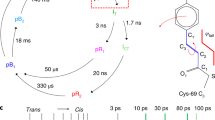
Abstract
X-ray crystallography visualizes the world at the atomic level. It has been used as the most powerful technique for observing the three-dimensional structures of biological macromolecules and has pioneered structural biology. To determine a crystal structure with high resolution, it was traditionally required to prepare large crystals (> 200 μm). Later, synchrotron radiation facilities, such as SPring-8, that produce powerful X-rays were built. They enabled users to obtain good quality X-ray diffraction images even with smaller crystals (ca. 200–50 μm). In recent years, one of the most important technological innovations in structural biology has been the development of X-ray free electron lasers (XFELs). The SPring-8 Angstrom Compact free electron LAser (SACLA) in Japan generates the XFEL beam by accelerating electrons to relativistic speeds and directing them through in-vacuum, short-period undulators. Since user operation started in 2012, we have been involved in the development of serial femtosecond crystallography (SFX) measurement systems using XFEL at the SACLA. The SACLA generates X-rays a billion times brighter than SPring-8. The extremely bright XFEL pulses enable data collection with microcrystals (ca. 50–1 μm). Although many molecular analysis techniques exist, SFX is the only technique that can visualize radiation-damage-free structures of biological macromolecules at room temperature in atomic resolution and fast time resolution. Here, we review the achievements of the SACLA-SFX Project in the past 5 years. In particular, we focus on: (1) the measurement system for SFX; (2) experimental phasing by SFX; (3) enzyme chemistry based on damage-free room-temperature structures; and (4) molecular movie taken by time-resolved SFX.



Similar content being viewed by others
References
Antonyuk SV, Hough MA (2011) Monitoring and validating active site redox states in protein crystals. Biochim Biophys Acta 1814(6):778–784. https://doi.org/10.1016/j.bbapap.2010.12.017
Antonyuk SV, Strange RW, Sawers G, Eady RR, Hasnain SS (2005) Atomic resolution structures of resting-state, substrate- and product-complexed Cu-nitrite reductase provide insight into catalytic mechanism. Proc Natl Acad Sci U S A 102(34):12041–12046. https://doi.org/10.1073/pnas.0504207102
Barends TR, Foucar L, Botha S, Doak RB, Shoeman RL, Nass K et al (2014) De novo protein crystal structure determination from X-ray free-electron laser data. Nature 505(7482):244–247. https://doi.org/10.1038/nature12773
Barends TRM, Foucar L, Ardevol A, Nass K, Aquila A, Botha S et al (2015) Direct observation of ultrafast collective motions in CO myoglobin upon ligand dissociation. Science 350(6259):445–450. https://doi.org/10.1126/science.aac5492
Barty A, Caleman C, Aquila A, Timneanu N, Lomb L, White TA et al (2012) Self-terminating diffraction gates femtosecond X-ray nanocrystallography measurements. Nat Photonics 6:35–40. https://doi.org/10.1038/nphoton.2011.297
Batyuk A, Galli L, Ishchenko A, Han GW, Gati C, Popov PA et al (2016) Native phasing of x-ray free-electron laser data for a G protein-coupled receptor. Sci Adv 2(9):e1600292. https://doi.org/10.1126/sciadv.1600292
Berglund GI, Carlsson GH, Smith AT, Szöke H, Henriksen A, Hajdu J (2002) The catalytic pathway of horseradish peroxidase at high resolution. Nature 417(6887):463–468. https://doi.org/10.1038/417463a
