Abstract
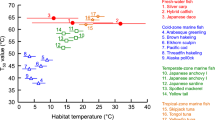
Complementary DNA encoding a part of myosin heavy chain was cloned from fast skeletal muscle of Japanese codling Physiculus japonicus. Japanese codling, a frequent catch off the Pacific coast of northeastern Japan, is a typical underutilized fish species, especially in summer. Phylogenetic analysis of the deduced amino acid sequence of Japanese codling myosin heavy chain loop 2 region revealed that this fish belongs to a group comprising white croaker Pennahia argentata and walleye pollack Gadus chalcogrammus, which are commonly used as raw materials for surimi-based products. Thus Japanese codling meat was considered a promising candidate ingredient for surimi-based products. Salt-ground meat was prepared from Japanese codling in the presence of 0.5 M NaCl and subjected to a two-step heating procedure: pre-heating at 15–70 °C for 30 min, and secondary heating at 85 °C for 20 min. Thermal gels with a maximum breaking strength of > 10 N were produced by this process. The breaking strength was comparable to that of white croaker and walleye pollack. On the basis of the phylogenetic analysis of myosin heavy chains, and the properties of the heat-induced surimi gel, we conclude that Japanese codling is a promising raw material for the production of surimi-based products.







Similar content being viewed by others
References
Chow S, Yanagimoto T, Matsuzaki K, Kofuji K, Hoshino K (2019) Little genetic difference between controversial Japanese codling species Physiculus japonicus and P. maximowiczi. Aquat Anim 2019:1–9
Fukushima H, Yoon SH, Watabe S (2003a) Differences in polymer formation through disulfide bonding of recombinant light meromyosin. J Agric Food Chem 51:4089–4095
Fukushima H, Satoh Y, Nakaya M, Ishizaki S, Watabe S (2003b) Thermal effects on fast skeletal myosins from walleye pollack, white croaker and rabbit in relation to gel formation. J Food Sci 68:1573–1577
Fukushima H, Satoh Y, Yoon SH, Togashi M, Nakaya M, Watabe S (2005) Rheological properties of fast skeletal myosin rod and light meromyosin from walleye pollack and white croaker: contribution of myosin fragments to thermal gel formation. J Agric Food Chem 53(9193–9198):9193
Goodson HV, Warrick HM, Spudich JA (1999) Specialized conservation of surface loops of myosin: evidence that loops are involved in determining functional characteristics. J Mol Biol 287:173–185
Hirayama Y, Watabe S (1997) Structural differences in the crossbridge head of temperature-associated myosin subfragment-1 isoforms from carp fast skeletal muscle. Eur J Biochem 246:380–387
Hossain MA, Ikeda D, Nomura A, Fukushima H, Watabe S (2008) cDNA cloning and complete primary structures of myosin heavy chains from brushtooth lizardfish (Sauridaundosquamis) and wanieso lizardfish (S. wanieso) fast skeletal muscles. Fish Sci 74:921–934
Ikeda D, Clark MS, Liang CS, Snell P, Edwards YJK, Elgar G, Watabe S (2004) Genomic structural analysis of the pufferfish (Takifugu rubripes) skeletal myosin heavy chain genes. Mar Biotechnol 6:S462–S467
Ikeda D, Ono Y, Snell P, Edwards YJK, Elgar G, Watabe S (2007) Divergent evolution of the myosin heavy chain gene family in fish and tetrapods; evidence from comparative genomic analysis. Physiol Genomics 32:1–15
Ikeda D, Nihei Y, Ono Y, Watabe S (2010) Three embryonic myosin heavy chain genes encoding different motor domain structures from common carp show distinct expression patterns in cranial muscles. Mar Genomics 3:1–9
Ikeguchi K, Kaneko G, Watabe S (2012) cDNA cloning and molecular characterization of bluefin tuna Thunnus orientalis transglutaminase. Fish Sci 78:667–674
Imai J, Hirayama Y, Kikuchi K, Kakinuma M, Watabe S (1997) cDNA cloning of myosin heavy chain isoforms from carp fast skeletal muscle and their gene expression associated with temperature acclimation. J Exp Biol 200:27–34
