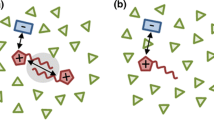
Abstract
Ionic liquids (ILs) are versatile solvents for a broad range of biotechnological applications. Recent experimental and simulation results highlight the potential benefits of dilute ILs in aqueous solution (aqueous ILs) in order to modify protein and DNA structures systematically. In contrast to a limited number of standard co-solutes like urea, ectoine, trimethylamine-N-oxide (TMAO), or guanidinium chloride, the large amount of possible cation and anion combinations in aqueous ILs can be used to develop tailor-made stabilizers or destabilizers for specific purposes. In this review article, we highlight common principles and differences between aqueous ILs and standard co-solutes with a specific focus on their underlying macromolecular stabilization or destabilization behavior. In combination with statistical thermodynamics theories, we present an efficient framework, which is used to classify structure modification effects consistently. The crucial importance of enthalpic and entropic contributions to the free energy change upon IL-assisted macromolecular unfolding in combination with a complex destabilization mechanism is described in detail. A special focus is also set on aqueous IL-DNA interactions, for which experimental and simulation outcomes are summarized and discussed in the context of previous findings.




Similar content being viewed by others
References
Ajloo D, Sangian M, Ghadamgahi M, Evini M, Saboury AA (2013) Effect of two imidazolium derivatives of ionic liquids on the structure and activity of adenosine deaminase. Int J Biol Macromol 55:47–61
Araújo J, Pereiro A, Alves F, Marrucho I, Rebelo L (2013) Nucleic acid bases in 1-alkyl-3-methylimidazolium acetate ionic liquids: a thermophysical and ionic conductivity analysis. J Chem Thermodyn 57:1–8
Atkins PW, de Paula J (2010) Physical chemistry. Oxford Univ Press, Oxford
Auton M, Bolen DW (2004) Additive transfer free energies of the peptide backbone unit that are independent of the model compound and the choice of concentration scale. Biochemistry 43(5):1329–1342
Baker SN, McCleskey TM, Pandey S, Baker GA (2004) Fluorescence studies of protein thermostability in ionic liquids. Chem Commun 8:940–941
Baker JL, Furbish J, Lindberg GE (2015) Influence of the ionic liquid [c4 mpy][tf2n] on the structure of the miniprotein trp-cage. J Mol Graph Model 62:202–212
Baynes BM, Trout BL (2003) Proteins in mixed solvents: a molecular-level perspective. J Phys Chem B 107(50):14058–14067
Ben-Naim A (2013) Statistical thermodynamics for chemists and biochemists. Springer Science & Business Media, Berlin
Benedetto A, Ballone P (2015) Room temperature ionic liquids meet biomolecules: a microscopic view of structure and dynamics. ACS Sustain Chem Eng 4(2):392–412
Bryngelson JD, Onuchic JN, Socci ND, Wolynes PG (1995) Funnels, pathways, and the energy landscape of protein folding: a synthesis. Prot Struct Funct Bioinf 21(3):167–195
Byrne N, Angell CA (2008) Protein unfolding, and the ‘tuning in’ of reversible intermediate states, in protic ionic liquid media. J Mol Biol 378(3):707–714
Canchi DR, García AE (2013a) Cosolvent effects on protein stability. Ann Rev Phys Chem 64:273–293
Canchi DR, García AE (2013b) Cosolvent effects on protein stability. Annu Rev Phys Chem 64:273–293
Cardoso L, Micaelo NM (2011) DNA molecular solvation in neat ionic liquids. Chemphyschem 12(2):275–277
Chandran A, Ghoshdastidar D, Senapati S (2012) Groove binding mechanism of ionic liquids: a key factor in long term stability of DNA in hydrated ionic liquids? J Am Chem Soc 134(50):20330–20339