Borshchevskiy VI, Round ES, Popov AN, Büldt G, Gordeliy VI (2011) X-ray-radiation-induced changes in bacteriorhodopsin structure. J Mol Biol 409(5):813–825. https://doi.org/10.1016/j.jmb.2011.04.038
Borshchevskiy V, Round E, Erofeev I, Weik M, Ishchenko A, Gushchin I et al (2014) Low-dose X-ray radiation induces structural alterations in proteins. Acta Crystallogr D Biol Crystallogr 70:2675–2685. https://doi.org/10.1107/S1399004714017295
Boulanger MJ, Murphy MEP (2001) Alternate substrate binding modes to two mutant (D98N and H255N) forms of nitrite reductase from Alcaligenes faecalis S-6: structural model of a transient catalytic intermediate. Biochemistry 40(31):9132–9141. https://doi.org/10.1021/bi0107400
Boulanger MJ, Kukimoto M, Nishiyama M, Horinouchi S, Murphy MEP (2000) Catalytic roles for two water bridged residues (Asp-98 and His-255) in the active site of copper-containing nitrite reductase. J Biol Chem 275(31):23957–23964. https://doi.org/10.1074/jbc.M001859200
Brenner S, Heyes DJ, Hay S, Hough MA, Eady RR, Hasnain SS, Scrutton NS (2009) Demonstration of proton-coupled electron transfer in the copper-containing nitrite reductases. J Biol Chem 284(38):25973–25983. https://doi.org/10.1074/jbc.M109.012245
Chapman HN, Fromme P, Barty A, White TA, Kirian RA, Aquila A et al (2011) Femtosecond X-ray protein nanocrystallography. Nature 470(7332):73–77. https://doi.org/10.1038/nature09750
Colletier JP, Sawaya MR, Gingery M, Rodriguez JA, Cascio D, Brewster AS et al (2016) De novo phasing with X-ray laser reveals mosquito larvicide BinAB structure. Nature 539(7627):43–47. https://doi.org/10.1038/nature19825
Cowtan K (2006) The Buccaneer software for automated model building. 1. Tracing protein chains. Acta Crystallogr D Biol Crystallogr 62(Pt 9):1002–1011. https://doi.org/10.1107/S0907444906022116
Dashti A, Schwander P, Langlois R, Fung R, Li W, Hosseinizadeh A et al (2014) Trajectories of the ribosome as a Brownian nanomachine. Proc Natl Acad Sci U S A 111(49):17492–17497. https://doi.org/10.1073/pnas.1419276111
Dashti A, Hail DB, Mashayekhi G, Schwander P, des Georges A, Frank J et al (2017) Conformational dynamics and energy landscapes of ligand binding in RyR1. bioRxiv 167080. https://doi.org/10.1101/167080
Fraser JS, Clarkson MW, Degnan SC, Erion R, Kern D, Alber T (2009) Hidden alternative structures of proline isomerase essential for catalysis. Nature 462(7273):669–673. https://doi.org/10.1038/nature08615
Fraser JS, van den Bedem H, Samelson AJ, Lang PT, Holton JM, Echols N et al (2011) Accessing protein conformational ensembles using room-temperature X-ray crystallography. Proc Natl Acad Sci U S A 108(39):16247–16252. https://doi.org/10.1073/pnas.1111325108
Fukuda Y, Inoue T (2015) High-temperature and high-resolution crystallography of thermostable copper nitrite reductase. Chem Commun (Camb) 51(30):6532–6535. https://doi.org/10.1039/c4cc09553g
Fukuda Y, Tse KM, Nakane T, Nakatsu T, Suzuki M, Sugahara M et al (2016a) Redox-coupled proton transfer mechanism in nitrite reductase revealed by femtosecond crystallography. Proc Natl Acad Sci U S A 113(11):2928–2933. https://doi.org/10.1073/pnas.1517770113
