Abstract
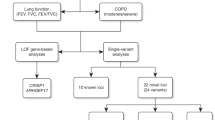
Rich collections of genomic and epigenomic annotations, availabilities of large population cohorts for genome-wide association studies (GWASs), and advancements in data integration techniques provide the unprecedented opportunity to accelerate discoveries in complex disease studies through integrative analyses. In this paper, we apply a variety of approaches to integrate GWAS summary statistics of chronic obstructive pulmonary disease (COPD) with functional annotations to illustrate how data integration could help researchers understand complex human diseases. We show that incorporating functional annotations can better prioritize GWAS signals at both the global and the local levels. Signal prioritization on severe COPD GWAS reveals multiple potential risk loci that are linked with pulmonary functions. Enrichment analysis provides novel insights on the pathogenesis of COPD and hints the existence of genetic contributions to muscle dysfunction and chronic lung inflammation, two symptoms that are often comorbid with COPD. Our results suggest that rich signals for COPD genetics are still buried under the Bonferroni-corrected genome-wide significance threshold. Many more biological findings are expected to emerge as more samples are recruited for COPD studies.




Similar content being viewed by others
References
Abecasis GR et al. (2012) An integrated map of genetic variation from 1,092 human genomes Nature 491:56–65. doi:10.1038/nature11632
Artigas MS et al (2011) Genome-wide association and large-scale follow up identifies 16 new loci influencing lung function. Nat Genet 43:1082–1090
Baekvad-Hansen M, Nordestgaard BG, Tybjaerg-Hansen A, Dahl M (2010) Two novel mutations in surfactant protein-C, lung function and obstructive lung disease. Respir Med 104:418–425. doi:10.1016/j.rmed.2009.10.012
Bernstein BE, Birney E, Dunham I, Green ED, Gunter C, Snyder M (2012) An integrated encyclopedia of DNA elements in the human genome. Nature 489:57–74. doi:10.1038/nature11247
Bhat TA, Panzica L, Kalathil SG, Thanavala Y (2015) Immune dysfunction in patients with chronic obstructive pulmonary disease. Ann Am Thorac Soc 12:S169–S175. doi:10.1513/AnnalsATS.201503-126AW
Bulik-Sullivan B et al (2015) An atlas of genetic correlations across human diseases and traits. Nat Genet 47:1236–1241. doi:10.1038/ng.3406
Casaburi R (2001) Skeletal muscle dysfunction in chronic obstructive pulmonary disease. Med Sci Sports Exerc 33:S662–S670
Cho MH et al (2010) Variants in FAM13A are associated with chronic obstructive pulmonary disease. Nat Genet 42:200–202. doi:10.1038/ng.535
Cho MH et al (2012) A genome-wide association study of COPD identifies a susceptibility locus on chromosome 19q13. Hum Mol Genet 21:947–957
Cho MH et al (2014) Risk loci for chronic obstructive pulmonary disease: a genome-wide association study and meta-analysis. Lancet Respir Med 2:214–225. doi:10.1016/s2213-2600(14)70002-5
Chung D, Yang C, Li C, Gelernter J, Zhao H (2014) GPA: a statistical approach to prioritizing GWAS results by integrating pleiotropy and annotation. PLoS Genet 10:e1004787. doi:10.1371/journal.pgen.1004787
DeMeo DL et al (2009) Integration of genomic and genetic approaches implicates IREB2 as a COPD susceptibility gene. Am J Hum Genet 85:493–502
Finucane HK et al (2015) Partitioning heritability by functional annotation using genome-wide association summary statistics. Nat Genet 47(11):1228–1235
Gusev A et al (2014) Partitioning heritability of regulatory and cell-type-specific variants across 11 common diseases. Am J Hum Genet 95:535–552
Hancock DB et al (2010) Meta-analyses of genome-wide association studies identify multiple loci associated with pulmonary function. Nat Genet 42:45–52
Hindorff LA, Sethupathy P, Junkins HA, Ramos EM, Mehta JP, Collins FS, Manolio TA (2009) Potential etiologic and functional implications of genome-wide association loci for human diseases and traits. Proc Natl Acad Sci USA 106:9362–9367. doi:10.1073/pnas.0903103106
Ingebrigtsen T et al (2010) Genetic influences on chronic obstructive pulmonary disease—a twin study. Respir Med 104:1890–1895
Kellis M et al (2014) Defining functional DNA elements in the human genome. Proc Natl Acad Sci USA. doi:10.1073/pnas.1318948111
Kichaev G et al (2014) Integrating functional data to prioritize causal variants in statistical fine-mapping studies. PLoS Genet 10:e1004722. doi:10.1371/journal.pgen.1004722
Kim HC, Mofarrahi M, Hussain SN (2008) Skeletal muscle dysfunction in patients with chronic obstructive pulmonary disease. Int J Chronic Obstr Pulm Dis 3:637
Kundaje A et al (2015) Integrative analysis of 111 reference human epigenomes. Nature 518:317–330
Li C, Yang C, Gelernter J, Zhao H (2014) Improving genetic risk prediction by leveraging pleiotropy. Hum Genet 133:639–650
