Abstract
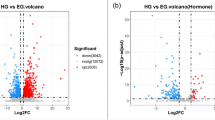
The progress of virology research in shrimp has been hampered by a lack of immortalized shrimp cell lines, and even long-term and stable primary cell cultures have not been perfectly achieved. Here, transcriptomic analysis of primary lymphoid cells in kuruma shrimp Marsupenaeus japonicus was performed to understand gene expression changes and stressors under existing culture conditions. In our optimized culture, the primary cells showed dead cell increases on days 3–4 and 6–7 after seeding. Next, we performed transcriptomic analysis at four time points (days 1, 3, 4, and 6 after tissue seeding), and RNA sequencing revealed that detected genes could be divided into three groups according to their expression changes: (1) downregulated on days 3–6, (2) upregulated on days 3–4, and (3) upregulated on day 6. Group 1 included platelet-derived growth factor receptor alpha and vascular endothelial growth factor 3 (MjVEGF3). Consistently, quantitative real-time polymerase chain reaction (qPCR) results showed a significant decrease in MjVEGF3 and its receptor (MjVEGFR) expression. These results suggest the importance of supplementation to recover VEGF signaling in the proper time-dependent manner for long and stable culture of shrimp lymphoid organ cells.





Similar content being viewed by others
References
Andrews S (2010) FastQC: a quality control tool for high throughput sequence data. http://www.bioinformatics.babraham.ac.uk/projects/fastqc
Anggraeni MS, Owens L (2000) The haemocytic origin of lymphoid organ spheroid cells in the penaeid prawn Penaeus monodon. Dis Aquat Organ 40:85–92
Anoop BS, Puthumana J, Vazhappilly CG, Kombiyil S, Philip R, Abdulaziz A, Bright Singh ISB (2021) Immortalization of shrimp lymphoid cells by hybridizing with the continuous cell line Sf9 leading to the development of “PmLyO-Sf9”. Fish Shellfish Immunol 113:196–207
Bautch VL (2012) VEGF-directed blood vessel patterning: from cells to organism. Cold Spring Harb Perspect Med 2:a006452
Chang PS, Chen HC, Wang YC (1998) Detection of white spot syndrome associated baculovirus (WSBV) in experimentally infected wild shrimps, crabs and lobsters by in situ hybridization. Aquaculture 164:23–47
Chen SN, Wang CS (1999) Establishment of cell culture systems from penaeid shrimp and their susceptibility to white spot disease and yellow head viruses. Methods Cell Sci 21:199–206
Chen SN, Chi SC, Kou GH, Liao IC (1986) Cell culture from tissues of grass prawn, Penaeus monodon. Fish Pathol 21:161–166
Chen X, Chen P, Wu D (1997) Study on a new bacilliform virus in cultured shrimps. Sci China (ser c) 27:415–420
Claydon K, Owens L (2008) Attempts at immortalization of crustacean primary cell cultures using human cancer genes. In Vitro Cell Dev Biol Anim 44:451–457
Claydon K, Tahir RAH, Said HM, Lakim MH, Tamat W (2010) Prevalence of shrimp viruses in wild Penaeus monodon from Brunei Darussalam. Aquaculture 308:71–74
da Huang W, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4:44–57
FAO (2020) FAO Yearbook Fishery and aquaculture statistics 2019. https://doi.org/10.4060/cb7874t
Flegel TW (2012) Historic emergence, impact and current status of shrimp pathogens in Asia. J Invertebr Pathol 110:166–173
Haigh JJ (2008) Role of VEGF in organogenesis. Organogenesis 4:247–256
Han Q, Li P, Lu X, Guo Z, Guo H (2013) Improved primary cell culture and subculture of lymphoid organs of the greasyback shrimp Metapenaeus ensis. Aquaculture 410–411:101–113
Hasson KW, Lightner DV, Mohney LL, Redman RM, White BM (1999) Role of lymphoid organ spheroids in chronic Taura syndrome virus (TSV) infections in Penaeus vannamei. Dis Aquat Organ 38:93–105
Itami T, Maeda M, Kondo M, Takahashi Y (1999) Primary culture of lymphoid organ cells and hemocytes of kuruma shrimp, Penaeus japonicus. Methods Cell Sci 21:237–244
Jayesh P, Seena J, Singh ISB (2012) Establishment of shrimp cell lines: perception and orientation. Indian J Virol 23:244–251
Jin Q, Zhu F (2019) Differentially expressed genes of the shrimp Marsupenaeus japonicus in response to infection by white spot syndrome virus or Vibrio alginolyticus. Fish Shellfish Immunol 92:348–355
