Abstract
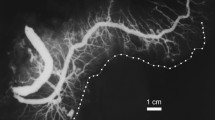
The salivary glands and pancreas have comparable anatomic and antigenic properties and can share common pathogenetic mechanisms involving toxic or autoimmune processes. The aim of this study is to assess the correlation in size between the normal submandibular glands and the pancreas. The study was based on human autopsy specimens of the pancreas, neck and oral base from 22 adults, both sexes (mean age, 57.9 years). The pancreatic and submandibular ducts were injected with a contrast medium, and the area of the salivary and pancreatic glandular ductograms was measured with the aid of software for quantification of visual information. Samples of tissue from the salivary glands and the pancreas were studied by means of light microscopy. A high correlation was found between the planimetric size of the pancreas and the submandibular glands (correlation coefficient 0.497 and 0.699 for the right and the left gland, respectively). This ratio was close to 5:1. There were no significant differences in size for the left vs. right submandibular gland (p = 0.39). The ductograms were significantly larger in size in males than in females (p < 0.001). This study has proven a positive correlation in planimetric size between the normal submandibular glands and pancreas, a result that is expected to have possible clinical implications in the long-term follow-up of patients with chronic pancreatitis.


Similar content being viewed by others
References
Bohl L, Merlo C, Carda C, Gómez De Ferraris ME, Carranza M (2008) Morphometric analysis of the parotid gland affected by alcoholic sialosis. J Oral Pathol Med 37:499–503. doi:10.1111/j.1600-0714.2008.00648.x
Bolondi L, Li Bassi S, Gaiani S, Barbara L (1989) Sonography of chronic pancreatitis. Radiol Clin North Am 27:815–833 (PMID: 2657858)
Carda C, Gomez de Ferraris ME, Arriaga A, Carranza M, Peydró A (2004) Alcoholic parotid sialosis: a structural and ultrastructural study. Med Oral 9:24–32 (PMID: 14704614)
Carranza M, Ferraris ME, Galizzi M (2005) Structural and morphometrical study in glandular parenchyma from alcoholic sialosis. J Oral Pathol Med 34:374–379. doi:10.1111/j.1600-0714.2005.00281.x
Dehghani SN, Tadjalli M, Masoumzadeh MH (2000) Sialography of sheep parotid and mandibular salivary glands. Res Vet Sci 68:3–7. doi:10.1053/rvsc.1999.0318
Delporte C (2014) Aquaporins in salivary glands and pancreas. Biochim Biophys Acta 1840:1524–1532. doi:10.1016/j.bbagen.2013.08.007
Djuric-Stefanovic A, Masulovic D, Kostic J, Randjic K, Saranovic D (2012) CT volumetry of normal pancreas: correlation with the pancreatic diameters measurable by the cross-sectional imaging, and relationship with the gender, age, and body constitution. Surg Radiol Anat 34:811–817. doi:10.1007/s00276-012-0962-7
Dutta SK, Dukehart M, Narang A, Latham PS (1989) Functional and structural changes in parotid glands of alcoholic cirrhotic patients. Gastroenterology 96:510–518 (PMID: 2910764)
Frulloni L, Morana G, Bovo P et al (1999) Salivary gland involvement in patients with chronic pancreatitis. Pancreas 19:33–38 (PMID: 10416689)
Fukui T, Okazaki K, Yoshizawa H et al (2005) A case of autoimmune pancreatitis associated with sclerosing cholangitis, retroperitoneal fibrosis and Sjögren’s syndrome. Pancreatology 5:86–91. doi:10.1159/000084494
Gorjup E, Danner S, Rotter N et al (2009) Glandular tissue from human pancreas and salivary gland yields similar stem cell populations. Eur J Cell Biol 88:409–421. doi:10.1016/j.ejcb.2009.02.187
Hayakawa T, Naruse S, Kitagawa M, Kondo T (2001) Clinical aspects of autoimmune pancreatitis in Sjogren’s syndrome. JOP 2:88–92 (PMID: 11870329)
Heo MS, Lee SC, Lee SS, Choi HM, Choi SC, Park TW (2001) Quantitative analysis of normal major salivary glands using computed tomography. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 92:240–244. doi:10.1067/moe.2001.114756