Fukuda Y, Tse KM, Suzuki M, Diederichs K, Hirata K, Nakane T et al (2016b) Redox-coupled structural changes in nitrite reductase revealed by serial femtosecond and microfocus crystallography. J Biochem 159(5):527–538. https://doi.org/10.1093/jb/mvv133
Ginn HM, Brewster AS, Hattne J, Evans G, Wagner A, Grimes JM et al (2015a) A revised partiality model and post-refinement algorithm for X-ray free-electron laser data. Acta Crystallogr D Biol Crystallogr 71(Pt 6):1400–1410. https://doi.org/10.1107/S1399004715006902
Ginn HM, Messerschmidt M, Ji X, Zhang H, Axford D, Gildea RJ et al (2015b) Structure of CPV17 polyhedrin determined by the improved analysis of serial femtosecond crystallographic data. Nat Commun 6:6435. https://doi.org/10.1038/ncomms7435
Haupts U, Tittor J, Oesterhelt D (1999) Closing in on bacteriorhodopsin: progress in understanding the molecule. Annu Rev Biophys Biomol Struct 28:367–399. https://doi.org/10.1146/annurev.biophys.28.1.367
Horrell S, Antonyuk SV, Eady RR, Hasnain SS, Hough MA, Strange RW (2016) Serial crystallography captures enzyme catalysis in copper nitrite reductase at atomic resolution from one crystal. IUCrJ 3(Pt 4):271–281. https://doi.org/10.1107/S205225251600823X
Hough MA, Antonyuk SV, Strange RW, Eady RR, Hasnain SS (2008) Crystallography with online optical and X-ray absorption spectroscopies demonstrates an ordered mechanism in copper nitrite reductase. J Mol Biol 378(2):353–361. https://doi.org/10.1016/j.jmb.2008.01.097
Hunter MS, Yoon CH, DeMirci H, Sierra RG, Dao EH, Ahmadi R et al (2016) Selenium single-wavelength anomalous diffraction de novo phasing using an X-ray-free electron laser. Nat Commun 7:13388. https://doi.org/10.1038/ncomms13388
Ishikawa T, Aoyagi H, Asaka T, Asano Y, Azumi N, Bizen T et al (2012) A compact X-ray free-electron laser emitting in the sub-angstrom region. Nat Photonics 6(8):540–544. https://doi.org/10.1038/nphoton.2012.141
Johansson LC, Arnlund D, Katona G, White TA, Barty A, DePonte DP et al (2013) Structure of a photosynthetic reaction centre determined by serial femtosecond crystallography. Nat Commun 4:2911. https://doi.org/10.1038/ncomms3911
Kabsch W (2014) Processing of X-ray snapshots from crystals in random orientations. Acta Crystallogr D Biol Crystallogr 70(Pt 8):2204–2216
Kataoka K, Furusawa H, Takagi K, Yamaguchi K, Suzuki S (2000) Functional analysis of conserved aspartate and histidine residues located around the type 2 copper site of copper-containing nitrite reductase. J Biochem 127(2):345–350
Keedy DA, van den Bedem H, Sivak DA, Petsko GA, Ringe D, Wilson MA et al (2014) Crystal cryocooling distorts conformational heterogeneity in a model Michaelis complex of DHFR. Structure 22(6):899–910. https://doi.org/10.1016/j.str.2014.04.016
Kern J, Alonso-Mori R, Hellmich J, Tran R, Hattne J, Laksmono H et al (2012) Room temperature femtosecond X-ray diffraction of photosystem II microcrystals. Proc Natl Acad Sci U S A 109(25):9721–9726. https://doi.org/10.1073/pnas.1204598109
Kern J, Tran R, Alonso-Mori R, Koroidov S, Echols N, Hattne J et al (2014) Taking snapshots of photosynthetic water oxidation using femtosecond X-ray diffraction and spectroscopy. Nat Commun 5:4371. https://doi.org/10.1038/ncomms5371
Kirian RA, White TA, Holton JM, Chapman HN, Fromme P, Barty A et al (2011) Structure–factor analysis of femtosecond microdiffraction patterns from protein nanocrystals. Acta Crystallogr A 67(Pt 2):131–140. https://doi.org/10.1107/S0108767310050981
