Abstract
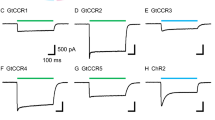
Optogenetics is a growing technique which allows manipulation of biological events simply by illumination. The technique is appreciated especially in the neuroscience field because of its availability in controlling neuronal functions. A light-gated cation channel, Cr_ChR2 from Chlamydomonas reinhardtii, is the first and mostly applied to optogenetics for activating neuronal excitability. In addition, the molecular mechanism of Cr_ChR2 has been intensively studied by electrophysiology, spectroscopy, X-ray structural studies, etc. Novel cation channelrhodopsins from Guillardia theta, namely, Gt_CCR1–4, were discovered in 2016 and 2017. These channelrhodopsins are more homologous to haloarchaeal rhodopsins, particularly the proton pumps. Thus these cryptophyte-type light-gated cation channels are structurally and mechanistically distinct from chlorophyte channelrhodopsin such as Cr_ChR2. We here compared the photocurrent properties, cation selectivity, and kinetics between well-known Cr_ChR2 and Gt_CCR4. The light sensitivity of Gt_CCR4 is significantly higher than that of Cr_ChR2, while the channel open lifetime is in the same range as that of Cr_ChR2. Gt_CCR4 shows high Na+ selectivity in which the selectivity ratio for Na+ was 37-fold larger than that for Cr_ChR2, which primarily conducts H+. On the other hand, Gt_CCR4 conducted almost no H+ and no Ca2+ under physiological conditions. Other unique features and the applicability of Gt_CCR4 for optogenetics were discussed.



Similar content being viewed by others
References
Berndt A, Yizhar O, Gunaydin LA et al (2009) Bi-stable neural state switches. Nat Neurosci 12:229–234. https://doi.org/10.1038/nn.2247
Boyden ES, Zhang F, Bamberg E et al (2005) Millisecond-timescale, genetically targeted optical control of neural activity. Nat Neurosci 8:1263–1268. https://doi.org/10.1038/nn1525
Chow BY, Han X, Dobry AS et al (2010) High-performance genetically targetable optical neural silencing by light-driven proton pumps. Nature 463:98–102. https://doi.org/10.1038/nature08652
Curtis BA, Tanifuji G, Maruyama S et al (2012) Algal genomes reveal evolutionary mosaicism and the fate of nucleomorphs. Nature 492:59–65. https://doi.org/10.1038/nature11681
Dawydow A, Gueta R, Ljaschenko D et al (2014) Channelrhodopsin-2-XXL, a powerful optogenetic tool for low-light applications. Proc Natl Acad Sci U S A 111:13972–13977. https://doi.org/10.1073/pnas.1408269111
Ernst OP, Lodowski DT, Elstner M et al (2014) Microbial and animal rhodopsins: structures, functions, and molecular mechanisms. Chem Rev 114:126–163. https://doi.org/10.1021/cr4003769
Govorunova EG, Sineshchekov OA, Li H et al (2013) Characterization of a highly efficient blue-shifted channelrhodopsin from the marine alga Platymonas subcordiformis. J Biol Chem 288:29911–29922. https://doi.org/10.1074/jbc.M113.505495
Govorunova EG, Sineshchekov OA, Janz R et al (2015) Neuroscience. Natural light-gated anion channels: a family of microbial rhodopsins for advanced optogenetics. Science (80- ) 349:647–650. https://doi.org/10.1126/science.aaa7484
Govorunova EG, Sineshchekov OA, Spudich JL (2016) Structurally distinct cation channelrhodopsins from cryptophyte algae. Biophys J 110:2302–2304. https://doi.org/10.1016/j.bpj.2016.05.001
Govorunova EG, Sineshchekov OA, Li H, Spudich JL (2017) Microbial rhodopsins: diversity, mechanisms, and optogenetic applications. Annu Rev Biochem 86:845–872. https://doi.org/10.1146/annurev-biochem-101910-144233