Johnson KN, Van Hulten MCW, Barnes AC (2008) ‘Vaccination’ of shrimp against viral pathogens: phenomenology and underlying mechanisms. Vaccine 26:4885–4892
Kampen KR, Ter Elst A, De Bont ES (2013) Vascular endothelial growth factor signaling in acute myeloid leukemia. Cell Mol Life Sci 70:1307–1317
Li S, Wang Z, Li F, Yu K, Xiang J (2017) A novel vascular endothelial growth factor receptor participates in white spot syndrome virus infection in Litopenaeus vannamei. Front Immunol 8:1457
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550
Luedeman RA, Lightner DV (1992) Development of an in vitro primary cell culture system from the penaeid shrimp, Penaeus stylirostris and Penaeus vannamei. Aquaculture 101:205–211
Ma J, Zeng L, Lu Y (2017) Penaeid shrimp cell culture and its applications. Rev Aquacult 9:88–98
Martin GG, Hose JE, Minka G, Rosenberg S (1996) Clearance of bacteria injected into the hemolymph of the ridge back prawn, Sicyonia ingentis (Crustacea: Decapoda): role of hematopoietic tissue. J Morphol 227:227–233
Nör JE, Christensen J, Mooney DJ, Polverini PJ (1999) Vascular endothelial growth factor (VEGF)-mediated angiogenesis is associated with enhanced endothelial cell survival and induction of Bcl-2 expression. Am J Pathol 154:375–384
Owens L, Smith J (1999) Early attempts at production of prawn cell lines. Methods Cell Sci 21:207–212
Puthumana J, Prabhakaran P, Philip R, Singh ISB (2015) Attempts on producing lymphoid cell line from Penaeus monodon by induction with SV40-T and 12S EIA oncogenes. Fish Shellfish Immunol 47:655–663
R Core Team (2019) A language and environment for statistical computing. R Foundation for Statistical Computing
Rinkevich B (1999) Cell cultures from marine invertebrates: obstacles, new approaches and recent improvements. J Biotechnol 70:133–153
Rusaini OL, Owens L (2010) Insight into the lymphoid organ of penaeid prawns: a review. Fish Shellfish Immunol 29:367–377
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504
Sivakumar S, Swaminathan TR, Anandan R, Kalaimani N (2019) Medium optimization and characterization of cell culture system from Penaeus vannamei for adaptation of white spot syndrome virus (WSSV). J Virol Methods 270:38–45
Tong SL, Miao H-Z (1996) Attempts to initiate cell cultures from Penaeus chinensis tissues. Aquaculture 147:151–157
Van de Braak CBT, Botterblom MHA, Taverne N, Van Muiswinkel WB, Rombout JHWM, Van der Knaap WPW (2002) The roles of hemocytes and the lymphoid organ in the clearance of injected Vibrio bacteria in Penaeus monodon shrimp. Fish Shellfish Immunol 13:293–309
Wang CH, Yang HN, Tang CY, Lu CH, Kou GH, Lo CF (2000) Ultrastructure of white spot syndrome virus development in primary lymphoid organ cell cultures. Dis Aquat Organ 41:91–104
Wang Z, Li S, Li F, Xie S, Xiang J (2016) Identification and function analysis of a novel vascular endothelial growth factor, LvVEGF3, in the Pacific whiteleg shrimp Litopenaeus vannamei. Dev Comp Immunol 63:111–120
Yang F, He J, Lin X, Li Q, Pan D, Zhang X, Xu X (2001) Complete genome sequence of the shrimp white spot bacilliform virus. J Virol 75:11811–11820
Zhan WB, Wang YH, Fryer JL, Yu KK, Fukuda H, Meng QX (1998) White spot syndrome virus infection of cultured shrimp in China. J Aquat Anim Health 10:405–410
Acknowledgements
We would like to thank Editage (www.editage.jp) for English language editing.
Funding
This work was supported by a Grant-in-Aid for Challenging Research (Pioneering) (KAKENHI grant no. 20K20299) from the Japan Society for the Promotion of Science, Japan.
Author information
Authors and Affiliations
Contributions
Conceptualization: AT, YO, TK, JH, and MS. Project administration: AT, YO, JH, and MS. Supervision: JH and MS. Writing–original draft: AT, YO and JH. Writing–review and editing: TK, MS, and JH. Funding acquisition: MS.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tsuchiya, A., Okamura, Y., Kono, T. et al. Transcriptomic analysis of cultured kuruma shrimp primary lymphoid organ cells for identifying key cascades hindering sustainable culture. Fish Sci 89, 449–462 (2023). https://doi.org/10.1007/s12562-023-01692-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-023-01692-8