Lu Q, Hu Y, Sun J, Cheng Y, Cheung K-H, Zhao H (2015) A statistical framework to predict functional non-coding regions in the human genome through integrated analysis of annotation data. Sci Rep. doi:10.1038/srep10576
Lu Q, Powles RL, Wang Q, He BJ, Zhao H (2016a) Integrative tissue-specific functional annotations in the human genome provide novel insights on many complex traits and improve signal prioritization in genome wide association studies. PLoS Genet 12:e1005947. doi:10.1371/journal.pgen.1005947
Lu Q, Yao X, Hu Y, Zhao H (2016b) GenoWAP: GWAS signal prioritization through integrated analysis of genomic functional annotation. Bioinformatics 32:542–548. doi:10.1093/bioinformatics/btv610
Maier R et al (2015) Joint analysis of psychiatric disorders increases accuracy of risk prediction for schizophrenia, bipolar disorder, and major depressive disorder. Am J Hum Genet 96:283–294
Naghavi M et al (2015) Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 385:117–171
Pickrell JK (2014) Joint analysis of functional genomic data and genome-wide association studies of 18 human traits. Am J Hum Genet 94:559–573
Pickrell J, Berisa T, Segurel L, Tung JY, Hinds D (2015) Detection and interpretation of shared genetic influences on 40 human traits. bioRxiv. doi:10.1101/019885
Pillai SG et al (2009) A genome-wide association study in chronic obstructive pulmonary disease (COPD): identification of two major susceptibility loci. PLoS Genet 5:e1000421
Pruim RJ et al (2010) LocusZoom: regional visualization of genome-wide association scan results. Bioinformatics 26:2336–2337
Purcell S et al (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81:559–575
Regan EA et al (2011) Genetic epidemiology of COPD (COPDGene) study design. COPD J Chronic Obstr Pulm Dis 7:32–43
Repapi E et al (2010) Genome-wide association study identifies five loci associated with lung function. Nat Genet 42:36–44. doi:10.1038/ng.501
Rovina N, Koutsoukou A, Koulouris NG (2013) Inflammation and immune response in COPD: Where do we stand? Mediat Inflamm 2013:413735
Visscher PM, Brown MA, McCarthy MI, Yang J (2012) Five years of GWAS discovery. Am J Hum Genet 90:7–24. doi:10.1016/j.ajhg.2011.11.029
Ward LD, Kellis M (2012) Interpreting noncoding genetic variation in complex traits and human disease. Nat Biotechnol 30:1095–1106. doi:10.1038/nbt.2422
Welter D et al (2014) The NHGRI GWAS Catalog, a curated resource of SNP-trait associations. Nucleic Acids Res 42:D1001–D1006. doi:10.1093/nar/gkt1229
Wilk JB et al (2009) A genome-wide association study of pulmonary function measures in the Framingham Heart Study. PLoS Genet 5:e1000429
Wüst RC, Degens H (2007) Factors contributing to muscle wasting and dysfunction in COPD patients. Int J Chronic Obstr Pulm Dis 2:289
Xu K, Moghal N, Egan SE (2012) Notch signaling in lung development and disease. In: Notch signaling in embryology and cancer. Springer, US, pp 89–98
Yang Y-S, Yang M-CW, Guo Y, Williams OW, Weissler JC (2009) PLAGL2 expression-induced lung epithelium damages at bronchiolar alveolar duct junction in emphysema: bNip3-and SP-C-associated cell death/injury activity. Am J Physiol Lung Cell Mol Physiol 297:L455–L466
Zhou X et al (2012) Identification of a chronic obstructive pulmonary disease genetic determinant that regulates HHIP. Hum Mol Genet 21:1325–1335. doi:10.1093/hmg/ddr569
Zhou JJ, Cho MH, Castaldi PJ, Hersh CP, Silverman EK, Laird NM (2013) Heritability of chronic obstructive pulmonary disease and related phenotypes in smokers. Am J Respir Crit Care Med 188:941–947
Acknowledgments
We thank all members of the Statistical and Applied Mathematical Sciences Institute (SAMSI) Data Integration: COPD Working Group as part of the SAMSI Beyond Bioinformatics Program. We also thank Dr. Michael Cho for sharing COPDGene GWAS summary statistics. We are grateful for the support of Drs. Sujit Ghosh and Snehalata Huzurbazar at SAMSI. This work was supported in part by the National Institutes of Health Grant R01 GM59507 and the VA Cooperative Studies Program of the Department of Veterans Affairs, Office of Research and Development. Funding for the COPDGene Study is provided by the National Heart, Lung, and Blood Institute Award Numbers R01HL089897 and R01HL089856. Further support provided by the COPD Foundation through contributions made to an Industry Advisory Board comprising AstraZeneca, Boehringer Ingelheim, Novartis Pfizer, Siemens, and Sunovion.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lu, Q., Jin, C., Sun, J. et al. Post-GWAS Prioritization Through Data Integration Provides Novel Insights on Chronic Obstructive Pulmonary Disease. Stat Biosci 9, 605–621 (2017). https://doi.org/10.1007/s12561-016-9151-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12561-016-9151-2