Abstract
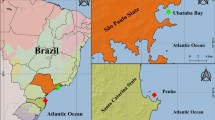
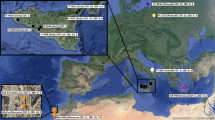
The genus Octopus has been treated as a “catch all” taxon because many species have morphological similarities. To investigate the taxonomic status of the Octopus species in the Tropical Northwestern Atlantic (TNA) and the Tropical Southwestern Atlantic (TSA), we sampled Octopus insularis Leite and Haimovici, 2008 in three areas of the northeastern Brazilian coast, four Brazilian oceanic islands and one island in Western Caribbean, Mexico. Samples of Octopus maya Voss and Solís, 1966 were obtained from the cultivation center of the Universidad Nacional Autónoma de México (UNAM) in Sisal city. Specimens previously identified as Octopus vulgaris Cuvier, 1797 were collected in two regions of southeast Brazil, in an industrial port in Progreso city (southern Gulf of Mexico) and from a fish market in Isla Mujeres, Mexico (Western Caribbean). Molecular species attribution was completed based on morphology of fresh specimens identified by an octopus specialist and then checked against previous identification (cultivation center and GenBank sequences). Molecular analysis of both mitochondrial (cytochrome oxidase I) and nuclear genes (elongation factor-1α), including GenBank data, confirmed that one specimen collected at the Western Caribbean from Mexico and identified as O. insularis, shared the same haplotype of the species from the type locality, indicating the occurrence of this species in the Caribbean Sea. Phylogenetic analyses indicated that 21 GenBank sequences from TNA identified as O. vulgaris grouped within the O. insularis clade and are most likely to be to the latter species. The COI analysis also showed that 18 individuals collected in fishing landings and fish markets, previously identified as O. vulgaris, had the identical haplotype of O. maya collected in the UNAM cultivation center. These results corroborate the misidentification of species in Mexican fisheries. Based on molecular and morphological data we extended the distribution of O. insularis to the TNA and revealed cases of misidentification among the most commercially exploited octopus species in this region.



Similar content being viewed by others
References
Acosta-Jofré MS, Sahade R, Laudien J, Chiappero MB (2012) A contribution to the understanding of phylogenetic relationships among species of the genus Octopus (Octopodidae: Cephalopoda). Sci Mar 76:311–318
Allcock AL, Barratt I, Eléaume M et al (2011) Cryptic speciation and the circumpolarity debate: a case study on endemic Southern Ocean octopuses using the COI barcode of life. Deep-Sea Res Pt II 58:242–249
Amor MD, Laptikhovsky V, Norman MD, Strugnell JM (2015) Genetic evidence extends the known distribution of Octopus insularis to the mid-Atlantic islands Ascension and St Helena. J Mar Biol Assoc UK 1–6
Arreguín-Sánchez F, Solís MJ, González De La Rosa ME (2000) Population dynamics and stock assessment for Octopus maya (Cephalopoda: Octopodidae) fishery in the Campeche Bank, Gulf of Mexico. Rev Biol Trop 48:323–331
Bortolus A (2008) Error cascades in the biological sciences: the unwanted consequences of using bad taxonomy in ecology. Ambio 37:114–118
Bouth HF, Leite TS, Lima FD, Lins-Oliveira JE (2011) Atol das Rocas: an oasis for Octopus insularis (Cephalopoda: Octopodidae). Zoologia 28:45–52
Boyle P, Rodhouse PG (2005) Cephalopods: ecology and fisheries. Blackwell Science, Oxford
Caddy JF, Rodhouse P (1998) Cephalopod and groundfish landings: evidence for ecological change in global fisheries. Rev Fish Biol Fish 8:431–444
Carlini DB, Young RE, Vecchione M (2001) A molecular phylogeny of the Octopoda (Mollusca: Cephalopoda) evaluated in light of morphological evidence. Mol Phylogenet Evol 21:388–97
Dai L, Zheng X, Kong L, Li Q (2012) DNA barcoding analysis of Coleoidea (Mollusca: Cephalopoda) from Chinese waters. Mol Ecol Resour 12:437–447