Chen M, Xiong H, Wen W, Zhang X, Gu H, Wang S (2013) Electrochemical biosensors for the assay of DNA damage initiated by ferric ions catalyzed oxidation of dopamine in room temperature ionic liquid. Electrochim Acta 114:265–270
Cheng DH, Chen XW, Wang JH, Fang ZL (2007) An abnormal resonance light scattering arising from ionic-liquid/DNA/ethidium interactions. Chem Eur J 13(17):4833–4839
Chevrot G, Fileti EE, Chaban VV (2016) Protein remains stable at unusually high temperatures when solvated in aqueous mixtures of amino acid based ionic liquids. J Mol Model 22(11):258
Clark KD, Nacham O, Yu H, Li T, Yamsek MM, Ronning DR, Anderson JL (2015a) Extraction of DNA by magnetic ionic liquids: tunable solvents for rapid and selective DNA analysis. Anal Chem 87(3):1552–1559
Clark KD, Yamsek MM, Nacham O, Anderson JL (2015b) Magnetic ionic liquids as PCR-compatible solvents for dna extraction from biological samples. Chem Commun 51(94):16771–16773
Clark KD, Sorensen M, Nacham O, Anderson JL (2016) Preservation of DNA in nuclease-rich samples using magnetic ionic liquids. RSC Adv 6(46):39846–39851
Collins KD (2004) Ions from the hofmeister series and osmolytes: effects on proteins in solution and in the crystallization process. Methods 34(3):300–311
Constantinescu D, Weingärtner H, Herrmann C (2007) Protein denaturation by ionic liquids and the hofmeister series: a case study of aqueous solutions of ribonuclease a. Angew Chem Int Ed 46(46):8887–8889
Constatinescu D, Herrmann C, Weingärtner H (2010) Patterns of protein unfolding and protein aggregation in ionic liquids. Phys Chem Chem Phys 12(8):1756–1763
Courtenay E, Capp M, Anderson C, Record M (2000) Vapor pressure osmometry studies of osmolyte-protein interactions: implications for the action of osmoprotectants in vivo and for the interpretation of “osmotic stress” experiments in vitro. Biochemistry 39(15):4455–4471
Dabirmanesh B, Daneshjou S, Sepahi AA, Ranjbar B, Khavari-Nejad RA, Gill P, Heydari A, Khajeh K (2011) Effect of ionic liquids on the structure, stability and activity of two related \(\alpha \)-amylases. Int J Biol Macromol 48(1):93–97
Dandia A, Singh R, Saini D (2013) Ionic liquid-mediated three-component synthesis of fluorinated spiro-thiazine derivatives and their antimycobacterial and DNA cleavage activities. J Chem Sci 125(5):1045–1053
Diddens D, Lesch V, Heuer A, Smiatek J (2017) Aqueous ionic liquids and their influence on peptide conformations: denaturation and dehydration mechanisms. Phys Chem Chem Phys 19(31):20430–20440
Ding Y, Zhang L, Xie J, Guo R (2010) Binding characteristics and molecular mechanism of interaction between ionic liquid and DNA. J Phys Chem B 114(5):2033–2043
Dommert F, Wendler K, Berger R, Delle Site L, Holm C (2012) Force fields for studying the structure and dynamics of ionic liquids: a critical review of recent developments. ChemPhysChem 13(7):1625–1637
Egorova KS, Gordeev EG, Ananikov VP (2017) Biological activity of ionic liquids and their application in pharmaceutics and medicine. Chem Rev 117(10):7132–7189
Eisenberg H (1976) Biological macromolecules and polyelectrolytes in solution. Clarendon Press, Oxford
Figueiredo AM, Sardinha J, Moore GR, Cabrita EJ (2013) Protein destabilisation in ionic liquids: the role of preferential interactions in denaturation. Phys Chem Chem Phys 15(45):19632–19643
Fujita K, Ohno H (2010) Enzymatic activity and thermal stability of metallo proteins in hydrated ionic liquids. Biopolymers 93(12):1093–1099
Fujita K, Ohno H (2012) Stable G-quadruplex structure in a hydrated ion pair: cholinium cation and dihydrogen phosphate anion. Chem Commun 48:5751–5753