Hille B (2001) Chapter 1 introduction. In: Ion Channels of Excitable Membranes
Hou SY, Govorunova EG, Ntefidou M et al (2012) Diversity of Chlamydomonas channelrhodopsins. Photochem Photobiol 88:119–128. https://doi.org/10.1111/j.1751-1097.2011.01027.x
Inoue K, Ono H, Abe-Yoshizumi R et al (2013) A light-driven sodium ion pump in marine bacteria. Nat Commun 4:1678. https://doi.org/10.1038/ncomms2689
Inoue K, Ito S, Kato Y et al (2016) A natural light-driven inward proton pump. Nat Commun 7:13415. https://doi.org/10.1038/ncomms13415
Ishizuka T, Kakuda M, Araki R, Yawo H (2006) Kinetic evaluation of photosensitivity in genetically engineered neurons expressing green algae light-gated channels. Neurosci Res 54:85–94. https://doi.org/10.1016/j.neures.2005.10.009
Ito S, Kato HE, Taniguchi R et al (2014) Water-containing hydrogen-bonding network in the active center of channelrhodopsin. J Am Chem Soc 136:3475–3482. https://doi.org/10.1021/ja410836g
Kandori H (2015) Ion-pumping microbial rhodopsins. Front Mol Biosci 2:52. https://doi.org/10.3389/fmolb.2015.00052
Kato HE, Zhang F, Yizhar O et al (2012) Crystal structure of the channelrhodopsin light-gated cation channel. Nature 482:369–374. https://doi.org/10.1038/nature10870
Klapoetke NC, Murata Y, Kim SS et al (2014) Independent optical excitation of distinct neural populations. Nat Methods 11:338–346. https://doi.org/10.1038/nmeth.2836
Kleinlogel S, Feldbauer K, Dempski RE et al (2011) Ultra light-sensitive and fast neuronal activation with the Ca2+−permeable channelrhodopsin catCh. Nat Neurosci 14:513–518. https://doi.org/10.1038/nn.2776
Lorenz-Fonfria VA, Resler T, Krause N et al (2013) Transient protonation changes in channelrhodopsin-2 and their relevance to channel gating. Proc Natl Acad Sci 110:E1273–E1281. https://doi.org/10.1073/pnas.1219502110
Matsuno-Yagi A, Mukohata Y (1977) Two possible roles of bacteriorhodopsin; a comparative study of strains of Halobacz’eril’m halobium differing in pigmentation. Biochem Biophys Res Commun 78:237–243
Nack M, Radu I, Gossing M et al (2010) The DC gate in channelrhodopsin-2: crucial hydrogen bonding interaction between C128 and D156. Photochem Photobiol Sci 9:194–198. https://doi.org/10.1039/b9pp00157c
Nagel G, Ollig D, Fuhrmann M et al (2002) Channelrhodopsin-1: a light-gated proton channel in green algae. Science (80- ) 296:2395–2398. https://doi.org/10.1126/science.1072068
Nagel G, Szellas T, Huhn W et al (2003) Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci U S A 100:13940–13945. https://doi.org/10.1073/pnas.1936192100
Oesterhelt D, Stoeckenius W (1971) Rhodopsin-like protein from the purple membrane of Halobacterium halobium. Nat New Biol 233:149–152. https://doi.org/10.1038/10.1038/newbio233149a0
Owen SF, Liu MH, Kreitzer AC (2019) Thermal constraints on in vivo optogenetic manipulations. Nat Neurosci 22:1061–1065. https://doi.org/10.1038/s41593-019-0422-3
Polovinkin V, Rodriguez-Valera F, Bueldt G et al (2017) Inward H + pump xenorhodopsin: mechanism and alternative optogenetic approach. Sci Adv 3:e1603187. https://doi.org/10.1126/sciadv.1603187
Prigge M, Schneider F, Tsunoda SP et al (2012) Color-tuned channelrhodopsins for multiwavelength optogenetics. J Biol Chem 287:31804–31812. https://doi.org/10.1074/jbc.M112.391185
Pushkarev A, Inoue K, Larom S et al (2018) Discovered Using Functional Metagenomics. Nature:1. https://doi.org/10.1038/s41586-018-0225-9