Drummond AJ, Suchard MA, Xie D, Rambaut A (2012) Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol 29:1969–73
Fadhlaoui-Zid K, Knittweis L, Aurelle D et al (2012) Genetic structure of Octopus vulgaris (Cephalopoda, Octopodidae) in the central Mediterranean Sea inferred from the mitochondrial COIII gene. Comptes Rendus - Biologies 335:625–636
FAO (2012) Fisheries and acquaculture statistics 2010. FAO yearbook, Rome
Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol 3:294–9
Gabrielsen T, Merkel B, Søreide J et al (2012) Potential misidentifications of two climate indicator species of the marine arctic ecosystem: Calanus glacialis and C. finmarchicus. Polar Biol 35:1621–1628
Garcia-Vazquez E, Machado-Schiaffino G, Campo D, Juanes F (2012) Species misidentification in mixed hake fisheries may lead to overexploitation and population bottlenecks. Fish Res 114:52–55
Gusmão J, Lazoski C, Solé-Cava AM (2000) A new species of Penaeus (Crustacea, Penaeidae) revealed by allozyme and cytochrome oxidase I analyses. Mar Biol 137:435–446
Guzik MT, Norman MD, Crozier RH (2005) Molecular phylogeny of the benthic shallow-water octopuses (Cephalopoda: Octopodinae). Mol Phylogenet Evol 37:235–48
Hanlon RT, Messenger JB (1996) Cephalopods behaviour. Cambridge University Press, Cambridge
Hebert PDN, Gregory TR (2005) The promise of DNA barcoding for taxonomy. Syst Biol 54:852–859
Hebert PDN, Cywinska A, Ball SL, deWaard JR (2003) Biological identifications through DNA barcodes. P Roy Soc Lond B Biol 270:313–321
Jassoud AFJ (2010) Genetic differentiation of eastern and western Atlantic Octopus vulgaris. Dissertation, University of Puerto Rico
Jereb P, Roper CFE, Norman MD, Finn JK (2014) Cephalopods of the world. An annotated and illustrated catalogue of cephalopod species known to date. Octopods and vampire squids. FAO species catalogue for fishery purposes, FAO, Rome
Juárez O, Rosas C, Arenta-Ortiz M (2012) Phylogenetic relationships of Octopus maya revealed by mtDNA sequences. Cienc Mar 38:563–575
Judkins HL (2009) Cephalopods of the broad Caribbean: distribution, abundance, and ecological importance. Dissertation, University of South Florida
Kaneko N, Kubodera T, Iguchis A (2011) Taxonomic study of shallow-water Octopuses (Cephalopoda: Octopodidae) in Japan and adjacent waters using mitochondrial genes with perspectives on octopus DNA barcoding. Malacologia 54:97–108
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C, Thierer T, Ashton B, Mentjies P, Drummond A (2012) Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649
Keskin E, Atar HH (2011) Genetic divergence of Octopus vulgaris species in the eastern Mediterranean. Biochem Syst Ecol 39:277–282
Keskin E, Atar HH (2013) DNA barcoding commercially important aquatic invertebrates of Turkey. Mitochondrial DNA 24:440–450
Knowlton N (1993) Sibling species in the sea. Annu Rev Ecol Syst 24:189–216
Knowlton N (2000) Molecular genetic analyses of species boundaries in the sea. Hydrobiologia 420:73–90
Leite TS, Haimovici M, Molina W, Warnke K (2008) Morphological and genetic description of Octopus insularis, a new cryptic species in the Octopus vulgaris complex (Cephalopoda: Octopodidae) from the tropical southwestern Atlantic. J Molluscan Stud 74:63–74
Leite TS, Haimovici M, Mather J, Oliveira JEL (2009) Habitat, distribution, and abundance of the commercial octopus (Octopus insularis) in a tropical oceanic island, Brazil: information for management of an artisanal fishery inside a marine protected area. Fish Res 98:85–91
Leite TS, Batista AT, Lima FD, Mather J (2016) Geographic variability of Octopus insularis diet: from oceanic island to continental populations. Aquat Biol 25:17–27
Lenz TM, Elias NH, Leite TS, Vidal EAG (2015) First description of the eggs and paralarvae of the tropical octopus, Octopus insularis, under culture conditions. Am Malacol Bull 33:1–9