Fujita K, MacFarlane DR, Forsyth M (2005) Protein solubilising and stabilising ionic liquids. Chem Commun 38:4804–4806
Fujita K, Forsyth M, MD R, Reid RW, Elliott GD (2006) Unexpected improvement in stability and utility of cytochrome c by solution in biocompatible ionic liquids. Biotechnol Bioeng 94:1209–1213
Fukumoto K, Yoshizawa M, Ohno H (2005) Room temperature ionic liquids from 20 natural amino acids. J Am Chem Soc 127(8):2398–2399
Fyta M, Netz RR (2012) Ionic force field optimization based on single-ion and ion-pair solvation properties: going beyond standard mixing rules. J Chem Phys 136(12):124103
Gee MB, Cox NR, Jiao Y, Bentenitis N, Weerasinghe S, Smith PE (2011) A kirkwood-buff derived force field for aqueous alkali halides. J Chem Theory Comput 7(5):1369–1380
Ghaemi M, Absalan G (2014) Study on the adsorption of DNA on Fe3O4 nanoparticles and on ionic liquid-modified Fe3O4 nanoparticles. Microchim Acta 181(1-2):45–53
Haberler M, Steinhauser O (2012) On the influence of of hydrated ionic liquids on the dynamical structure of model proteins: a computational study. Phys Chem Chem Phys 13(40):17994–18004
Haberler M, Schröder C, Steinhauser O (2011) Solvation studies of a zinc finger protein in hydrated ionic liquids. Phys Chem Chem Phys 13(15):6955–6969
Haberler M, Schröder C, Steinhauser O (2012a) Hydrated ionic liquids with and without solute: the influence of water content and protein solutes. J Chem Theory Comput 8(10):3911–3928
Haberler M, Schröder C, Steinhauser O (2012b) Hydrated ionic liquids with and without solute: the influence of water content and protein solutes. J Chem Theo Comput 8(10):3911–3928
Hahn MB, Solomun T, Wellhausen R, Hermann S, Seitz H, Meyer S, Kunte HJ, Zeman J, Uhlig F, Smiatek J, Sturm H (2015) Influence of the compatible solute ectoine on the local water structure: implications for the binding of the protein g5p to dna. J Phys Chem B 119(49):15212–15220
Hahn MB, Uhlig F, Solomun T, Smiatek J, Sturm H (2016) Combined influence of ectoine and salt: spectroscopic and numerical evidence for compensating effects on aqueous solutions. Phys Chem Chem Phys 18(41):28398–28402
Hall D (1971) Kirkwood-buff theory of solutions. An alternative derivation of part of it and some applications. Transact Farad Soc 67:2516–2524
Haque A, Khan I, Hassan SI, Khan MS (2017) Interaction studies of cholinium-based ionic liquids with calf thymus DNA: spectrophotometric and computational methods. J Mol Liq 237:201–207
Harries D, Rösgen J (2008) A practical guide on how osmolytes modulate macromolecular properties. Methods Cell Biol 84:679–735
He Y, Shang Y, Liu Z, Shao S, Liu H, Hu Y (2013) Interactions between ionic liquid surfactant C(12)mim Br and DNA in dilute brine. Colloids Surf B 101:398–404
Heyda J, Okur HI, Hladílkova J, Rembert KB, Hunn W, Yang T, Dzubiella J, Jungwirth P, Cremer PS (2017) Guanidinium can both cause and prevent the hydrophobic collapse of biomacromolecules. J Am Chem Soc 139(2):863–870
Horinek D, Netz RR (2011) Can simulations quantitatively predict peptide transfer free energies to urea solutions? Thermodynamic concepts and force field limitations. J Phys Chem A 115(23):6125–6136
Jha SK, Marqusee S (2014) Kinetic evidence for a two-stage mechanism of protein denaturation by guanidinium chloride. Proc Natl Acad Sci USA 111(13):4856–4861
Jumbri K, Rahman MBA, Abdulmalek E, Ahmad H, Micaelo NM (2014) An insight into structure and stability of DNA in ionic liquids from molecular dynamics simulation and experimental studies. PCCP 16(27):14036–14046