Iwami Y, Ojima T, Inoue A, Nishita K (2002) Primary structure of myosin heavy chain from fast skeletal muscle of chum salmon Oncorhynchus keta. Comp Biochem Physiol B Biochem Mol Biol 133:257–267
Kato N, Hashimoto A, Nozaki H, Arai K (1984) Effect of temperature on the rate for the setting of meat pastes from Alaska pollack, white croaker and tilapia. Bull Jpn Soc Sci Fish 50:2103–2108
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Matsuoka Y, Wan J, Ushio H, Watabe S (2013) Thermal gelation properties of white croaker, walleye pollack and deepsea bonefish surimi after suwari treatment at various temperatures. Fish Sci 79:715–724
Muramatsu-Uno M, Kikuchi K, Suetake H, Watabe S (2005) The complete nucleotide sequence of carp myosin heavy chain gene. Gene 349:143–151
Niwa E, Kanoh S, Nakayama T (1986) Function of salt in the setting of fish flesh sol. Nippon Suisan Gakkaishi 60:279–285
Nozawa H, Mamegoshi S, Seki N (1997) Partial purification and characterization of six transglutaminases from ordinary muscles of various fishes and marine invertebrates. Comp Biochem Physiol B Biochem Mol Biol 118:313–317
Sano K, Nakanishi K, Nakamura N, Motoki M, Yasueda H (1996) Cloning and sequence analysis of a cDNA encoding salmon (Onchorhynchus keta) liver transglutaminase. Biosci Biotechnol Biochem 60:1790–1794
Satoh Y, Nakaya M, Ochiai Y, Watabe S (2006) Enzymatic and structural properties of white croaker fast skeletal myosins in comparison of that from walleye pollack. Fish Sci 72:646–655
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Frovenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 19:76–85
Spudich JA (1994) How molecular motors work. Nature 372:515–518
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Togashi M, Kakinuma M, Hirayama Y, Fukushima H, Watabe S, Ojima T, Nishita K (2000) cDNA cloning of myosin rod and complete primary structure of myosin heavy chain of walleye pollack fast skeletal muscle. Fish Sci 66:349–357
Wan J, Kimura I, Seki N (1995) Inhibitory factors of transglutaminase in salted salmon meat paste. Fish Sci 61:968–972
Watabe S (2002) Temperature plasticity of contractile proteins in fish muscle. J Exp Biol 205:2231–2236
Watabe S, Ikeda D (2006) Diversity of the pufferfish Takifugu rubripes fast skeletal myosin heavy chain genes. Comp Biochem Physiol Part D 1:28–34
Watabe S, Imai J, Nakaya M, Hirayama Y, Okamoto Y, Masaki H, Uozumi T, Hirono I, Aoki T (1995) Temperature acclimation induces light meromyosin isoforms with different primary structures in carp fast skeletal muscle. Biochem Biophys Res Commun 208:118–125
Weber K, Osborn M (1969) The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem 244:4406–4412
Yoon SH, Kakinuma M, Hirayama Y, Yamamoto T, Watabe S (2000) cDNA cloning and characterization of the complete primary structure for myosin heavy chain of white croaker fast skeletal muscle. Fish Sci 66:1158–1162
Acknowledgments
This study was partly supported by Sanriku Recovery Fund, a fund for Private University from the Ministry of Education, Science, Culture and Sports of Japan, the President Fund of Kitasato University, and a Kitasato University Research Grant for Young Researchers. Mr. Makoto Endo, captain of the Sakae-maru, is also acknowledged for collecting the Japanese codling in Okkirai Bay, Iwate Prefecture, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Watabe, S., Ikeda, D., Mashiro, T. et al. Suitability of Japanese codling as a raw material for surimi-based products revealed by primary sequence analysis of myosin heavy chain and thermal gel properties. Fish Sci 86, 711–719 (2020). https://doi.org/10.1007/s12562-020-01430-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-020-01430-4