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452
Lima FD, Leite TS, Haimovici M, Oliveira JEL (2014a) Gonadal development and reproductive strategies of the tropical octopus (Octopus insularis) in northeast Brazil. Hydrobiologia 725:7–21
Lima FD, Leite TS, Haimovici M, Nóbrega MF, Oliveira JEL (2014b) Population structure and reproductive dynamics of Octopus insularis (Cephalopoda: Octopodidae) in a coastal reef environment along northeastern Brazil. Fish Res 152:86–92
Lü ZM, Cui WT, Liu LQ, Li HM, Wu CW (2013) Phylogenetic relationships among Octopodidae species in coastal waters of China inferred from two mitochondrial DNA gene sequences. Genet Mol Res 12:3755–3765
Lumpkin R, Garzoli SL (2005) Near-surface circulation in the Tropical Atlantic Ocean. Deep-Sea Res Pt II 52:495–518
Mangold K (1986) Reproduction. In: Boyle PR (ed) Cephalopod life cycles. II - Comparative Reviews. Academic Press, London, pp 157–200
Mather JA (1988) Daytime activity of juvenile Octopus vulgaris in Bermuda. Malacologia 29:69–76
Mather JA (1993) Octopuses as predators: implications for management. In: Okutani T, O’Dor RK, Kubodera T (eds) Recent advances in cephalopod fisheries biology. Tokai University Press, Tokyo
Mather JA, Mather DL (1994) Skin colours and patterns of juvenile Octopus vulgaris (Mollusca, Cephalopoda) in Bermuda. Vie Milieu 44:267–272
Mather JA, O’Dor RK (1991) Foraging strategies and predation risk shape the natural history of juvenile Octopus vulgaris. B Mar Sci 49:256–269
Moguel C, Mascaró M, Avila-Poveda OH, Caamal C, Sanchez A, Pascual C, Rosas C (2010) Morphological, physiological, and behavioral changes during post-hatching development of Octopus maya (Mollusca: Cephalopoda) with special focus on digestive system. Aquat Biol 9:35–48
Norman MD, Hochberg FG (2005) The current state of octopus taxonomy. Phuket Mar Biol Cent Res Bul 66:127–154
Norman MD, Hochberg FG, Finn JK (2014) World octopod fisheries. In: Jereb P, Roper CFE, Norman MD, Finn JK (eds) Cephalopods of the world. An annotated and illustrated catalogue of cephalopod species known to Date. Octopods and vampire squids, Food and Agriculture Organization of the United Nations, Rome, pp 9–21
Opitz (1993) A quantitative model of the trophic interactions in a Caribbean coral reef ecosystem. In: Christensen V, Pauly D (eds) Trophic models of aquatic ecosystems. ICLARM Conf Proc, Makati, pp 259–267
Otero J, González AF, Sieiro MP, Guerra A (2007) Reproductive cycle and energy allocation of Octopus vulgaris in Galician waters, NE Atlantic. Fish Res 85:122–129
Pliego-Cárdenas R, Hochberg FG, León FJG, Barriga-Sosa IDLA (2014) Close genetic relationships between two american octopuses: Octopus hubbsorum Berry, 1953, and Octopus mimus Gould, 1852. J Shellfish Res 33:293–303
Posada D (2008) jModelTest: phylogenetic model averaging. Mol Biol Evol 25:1253–1256
Rambaut A, Suchard MA, Xie D, Drummond AJ (2014) Tracer v1.6. http://beast.bio.ed.ac.uk/Tracer. Accessed 26 November 2015
Rocha LA, Bowen BW (2008) Speciation in coral-reef fishes. J Fish Biol 72:1101–21
Rudorff CAG, Lorenzzetti JA, Gherardi DFM, Lins-Oliveira JE (2009) Modeling spiny lobster larval dispersion in the Tropical Atlantic. Fish Res 96:206–215
SAGARPA (2014) Acuerdo por el que se da a conocer el Plan de Manejo Pesquero de pulpo (O. maya y O. vulgaris) del Golfo de México y Mar Caribe, Mexico
Sales JBDL, Rego PS, Hilsdorf AWS, Moreira AA et al (2013) Phylogeographical features of Octopus vulgaris and Octopus insularis in the Southeastern Atlantic based on the analysis of mitochondrial markers. J Shellfish Res 32:325–339
Schwartz MK, Luikart G, Waples RS (2006) Genetic monitoring as a promising tool for conservation and management. Trends Ecol Evol 22:25–33
Solé-Cava AM, Cunha HA (2012) A genética e a conservação da natureza. In: Matioli SR, Fernandes FMC (eds) Biologia molecular e evolução, 2nd edn. Holos, Ribeirão Preto, pp 217–228
