Abstract
Background
Jaundice is a common neonatal problem. This study was conducted to determine the effect of metoclopramide on neonatal bilirubin and maternal prolactin (primary outcomes) and milk volume (secondary outcome).
Methods
This triple-blind, randomized, controlled, clinical trial was conducted on 112 mothers. The participants were assigned to the intervention (metoclopramide) and control groups (placebo) using block randomization. Ten-mg metoclopramide and placebo tablets were taken by the participants three times a day. The intervention began in the first 2–10 hours after childbirth and continued until the fifth day. The mothers’ prolactin level was measured on the first morning after the intervention and on the sixth day (1 day after the intervention was over). Neonatal total bilirubin was also measured before the intervention and on the sixth day.
Results
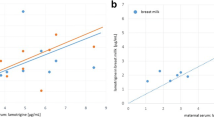
After the intervention, the two groups did not differ significantly in terms of the mean neonatal indirect bilirubin (P = 0.565) and milk volume (P = 0.261), but the mean serum prolactin was significantly higher in the metoclopramide group compared to the placebo group (adjusted mean difference 37; 95% confidence interval 58.1–16.5; P = 0.001).
Conclusions
Metoclopramide increased maternal serum prolactin but had no effects on neonatal jaundice. The insufficient numbers of studies on this subject mandate further research.

Similar content being viewed by others
References
Olusanya BO, Osibanjo FB, Slusher TM. Risk Factors for severe neonatal hyperbilirubinemia in low and middle-income countries: a systematic review and meta-analysis. PLoS One. 2015;10:e0117229.
Dennery PA, Seidman DS, Stevenson DK. Neonatal hyperbilirubinemia. N Engl J Med. 2001;344:581–90.
Kaplan M, Muraca M, Hammerman C, Rubaltelli FF, Vilei MT, Vreman HJ, et al. Imbalance between production and conjugation of bilirubin: a fundamental concept in the mechanism of neonatal jaundice. Pediatrics. 2002;110:e47.
Cohen RS, Wong RJ, Stevenson DK. Understanding neonatal jaundice: a perspective on causation. Pediatr Neonatol. 2010;51:143–8.
Bhutani VK, Gourley GR, Adler S, Kreamer B, Dalin C, Johnson LH. Noninvasive measurement of total serum bilirubin in a multiracial predischarge newborn population to assess the risk of severe hyperbilirubinemia. Pediatrics. 2000;106:E17.
Maisels MJ, McDonagh AF. Phototherapy for neonatal jaundice. N Engl J Med. 2008;358:920–8.
Woodgate P, Jardine LA. Neonatal jaundice: phototherapy. BMJ Clin Evid. 2015;2015:0319.
Cortey A, Elzaabi M, Waegemans T, Roch B, Aujard Y. Efficacy and safety of intravenous immunoglobulins in the management of neonatal hyperbilirubinemia due to ABO incompatibility: a meta-analysis. Arch Pediatr. 2014;21:976–83 (in French).
Dennery PA. Pharmacological interventions for the treatment of neonatal jaundice. Semin Neonatol. 2002;7:111–9.
American Academy of Pediatrics Subcommittee on Hyperbilirubinemia. Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation. Pediatrics. 2004;114:297–316.
Gabay MP. Galactogogues: medications that induce lactation. J Hum Lact. 2002;18:274–9.
Mishriky BM, Habib AS. Metoclopramide for nausea and vomiting prophylaxis during and after Caesarean delivery: a systematic review and meta-analysis. Br J Anaesth. 2012;108:374–83.
Metoclopramide. https://www.drugbank.ca/drugs/DB01233. Accessed 14 Jun 2017.
Zuppa AA, Sindico P, Orchi C, Carducci C, Cardiello V, Romagnoli C. Safety and efficacy of galactogogues: substances that induce, maintain and increase breast milk production. J Pharm Pharm Sci. 2010;13:162–74.
Jo YY, Kim YB, Yang MR, Chang YJ. Extrapyramidal side effects after metoclopramide administration in a post-anesthesia care unit—a case report. Korean J Anesthesiol. 2012;63:274–6.
Food & Drug Administration. FDA requires boxed warning and risk mitigation strategy for metoclopramide-containing drugsagency warns against chronic use of these products to treat gastrointestinal disorders. 2009. http://www.fda.gov/bbs/topics/NEWS/2009/NEW01963.html. Accessed 28 Feb 2017.