Jumbri K, Ahmad H, Abdulmalek E, Rahman MBA (2016) Binding energy and biophysical properties of ionic liquid-DNA complex: understanding the role of hydrophobic interactions. J Mol Liq 223:1197–1203
Kanduċ M, Chudoba R, Palczynski K, Kim WK, Roa R, Dzubiella J (2017) Selective solute adsorption and partitioning around single pnipam chains. Phys Chem Chem Phys 19(8):5906–5916
Khandelwal G, Bhyravabhotla J (2010) A phenomenological model for predicting melting temperatures of DNA sequences. PLoS One 5(8):e12433
Kirkwood JG, Buff FP (1951) The statistical mechanical theory of solutions. I. J Chem Phys 19(6):774–777
Klähn M, Lim GS, Sedurama nA, Wu P (2011) On the different roles of anions and cations in th e solvation of enzymes in ionic liquids. Phys Chem Chem Phys 13(4):1649–1662
Kobayashi T, Reid JE, Shimizu S, Fyta M, Smiatek J (2017) The properties of residual water molecules in ionic liquids: a comparison between direct and inverse Kirkwood–Buff approaches. Phys Chem Chem Phys 19:18924–18937
Krishnamoorthy AN, Zeman J, Holm C, Smiatek J (2016) Preferential solvation and ion association properties in aqueous dimethyl sulfoxide solutions. Phys Chem Chem Phys 18(45):31312–31322
Krishnamoorthy AN, Holm C, Smiatek J (2018) The influence of co-solutes on the chemical equilibrium — a Kirkwood-Buff theory for ion pair association-dissociation processes in ternary electrolyte solutions. in revision
Kulkarni M, Mukherjee A (2016) Ionic liquid prolongs DNA translocation through graphene nanopores. RSC Adv 6(51):46019–46029
Kumar A, Venkatesu P (2013) Prevention of insulin self-aggregation by a protic ionic liquid. RSC Adv 3:362–367
Kumar P, Franzese G, Buldyrev SV, Stanley HE (2006) Molecular dynamics study of orientational cooperativity in water. Phys Rev E 73:0415051–0415058
Kumar P, Buldyrev SV, Stanley HE (2009) A tetrahedral entropy for water. Proc Natl Acad Sci USA 106:22130–22134
Kunz W (2010) Specific ion effects. World Scientific
Kusalik PG, Patey G (1987) The thermodynamic properties of electrolyte solutions: some formal results. J Chem Phys 86(9):5110–5116
Kusalik PG, Patey G (1988) On the molecular theory of aqueous electrolyte solutions. i. The solution of the rhnc approximation for models at finite concentration. J Chem Phys 88(12):7715–7738
Lesch V, Heuer A, Holm C, Smiatek J (2015a) Solvent effects of 1-ethyl-3-methylimidazolium acetate: solvation and dynamic behavior of polar and apolar solutes. Phys Chem Chem Phys 17(13):8480–8490
Lesch V, Heuer A, Tatsis VA, Holm C, Smiatek J (2015b) Peptides in the presence of aqueous ionic liquids: tunable co-solutes as denaturants or protectants? Phys Chem Chem Phys 17(39):26049–26053
Lesch V, Heuer A, Holm C, Smiatek J (2016) Properties of apolar solutes in alkyl imidazolium-based ionic liquids: the importance of local interactions. ChemPhysChem 17(3):387–394
Li T, Joshi MD, Ronning DR, Anderson JL (2013a) Ionic liquids as solvents for in situ dispersive liquid–liquid microextraction of DNA. J Chromatogr A 1272:8–14
Li X, Ma J, Lei W, Li J, Zhang Y, Li Y (2013b) Cloning of cytochrome P450 3A137 complementary DNA in silver carp and expression induction by ionic liquid. Chemosphere 92(9):1238–1244
Lo Nostro P, Ninham BW (2012) Hofmeister phenomena: an update on ion specificity in biology. Chem Rev 112(4):2286–2322
Machado I, Özalp VC, Rezabal E, Schäfer T (2014) DNA aptamers are functional molecular recognition sensors in protic ionic liquids. Chem Eur J 20(37):11820–11825
Mann JP, McCluskey A, Atkin R (2009) Activity and thermal stability of lysozyme in alkylammonium formate ionic liquids—influence of cation modification. Green Chem 11(6):785–792