Spalding MD, Fox HE, Allen GR et al (2007) Marine Ecoregions of the world : A bioregionalization of coastal and shelf areas. Bioscience 57:573–583
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Taylor AL, McKeown NJ, Shaw PW (2012) Molecular identification of three co-occurring and easily misidentified octopus species using PCR-RFLP techniques. Conserv Genet Resour 4:885–887
Teske PR, Oosthuizen A, Papadopoulos I, Abstract NPB (2007) Phylogeographic structure of Octopus vulgaris in South Africa revisited: Identification of a second lineage near Durban harbour. Mar Biol 151:2119–2122
Thorpe JP, Sole-Cava AM, Watts PC (2000) Exploited marine invertebrates: genetics and fisheries. Hydrobiologia 420:165–184
Tillett BJ, Meekan MG, Broderick D, Field IC, Cliff G, Ovenden JR (2012) Pleistocene isolation, secondary introgression and restricted contemporary gene flow in the pig-eye shark, Carcharhinus amboinensis across northern Australia. Conserv Genet 13:99–115
Tourinho JL, Solé-Cava AM, Lazoski C (2012) Cryptic species within the commercially most important lobster in the tropical Atlantic, the spiny lobster Panulirus argus. Mar Biol 159:1897–1906
von der Heyden S, Beger M, Toonen RJ et al (2014) The application of genetics to marine management and conservation: Examples from the Indo-Pacific. Bull Mar Sci 90:123–58
Vecchione M, Mickevich MF, Fauchald K, Collette BB, Williams AB, Munroe TA, Young RE (2000) Importance of assessing taxonomic adequacy in determining fishing effects on marine biodiversity. ICES J Mar Sci 57:677–681
Villanueva R, Nozais C, Boletzky SV (1996) Swimming behavior and food searching in planktonic Octopus vulgaris Cuvier from hatching to settlement. J Exp Mar Biol Ecol 208:169–184
Voight JR (1991) Morphological variation in octopod specimens: reassessing the assumption of preservation-induced deformation. Malacologia 33:241–253
Voss GL, Solis-Ramirez M (1966) Octopus maya, a new species from the Bay of Campeche, Mexico. B Mar Sci 16:615–625
Waples RS, Punt AE, Cope JM (2008) Integrating genetic data into management of marine resources: how can we do it better? Fish Fish 9:423–449
Ward RD (2000) Genetics in fisheries management. Hydrobiologia 420:191–201
Warnke K, Söller R, Blohm D, Saint-Paul U (2004) A new look at geographic and phylogenetic relationships within the species group surrounding Octopus vulgaris (Mollusca, Cephalopoda): indications of very wide distribution from mitochondrial DNA sequences. J Zool Syst Evol Res 42:306–312
Wenne R, Boudry P, Hemmer-Hansen J, Lubieniecki KP, Was A, Kause A (2007) What role for genomics in fisheries management and aquaculture? Aquat Living Resour 20:241–255
Acknowledgements
We are thankful to the Chico Mendes Institute for Biodiversity Conservation (MMA/ICMBIO) and the Brazilian Navy for logistics support in the field work. We thank the Brazilian National Research Council (CNPq 481492/2013-9) and Coordination for the Improvement of Higher Education Personnel (CAPES 23038.004807/2014-01) for financial support and research grant (FD). WMBF receives a Ph.D. grant from Science without Borders Program/CNPq (process 233161/2014-7). We are grateful to Dr. Jan Strugnell for reviewing an earlier version of this manuscript. We would also like to thank Nikeisha Caruana and Adam Amato for reviewing the English grammar.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Jorgensen
Rights and permissions
About this article
Cite this article
Lima, F.D., Berbel-Filho, W.M., Leite, T.S. et al. Occurrence of Octopus insularis Leite and Haimovici, 2008 in the Tropical Northwestern Atlantic and implications of species misidentification to octopus fisheries management. Mar Biodiv 47, 723–734 (2017). https://doi.org/10.1007/s12526-017-0638-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-017-0638-y