Hosoyamada Y, Sakai T (2003) The ultrastructure of periductal connective tissue and distinctive populations of collagen fibrils associated with ductal epithelia of exocrine glands. Arch Histol Cytol 66:407–418. doi:10.1679/aohc.66.407
Kalinowski M, Heverhagen JT, Rehberg E, Klose KJ, Wagner HJ (2002) Comparative study of MR sialography and digital subtraction sialography for benign salivary gland disorders. AJNR Am J Neuroradiol 23:1485–1492 (PMID: 12372736)
Kamisawa T, Tu Y, Egawa N, Sakaki N, Inokuma S, Kamata N (2003) Salivary gland involvement in chronic pancreatitis of various etiologies. Am J Gastroenterol 98:323–326. doi:10.1111/j.1572-0241.2003.07230.x
Metzger ED, Levine JM, Mcardle CR, Wolfe BE, Jimerson DC (1999) Salivary gland enlargement and elevated serum amylase in bulimia nervosa. Biol Psychiatry 45:1520–1522. doi:10.1016/S0006-3223(98)00221-2
Morimoto Y, Tanaka T, Yoshioka I, Masumi S, Yamashita M, Ohba T (2002) Virtual endoscopic view of salivary gland ducts using MR sialography data from three dimension fast asymmetric spin-echo (3D-FASE) sequences: a preliminary study. Oral Dis 8:268–274. doi:10.1034/j.1601-0825.2002.01819.x
Rakonczay Z Jr, Vág J, Földes A et al (2014) Chronic inflammation in the pancreas and salivary glands—lessons from similarities and differences in pathophysiology and treatment modalities. Curr Pharm Des 20:1104–1120. doi:10.2174/13816128113199990415
Sagatelian MA, Fravel J, Gallo SH, Makk LJ, Looney SW, Wright RA (1998) Do parotid duct abnormalities occur in patients with chronic alcoholic pancreatitis? Am J Gastroenterol 93:197–200 (PMID: 9468241)
Schrader H, Menge BA, Schneider S, Belyaev O, Tannapfel A, Uhl W et al (2009) Reduced pancreatic volume and beta-cell area in patients with chronic pancreatitis. Gastroenterology 136:513–522. doi:10.1053/j.gastro.2008.10.083
Stimec B, Bulajic M, Ugljesic M (2000) Computerized planimetry of normal and abnormal postmortem pancreatograms. Ann Anat 182:479–482. doi:10.1016/S0940-9602(00)80063-1
Stimec BV, Draskic M, Fasel JH (2010) Cadaver procurement for anatomy teaching: legislative challenges in a transition-related environment. Med Sci Law 50:45–49. doi:10.1258/msl.2009.009008
Tylenda CA, Ship JA, Fox PC, Baum BJ (1988) Evaluation of submandibular salivary flow rate in different age groups. J Dent Res 67:1225–1228. doi:10.1177/00220345880670091501
Udupa JK, Odhner D, Zhao L et al (2014) Body-wide hierarchical fuzzy modeling, recognition, and delineation of anatomy in medical images. Med Image Anal 18:752–771. doi:10.1016/j.media.2014.04.003
Varghese JC, Thornton F, Lucey BC, Walsh M, Farrell MA, Lee MJ (1999) A prospective comparative study of MR sialography and conventional sialography of salivary duct disease. AJR Am J Roentgenol 173:1497–1503. doi:10.2214/ajr.173.6.10584790
Weissman JL (1995) Imaging of the salivary glands. Semin Ultrasound CT MR 16:546–568 (PMID: 8747417)
World Medical Association (2008) Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects. 59th WMA General Assembly, Seoul, October 2008 (accessed Oct 03, 2016). Available from: http://www.wma.net/en/30publications/10policies/b3/
Yousem DM, Kraut MA, Chalian AA (2000) Major salivary gland imaging. Radiology 216:19–29. doi:10.1148/radiology.216.1.r00jl4519
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest, either primary or secondary.
Rights and permissions
About this article
Cite this article
Stimec, B.V., Rakocevic, Z., Ignjatovic, D. et al. Planimetric correlation between the submandibular glands and the pancreas: a postmortem ductographic study. Anat Sci Int 93, 114–118 (2018). https://doi.org/10.1007/s12565-016-0382-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-016-0382-6