Marcus Y (2009) Effect of ions on the structure of water: structure making and breaking. Chem Rev 109(3):1346–1370
Martinez L, Shimizu S (2017) Molecular interpretation of preferential interactions in protein solvation: a solvent-shell perspective by means of minimum-distance distribution functions. J Chem Theory Comput 13(12):6358–6372
Marušič M, Tateishi-Karimata H, Sugimoto N, Plavec J (2015) Structural foundation for DNA behavior in hydrated ionic liquid: an NMR study. Biochimie 108:169–177
Mazid RR, Cooper A, Zhang Y, Vijayaraghavan R, MacFarlane DR, Cortez-Jugo C, Cheng W (2015) Enhanced enzymatic degradation resistance of plasmid DNA in ionic liquids. RSC Adv 5(54):43839–43844
Micaêlo NM, Soares CM (2008) Protein structure and dynamics in ionic liquids. Insights from molecular dynamics simulation studies. J Phys Chem B 112(9):2566–2572
Micciulla S, Michalowsky J, Schroer MA, Holm C, von Klitzing R, Smiatek J (2016) Concentration dependent effects of urea binding to poly (n-isopropylacrylamide) brushes: a combined experimental and numerical study. Phys Chem Chem Phys 18:5324–5335
Mishra A, Ekka MK, Maiti S (2016) Influence of ionic liquids on thermodynamics of small molecule DNA interaction: the binding of ethidium bromide to calf thymus DNA. J Phys Chem B 120(10):2691–2700
Mukesh C, Mondal D, Sharma M, Prasad K (2013) Rapid dissolution of DNA in a novel bio-based ionic liquid with long-term structural and chemical stability: successful recycling of the ionic liquid for reuse in the process. Chem Commun 49(61):6849–6851
Nakano M, Tateishi-Karimata H, Tanaka S, Sugimoto N (2014) Choline ion interactions with DNA atoms explain unique stabilization of A–T base pairs in DNA duplexes: a microscopic view. J Phys Chem B 118(2):379–389
Narayanan Krishnamoorthy A, Holm C, Smiatek J (2014) Local water dynamics around antifreeze protein residues in the presence of osmolytes: the importance of hydroxyl and disaccharide groups. J Phys Chem B 118(40):11613–11621
Newman KE (1994) Kirkwood–buff solution theory: derivation and applications. Chem Soc Rev 23(1):31–40
Nishimura N, Nomura Y, Nakamura N, Ohno H (2005) DNA strands robed with ionic liquid moiety. Biomaterials 26(27):5558–5563
Okur HI, Hladílková J, Rembert KB, Cho Y, Heyda J, Dzubiella J, Cremer PS, Jungwirth P (2017) Beyond the hofmeister series: ion-specific effects on proteins and their biological functions. J Phys Chem B 121(9):1997–2014
Oprzeska-Zingrebe EA, Smiatek J (2018) Preferential binding of urea to single-stranded DNA structures:a molecular dynamics study. Biophys J (to be published)
Pabbathi A, Samanta A (2015) Spectroscopic and molecular docking study of the interaction of DNA with a morpholinium ionic liquid. J Phys Chem B 119(34):11099–11105
Pandey PK, Rawat K, Aswal VK, Kohlbrecher J, Bohidar HB (2018) Imidazolium based ionic liquid induced DNA gelation at remarkably low concentration. Coll Surf 538:184–191
Patel R, Kumari M, Khan AB (2014a) Recent advances in the application of ionic liquids in protein stability and activity: a review. Appl Biochem Biotechnol 172:3701–3720
Patel R, Kumari M, Khan AB (2014b) Recent advances in the application of ionic liquids in protein stability and activity: a review. Appl Biochem Biotechnol 172:3701–3720
Pierce V, Kang M, Aburi M, Weerasinghe S, Smith PE (2008) Recent applications of kirkwood-buff theory to biological systems. Cell Biochem Biophys 50:1–22
Qin W, Li SFY (2003) Electrophoresis of DNA in ionic liquid coated capillary. Analyst 128:37–41