Kobayashi K, Tagawa S, Deligeer, Suzuki S (1999) The pH-dependent changes of intramolecular electron transfer on copper-containing nitrite reductase. J Biochem 126(2):408–412
Kubo M, Nango E, Tono K, Kimura T, Owada S, Song CY et al (2017) Nanosecond pump–probe device for time-resolved serial femtosecond crystallography developed at SACLA. J Synchrotron Radiat 24:1086–1091. https://doi.org/10.1107/S160057751701030x
Kupitz C, Basu S, Grotjohann I, Fromme R, Zatsepin NA, Rendek KN et al (2014) Serial time-resolved crystallography of photosystem II using a femtosecond X-ray laser. Nature 513(7517):261–265. https://doi.org/10.1038/nature13453
Kupitz C, Olmos JL Jr, Holl M, Tremblay L, Pande K, Pandey S et al (2017) Structural enzymology using X-ray free electron lasers. Struct Dyn 4(4):044003. https://doi.org/10.1063/1.4972069
Leferink NGH, Han C, Antonyuk SV, Heyes DJ, Rigby SEJ, Hough MA et al (2011) Proton-coupled electron transfer in the catalytic cycle of Alcaligenes xylosoxidans copper-dependent nitrite Reductase. Biochemistry 50(19):4121–4131. https://doi.org/10.1021/bi200246f
Li Y, Hodak M, Bernholc J (2015) Enzymatic mechanism of copper-containing nitrite reductase. Biochemistry 54(5):1233–1242. https://doi.org/10.1021/bi5007767
Liu Q, Hendrickson WA (2015) Crystallographic phasing from weak anomalous signals. Curr Opin Struct Biol 34:99–107. https://doi.org/10.1016/j.sbi.2015.08.003
Liu W, Wacker D, Gati C, Han GW, James D, Wang D et al (2013) Serial femtosecond crystallography of G protein-coupled receptors. Science 342(6165):1521–1524. https://doi.org/10.1126/science.1244142
MacKay DJC (1998) Introduction to monte carlo methods. NATO ASI Ser D Behav Soc Sci 89:175–204
Mafuné F, Miyajima K, Tono K, Takeda Y, Kohno JY, Miyauchi N et al (2016) Microcrystal delivery by pulsed liquid droplet for serial femtosecond crystallography. Acta Crystallogr D Struct Biol 72:520–523. https://doi.org/10.1107/S2059798316001480
Maia FRNC (2012) The coherent X-ray imaging data bank. Nat Methods 9(9):854–855. https://doi.org/10.1038/nmeth.2110
Matsui Y, Sakai K, Murakami M, Shiro Y, Adachi S, Okumura H et al (2002) Specific damage induced by X-ray radiation and structural changes in the primary photoreaction of bacteriorhodopsin. J Mol Biol 324(3):469–481. https://doi.org/10.1016/S0022-2836(02)01110-5
Merk A, Bartesaghi A, Banerjee S, Falconieri V, Rao P, Davis MI et al (2016) Breaking Cryo-EM resolution barriers to facilitate drug discovery. Cell 165(7):1698–1707. https://doi.org/10.1016/j.cell.2016.05.040
Moffat K (2001) Time-resolved biochemical crystallography: a mechanistic perspective. Chem Rev 101(6):1569–1581. https://doi.org/10.1021/cr990039q
Nakane T, Song C, Suzuki M, Nango E, Kobayashi J, Masuda T et al (2015) Native sulfur/chlorine SAD phasing for serial femtosecond crystallography. Acta Crystallogr D Biol Crystallogr 71(Pt 12):2519–2525. https://doi.org/10.1107/S139900471501857X
Nakane T, Hanashima S, Suzuki M, Saiki H, Hayashi T, Kakinouchi K et al (2016a) Membrane protein structure determination by SAD, SIR, or SIRAS phasing in serial femtosecond crystallography using an iododetergent. Proc Natl Acad Sci U S A 113(46):13039–13044. https://doi.org/10.1073/pnas.1602531113
Nakane T, Joti Y, Tono K, Yabashi M, Nango E, Iwata S et al (2016b) Data processing pipeline for serial femtosecond crystallography at SACLA. J Appl Crystallogr 49:1035–1041. https://doi.org/10.1107/S1600576716005720