Ritter E, Stehfest K, Berndt A et al (2008) Monitoring light-induced structural changes of channelrhodopsin-2 by UV-visible and Fourier transform infrared spectroscopy. J Biol Chem 283:35033–35041. https://doi.org/10.1074/jbc.M806353200
Schneider F, Grimm C, Hegemann P (2015) Biophysics of channelrhodopsin. Annu Rev Biophys 44:167–186. https://doi.org/10.1146/annurev-biophys-060414-034014
Shigemura S, Hososhima S, Kandori H, Tsunoda SP (2019) Ion channel properties of a cation channelrhodopsin, Gt_CCR4. Appl Sci 9. https://doi.org/10.3390/app9173440
Sineshchekov OA, Govorunova EG, Li H, Spudich JL (2017) Bacteriorhodopsin-like channelrhodopsins: alternative mechanism for control of cation conductance. Proc Natl Acad Sci 114:E9512–E9519. https://doi.org/10.1073/pnas.1710702114
Sugiyama Y, Wang H, Hikima T et al (2009) Photocurrent attenuation by a single polar-to-nonpolar point mutation of channelrhodopsin-2. Photochem Photobiol Sci 8:328–336. https://doi.org/10.1039/b815762f
Tsunoda SP, Hegemann P (2009) Glu 87 of channelrhodopsin-1 causes pH-dependent color tuning and fast photocurrent inactivation. Photochemistry and Photobiology, In, pp 564–569
Vierock J, Grimm C, Nitzan N, Hegemann P (2017) Molecular determinants of proton selectivity and gating in the red-light activated channelrhodopsin Chrimson. Sci Rep 7:1–15. https://doi.org/10.1038/s41598-017-09600-8
Volkov O, Kovalev K, Polovinkin V et al (2017) Structural insights into ion conduction by channelrhodopsin 2. Science (80- ) 358. https://doi.org/10.1126/science.aan8862
Watanabe S, Ishizuka T, Hososhima S et al (2016) The regulatory mechanism of ion permeation through a channelrhodopsin derived from Mesostigma viride (MvChR1). Photochem Photobiol Sci 15:365–374. https://doi.org/10.1039/c5pp00290g
Wen L, Wang H, Tanimoto S et al (2010) Opto-current-clamp actuation of cortical neurons using a strategically designed channelrhodopsin. PLoS One. https://doi.org/10.1371/journal.pone.0012893
Wietek J, Wiegert JS, Adeishvili N et al (2014) Conversion of channelrhodopsin into a light-gated chloride channel. Science 344:409–412. https://doi.org/10.1126/science.1249375
Yamauchi Y, Konno M, Ito S et al (2017) Molecular properties of a DTD channelrhodopsin from Guillardia theta. Biophys Physicobiology 14:57–66. https://doi.org/10.2142/biophysico.14.0
Yizhar O, Fenno LE, Prigge M et al (2011) Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature 477:171–178. https://doi.org/10.1038/nature10360
Yoshizawa S, Kumagai Y, Kim H et al (2014) Functional characterization of flavobacteria rhodopsins reveals a unique class of light-driven chloride pump in bacteria. Proc Natl Acad Sci U S A 111:6732–6737. https://doi.org/10.1073/pnas.1403051111
Zhang F, Wang L-P, Brauner M et al (2007) Multimodal fast optical interrogation of neural circuitry. Nature 446:633–639. https://doi.org/10.1038/nature05744
Funding
This work was supported by the Japanese Ministry of Education, Culture, Sports, Science and Technology (25104009, 15H02391 to H.K and 18 K06109 to S.P.T.), a JST CREST grant (JPMJCR1753 to H.K.), and a JST PRESTO grant (JPMJPR1688 to S.P.T). S.H. is a Research Fellow of the Japan Society for the Promotion of Science (JSPS Research Fellow).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hososhima, S., Shigemura, S., Kandori, H. et al. Novel optogenetics tool: Gt_CCR4, a light-gated cation channel with high reactivity to weak light. Biophys Rev 12, 453–459 (2020). https://doi.org/10.1007/s12551-020-00676-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-020-00676-7