Reid JE, Walker AJ, Shimizu S (2015) Residual water in ionic liquids: clustered or dissociated? Phys Chem Chem Phys 17(22):14710–14718
Reid JESJ, Gammons RJ, Slattery JM, Walker AJ, Shimizu S (2017) Interactions in water–ionic liquid mixtures: comparing protic and aprotic systems. J Phys Chem B. https://doi.org/10.1021/acs.jpcb.6b10562
Rodríguez-Ropero F, van der Vegt NF (2015) On the urea induced hydrophobic collapse of a water soluble polymer. Phys Chem Chem Phys 17(13):8491–8498
Rösgen J, Pettitt BM, Bolen DW (2004) Uncovering the basis for nonideal behavior of biological molecules. Biochemistry 43(45):14472–14484
Rösgen J, Pettitt BM, Bolen DW (2005) Protein folding, stability, and solvation structure in osmolyte solutions. Biophys J 89(5):2988–2997
Rösgen J, Pettitt BM, Bolen DW (2007) An analysis of the molecular origin of osmolyte-dependent protein stability. Protein Sci 16(4):733–743
Roy A, Banerjee P, Dutta R, Kundu S, Sarkar N (2016) Probing the interaction between a DNA nucleotide (adenosine-5 ’-monophosphate disodium) and surface active ionic liquids by rotational relaxation measurement and fluorescence correlation spectroscopy. Langmuir 32(42):10946–10956
Saha D, Mukherjee A (2018) Effect of water and ionic liquids on biomolecules. Biophys Rev, 1–14
Saha D, Kulkarni M, Mukherjee A (2016) Water modulates the ultraslow dynamics of hydrated ionic liquids near CG rich DNA: consequences for DNA stability. Phys Chem Chem Phys 18(47):32107–32115
Sapir L, Harries D (2017) Wisdom of the crowd. Bunsen-Magazin 19:152–162
Satpathi S, Kulkarni M, Mukherjee A, Hazra P (2016) Ionic liquid induced G-quadruplex formation and stabilization: spectroscopic and simulation studies. Phys Chem Chem Phys 18(43):29740–29746
Schröder C (2017) Proteins in ionic liquids: current status of experiments and simulations. Top Curr Chem 375(2):25
Schroer MA, Michalowsky J, Fischer B, Smiatek J, Grübel G (2016) Stabilizing effect of tmao on globular pnipam states: preferential attraction induces preferential hydration. Phys Chem Chem Phys 18(46):31459–31470
Schurr JM, Rangel DP, Aragon SR (2005) A contribution to the theory of preferential interaction coefficients. Biophys J 89(4):2258–2276
Senske M, Constantinescu-Aruxandei D, Havenith M, Herrmann C, Weingärtner H, Ebbinghaus S (2016) The temperature dependence of the hofmeister series: thermodynamic fingerprints of cosolute–protein interactions. Phys Chem Chem Phys 18(43):29698–29708
Sharma M, Mondal D, Singh N, Trivedi N, Bhattab J, Prasad K (2015) High concentration DNA solubility in bio-ionic liquids with long-lasting chemical and structural stability at room temperature. RSC Adv 5:40546–40551
Shimizu S (2004) Estimating hydration changes upon biomolecular reactions from osmotic stress, high pressure, and preferential hydration experiments. Proc Natl Acad Sci USA 101(5):1195–1199
Shimizu S, Matubayasi N (2018) A unified perspective on preferential solvation and adsorption based on inhomogeneous solvation theory. Physica A 492:1988–1996
Shimizu S, Smith DJ (2004) Preferential hydration and the exclusion of cosolvents from protein surfaces. J Chem Phys 121(2):1148–1154
Singh PK, Sujana J, Mora AK, Nath S (2012) Probing the dna-ionic liquid interaction using an ultrafast molecular rotor. J Photochem Photobiol A 246:16–22
Smiatek J (2014) Osmolyte effects: impact on the aqueous solution around charged and neutral spheres. J Phys Chem B 118(3):771–782
Smiatek J (2017) Aqueous ionic liquids and their influence on protein structures: an overview on recent theoretical and experimental results. J Phys Condens Matter 29:233001
Smiatek J, Heuer A (2014) Deprotonation mechanism of a single-stranded DNA i-motif. RSC Adv 4:17110–17113