Nango E, Royant A, Kubo M, Nakane T, Wickstrand C, Kimura T et al (2016) A three-dimensional movie of structural changes in bacteriorhodopsin. Science 354(6319):1552–1557. https://doi.org/10.1126/science.aah3497
Nass K, Meinhart A, Barends TR, Foucar L, Gorel A, Aquila A et al (2016) Protein structure determination by single-wavelength anomalous diffraction phasing of X-ray free-electron laser data. IUCrJ 3(Pt 3):180–191. https://doi.org/10.1107/S2052252516002980
Neutze R, Wouts R, van der Spoel D, Weckert E, Hajdu J (2000) Potential for biomolecular imaging with femtosecond X-ray pulses. Nature 406(6797):752–757. https://doi.org/10.1038/35021099
Neutze R, Pebay-Peyroula E, Edman K, Royant A, Navarro J, Landau EM (2002) Bacteriorhodopsin: a high-resolution structural view of vectorial proton transport. Biochim Biophys Acta Biomembr 1565(2):144–167. https://doi.org/10.1016/S0005-2736(02)00566-7
Pande K, Hutchison CDM, Groenhof G, Aquila A, Robinson JS, Tenboer J et al (2016) Femtosecond structural dynamics drives the trans/cis isomerization in photoactive yellow protein. Science 352(6286):725–729. https://doi.org/10.1126/science.aad5081
Sauter NK (2015) XFEL diffraction: developing processing methods to optimize data quality. J Synchrotron Radiat 22(Pt 2):239–248. https://doi.org/10.1107/S1600577514028203
Scapin G (2013) Molecular replacement then and now. Acta Crystallogr D Biol Crystallogr 69(Pt 11):2266–2275
Schlichting I, Berendzen J, Chu K, Stock AM, Maves SA, Benson DE et al (2000) The catalytic pathway of cytochrome p450cam at atomic resolution. Science 287(5458):1615–1622
Sheldrick GM (2010) Experimental phasing with SHELXC/D/E: combining chain tracing with density modification. Acta Crystallogr D Biol Crystallogr 66(Pt 4):479–485. https://doi.org/10.1107/S0907444909038360
Stagno JR, Liu Y, Bhandari YR, Conrad CE, Panja S, Swain M et al (2017) Structures of riboswitch RNA reaction states by mix-and-inject XFEL serial crystallography. Nature 541(7636):242–246. https://doi.org/10.1038/nature20599
Suga M, Akita F, Sugahara M, Kubo M, Nakajima Y, Nakane T et al (2017) Light-induced structural changes and the site of O=O bond formation in PSII caught by XFEL. Nature 543(7643):131–135. https://doi.org/10.1038/nature21400
Sugahara M, Mizohata E, Nango E, Suzuki M, Tanaka T, Masuda T et al (2015) Grease matrix as a versatile carrier of proteins for serial crystallography. Nat Methods 12(1):61–63. https://doi.org/10.1038/nmeth.3172
Sugahara M, Nakane T, Masuda T, Suzuki M, Inoue S, Song CY et al (2017) Hydroxyethyl cellulose matrix applied to serial crystallography. Sci Rep 7:703. https://doi.org/10.1038/s41598-017-00761-0
Taylor GL (2010) Introduction to phasing. Acta Crystallogr D Biol Crystallogr 66(Pt 4):325–338
Tenboer J, Basu S, Zatsepin N, Pande K, Milathianaki D, Frank M et al (2014) Time-resolved serial crystallography captures high-resolution intermediates of photoactive yellow protein. Science 346(6214):1242–1246. https://doi.org/10.1126/science.1259357
Tocheva EI, Rosell FI, Mauk AG, Murphy ME (2004) Side-on copper-nitrosyl coordination by nitrite reductase. Science 304(5672):867–870. https://doi.org/10.1126/science.1095109