Dahl E, Diskin AL. Long-lasting adverse effects after short-term low-dose treatment with metoclopramide for vomiting. Int Marit Health. 2014;65:16–9.
Guzmán V, Toscano G, Canales ES, Zárate A. Improvement of defective lactation by using oral metoclopramide. Acta Obstet Gynecol Scand. 1979;58:53–5.
Donovan TJ, Buchanan K. Medications for increasing milk supply in mothers expressing breastmilk for their preterm hospitalised infants. Cochrane Database Syst Rev. 2012;3:CD005544.
Dastgerdi E, Shirazi M, Mohammadzadeh A, Shahrarha A, Amiri R. Effect of metoclopramide on increased milk production in mothers of preterm infants. J Gyn Ir. 2011;14:32–6 (in Persian).
Ingram J, Taylor H, Churchill C, Pike A, Greenwood R. Metoclopramide or domperidone for increasing maternal breast milk output: a randomised controlled trial. Arch Dis Child Fetal Neonatal Ed. 2012;97:F241–5.
Torre DL, Falorni A. Pharmacological causes of hyperprolactinemia. Ther Clin Risk Manag. 2007;3:929–51.
Lafuente A, Marco J, Esquifino AI. Opioids and the pulsatile prolactin secretory pattern: effects of hyperprolactinemia. Vet Hum Toxicol. 1994;36:524–8.
Lawrence R, Lawrence R. Breastfeeding: a guide for the medical professional. 7th ed. Philadelphia: Saunders; 2011.
Gullu S. Prolactin: an underestimated hormone. Endocrine abstracts. 2008;16:ME9. https://www.endocrine-abstracts.org/ea/0016/ea0016me9. Accessed 12 Nov 2017.
Saeidi R, Gholami Robatsangi M, Mirza Rahimi M, Amiri Tehrani Zadeh M. Comparison of serum bilirubin measurement in laboratory and transcutaneous bilirubinometry. J Sabzevar Univ Med Sci. 1970;16:150–4 (in Persian).
Kauppila A, Anunti P, Kivinen S, Koivisto M, Ruokonen A. Metoclopramide and breast feeding: efficacy and anterior pituitary responses of the mother and the child. Eur J Obstet Gynecol Reprod Biol. 1985;19:19–22.
Hansen WF, McAndrew S, Harris K, Zimmerman MB. Metoclopramide effect on breastfeeding the preterm infant: a randomized trial. Gynecol Obstet. 2005;105:383–9.
Cooke M, Sheehan A, Schmied V. A description of the relationship between breastfeeding experiences, breastfeeding satisfaction, and weaning in the first 3 months after birth. J Hum Lact. 2003;19:145–56.
Ștefănescu E, Pleșu M, Scutari C, Junghină A, Mireșan H. Metoclopramide neurological side effects screening; a pharmacovigilence study in Romanian community pharmacies. J Mind Med Sci. 2015;2:55–66.
Kauppila A, Kivinen S, Ylikorkala O. A dose response relation between improved lactation and metoclopramide. Lancet. 1981;1:1175–7.
Acknowledgements
We wish to express our gratitude to Mr. Neginfar (Alzahra Laboratory Technician) and all the participants.
Funding
This study was funded by Tabriz University of Medical Sciences.
Author information
Authors and Affiliations
Contributions
SOT and RS contributed to the study design, data collection, data interpretation and writing of the manuscript. MM, AJD, and SM contributed to study design, data interpretation and revision of the manuscript. YJ contributed to study design, data interpretation and revision of the manuscript, and prepared drug and placebo. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Ethics Committee of the Research and Technology Deputy of Tabriz University of Medical Sciences (IR.TBZMED.REC.1395.1020). Written informed consent was obtained from all individual participants included in the study.
Conflict of interest
No financial or nonfinancial benefits have been received or will be received from any party related directly or indirectly to the subject of this article.
Rights and permissions
About this article
Cite this article
Tabrizi, S.O., Mirghafourvand, M., Dost, A.J. et al. Effect of metoclopramide administration to mothers on neonatal bilirubin and maternal prolactin: a randomized, controlled, clinical trial. World J Pediatr 15, 135–142 (2019). https://doi.org/10.1007/s12519-018-0217-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-018-0217-8