Smiatek J, Harishchandra RK, Rubner O, Galla HJ, Heuer A (2012) Properties of compatible solutes in aqueous solution. Biophys Chem 160(1):62–68
Smith PE (1999) Computer simulation of cosolvent effects on hydrophobic hydration. J Phys Chem B 103(3):525–534
Smith PE (2004) Cosolvent interactions with biomolecules: relating computer simulation data to experimental thermodynamic data. J Phys Chem B 108:18716–18724
Smith PE (2006) Chemical potential derivatives and preferential interaction parameters in biological systems from Kirkwood-Buff theory. Biophys J 91(3):849–856
Smith PE, Matteoli E, O’Connell JP (2013) Fluctuation theory of solutions: applications in chemistry, chemical engineering, and biophysics. CRC Press
Soni SK, Sarkar S, Mirzadeh N, Selvakannan PR, Bhargava SK (2015) Self-assembled functional nanostructure of plasmid DNA with ionic liquid Bmim PF6 : enhanced efficiency in bacterial gene transformation. Langmuir 31(16):4722–4732
Sukenik S, Sapir L, Gilman-Politi R, Harries D (2013a) Diversity in the mechanisms of cosolute action on biomolecular processes. Farad Discuss 160:225–237
Sukenik S, Sapir L, Harries D (2013b) Balance of enthalpy and entropy in depletion forces. Curr Opin Coll Interface Sci 18:495–501
Sun W, Zhang Y, Hu A, Lu Y, Shi F, Lei B, Sun Z (2013) Electrochemical DNA biosensor based on partially reduced graphene oxide modified carbon ionic liquid electrode for the detection of transgenic soybean A2704-12 gene sequence. Electroanal 25(6):1417–1424
Tanford C (1964) Isothermal unfolding of globular proteins in aqueous urea solutions. J Am Chem Soc 86(10):2050–2059
Tanford C (1969) Extension of the theory of linked functions to incorporate the effects of protein hydration. J Mol Biol 39(3):539–544
Tateishi-Karimata H, Sugimoto N (2012) A-T base pairs are more stable than G-C base pairs in a hydrated ionic liquid. Angew Chem Int Ed 51(6):1416–1419
Tateishi-Karimata H, Sugimoto N (2014) Structure, stability and behaviour of nucleic acids in ionic liquids. Nucleic Acids Res 42(14):8831–8844
Tateishi-Karimata H, Nakano M, Pramanik S, Tanaka S, Sugimoto N (2015) i-motifs are more stable than G-quadruplexes in hydrated ionic liquid. Chem Commun 51(32):6909–6912
Timasheff SN (2002) Protein hydration, thermodynamic binding, and preferential hydration. Biochemistry 41(46):13473–13482
Tung HJ, Pfaendtner J (2016) Kinetics and mechanism of ionic-liquid induced protein unfolding: application to the model protein hp35. Mol Syst Des Eng 1(4):382–390
Uhlig F, Zeman J, Smiatek J, Holm C (2018) First-principles parameterization of polarizable coarse-grained force fields for ionic liquids. J Chem Theory Comput. https://doi.org/10.1021/acs.jctc.7b00903
van Rantwijk F, Sheldon RA (2007) Biocatalysis in ionic liquids. Chem Rev 107(6):2757–2785
Vijayaraghavan R, Izgorodin A, Ganesh V, Surianarayanan M, MacFarlane DR (2010) Long-term structural and chemical stability of DNA in hydrated ionic liquids. Angew Chem Int Ed 49(9):1631–1633
Wang JH, Cheng DH, Chen XW, Du Z, Fang ZL (2007) Direct extraction of double-stranded DNA into ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate and its quantification. Anal Chem 79(2):620–625
Wang H, Wang J, Zhang S (2011) Binding Gibbs energy of ionic liquids to calf thymus DNA: a fluorescence spectroscopy study. PCCP 13(9):3906–3910
Weerasinghe S, Smith PE (2003) A Kirkwood–uff derived force field for sodium chloride in water. J Chem Phys 119(21):11342–11349
Weingärtner H, Cabrele C, Herrmann C (2012) How ionic liquids can help to stabilize native proteins. Phys Chem Chem Phys 14(2):415–426