Tono K, Nango E, Sugahara M, Song C, Park J, Tanaka T et al (2015) Diverse application platform for hard X-ray diffraction in SACLA (DAPHNIS): application to serial protein crystallography using an X-ray free-electron laser. J Synchrotron Radiat 22(Pt 3):532–537. https://doi.org/10.1107/S1600577515004464
Uervirojnangkoorn M, Zeldin OB, Lyubimov AY, Hattne J, Brewster AS, Sauter NK et al (2015) Enabling X-ray free electron laser crystallography for challenging biological systems from a limited number of crystals. Elife 4:e05421. https://doi.org/10.7554/eLife.05421
Weierstall U, James D, Wang C, White TA, Wang D, Liu W et al (2014) Lipidic cubic phase injector facilitates membrane protein serial femtosecond crystallography. Nat Commun 5:3309. https://doi.org/10.1038/ncomms4309
White TA, Kirian RA, Martin AV, Aquila A, Nass K, Barty A et al (2012) CrystFEL: a software suite for snapshot serial crystallography. J Appl Crystallogr 45:335–341. https://doi.org/10.1107/S0021889812002312
White TA, Mariani V, Brehm W, Yefanov O, Barty A, Beyerlein KR et al (2016) Recent developments in CrystFEL. J Appl Crystallogr 49(Pt 2):680–689. https://doi.org/10.1107/S1600576716004751
Wickstrand C, Dods R, Royant A, Neutze R (2015) Bacteriorhodopsin: would the real structural intermediates please stand up? Biochim Biophys Acta Gen Subj 1850(3):536–553. https://doi.org/10.1016/j.bbagen.2014.05.021
Wijma HJ, Jeuken LJC, Verbeet MP, Armstrong FA, Canters GW (2006) A random-sequential mechanism for nitrite binding and active site reduction in copper-containing nitrite reductase. J Biol Chem 281(24):16340–16346. https://doi.org/10.1074/jbc.M601610200
Yamashita K, Pan D, Okuda T, Sugahara M, Kodan A, Yamaguchi T et al (2015) An isomorphous replacement method for efficient de novo phasing for serial femtosecond crystallography. Sci Rep 5:14017. https://doi.org/10.1038/srep14017
Yamashita K, Kuwabara N, Nakane T, Murai T, Mizohata E, Sugahara M et al (2017) Experimental phase determination with selenomethionine or mercury-derivatization in serial femtosecond crystallography. IUCrJ 4:639–647. https://doi.org/10.1107/S2052252517008557
Acknowledgements
We appreciate all the members of the SACLA-SFX Project, especially Dr. Keitaro Yamashita for the valuable comments on the manuscript. This work was supported by the X-ray Free Electron Laser Priority Strategy Program of the Ministry of Education, Culture, Sports, Science and Technology in Japan and partially by the Strategic Basic Research Programs of the Japan Science and Technology Agency.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Eiichi Mizohata declares that he has no conflict of interest. Takanori Nakane declares that he has no conflict of interest. Yohta Fukuda declares that he has no conflict of interest. Eriko Nango declares that she has no conflict of interest. So Iwata declares that he has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
This article is part of a Special Issue on ‘Biomolecules to Bio-nanomachines - Fumio Arisaka 70th Birthday’ edited by Damien Hall, Junichi Takagi and Haruki Nakamura.
Rights and permissions
About this article
Cite this article
Mizohata, E., Nakane, T., Fukuda, Y. et al. Serial femtosecond crystallography at the SACLA: breakthrough to dynamic structural biology. Biophys Rev 10, 209–218 (2018). https://doi.org/10.1007/s12551-017-0344-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-017-0344-9