Wilkes JS (2004) Properties of ionic liquid solvents for catalysis. J Mol Catal A 214(1):11–17
Wyman Jr, J (1964) Linked functions and reciprocal effects in hemoglobin: a second look. Adv Prot Chem 19:223–286
Xia Z, Das P, Shakhnovich EI, Zhou R (2012) Collapse of unfolded proteins in a mixture of denaturants. J Am Chem Soc 134(44):18266–18274
Xie YN, Wang SF, Zhang ZL, Pang DW (2008) Interaction between room temperature ionic liquid bmim BF4 and DNA investigated by electrochemical micromethod. J Phys Chem B 112(32):9864–9868
Yan H, Wu J, Dai G, Zhong A, Chen H, Yang J, Han D (2012) Interaction mechanisms of ionic liquids [cnmim] br (n = 4, 6, 8, 10) with bovine serum albumin. J Lumin 132(3):622–628
Yancey PH (2005) Organic osmolytes as compatible, metabolic and counteracting cytoprotectants in high osmolarity and other stresses. J Exp Biol 208(15):2819–2830
Yang Z (2009) Hofmeister effects: an explanation for the impact of ionic liquids on biocatalysis. J Biotechnol 144(1):12–22
Zeindlhofer V, Khlan D, Bica K, Schröder C (2017) Computational analysis of the solvation of coffee ingredients in aqueous ionic liquid mixtures. RSC Adv 7(6):3495–3504
Zeindlhofer V, Berger M, Steinhauser O, Schröder C (2018) A shell-resolved analysis of preferential solvation of coffee ingredients in aqueous mixtures of the ionic liquid 1-ethyl-3-methylimidazolium acetate. J Chem Phys 148(19):193819
Zeman J, Uhlig F, Smiatek J, Holm C (2017) A coarse-grained polarizable force field for the ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate. J Phys Condens Matter 29:504004
Zhang Y, Chen X, Lan J, You J, Chen L (2009) Synthesis and biological applications of imidazolium-based polymerized ionic liquid as a gene delivery vector. Chem Biol Drug Des 74(3):282–288
Zhao H (2005) Effect of ions and other compatible solutes on enzyme activity, and its implication for biocatalysis using ionic liquids. J Mol Cat B 37(1-6):16–25
Zhao H (2015a) DNA stability in ionic liquids and deep eutectic solvents. J Chem Technol Biotechnol 90(1):19–25
Zhao H (2015b) Protein stabilization and enzyme activation in ionic liquids: specific ion effects. J Chem Technol Biotechnol 91:25–50
Zheng W, Borgia A, Buholzer K, Grishaev A, Schuler B, Best RB (2016) Probing the action of chemical denaturant on an intrinsically disordered protein by simulation and experiment. J Am Chem Soc 138(36):11702–11713
Acknowledgements
The authors acknowledge helpful discussions with Diddo Diddens, Volker Lesch, Andreas Heuer, Hans-Joachim Galla, Julian Michalowsky, Miriam Kohagen, Frank Uhlig, Johannes Zeman, Maria Fyta, Takeshi Kobayashi, Anand Narayanan Krishnamoorthy, Samantha Micciulla, Martin Grininger, Wilhelm-Maximilian Hützler, Marc-Benjamin Hahn, Tihomir Solomun, Heinz Sturm, Martin Schroer, Christian Schröder, and Christian Holm.
Funding
This work was funded by the Deutsche Forschungsgemeinschaft through the Sonderforschungsbereich 716 (SFB 716)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Ewa Anna Oprzeska-Zingrebe declares that she has no conflicts of interest. Jens Smiatek declares that he has no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
This article is part of a Special Issue on “Ionic Liquids and Biomolecules” edited by Antonio Benedetto and Hans-Joachim Galla
Rights and permissions
About this article
Cite this article
Oprzeska-Zingrebe, E.A., Smiatek, J. Aqueous ionic liquids in comparison with standard co-solutes. Biophys Rev 10, 809–824 (2018). https://doi.org/10.1007/s12551-018-0414-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-018-0414-7