Abstract
Background
Giant axonal neuropathy (GAN) is a rare neurodegenerative disease transmitted in an autosomal recessive mode. This disorder presents motor and sensitive symptoms with an onset in early childhood. Progressive neurodegeneration makes the patients wheelchair dependent by the end of the second decade of life. Affected individuals do not survive beyond the third decade of life. Molecular analysis has identified mutations in the gene GAN in patients with this disorder. This gene produces a protein called gigaxonin which is presumably involved in protein degradation via the ubiquitin–proteasome system. However, the underlying molecular mechanism is not clearly understood yet.
Methods
Here we present the first patient from Mexico with clinical data suggesting GAN. Sequencing of the GAN gene was carried out. Changes in the nucleotide sequence were investigated for their possible impact on protein function and structure using the publicly available prediction tools PolyPhen-2 and PANTHER.
Results
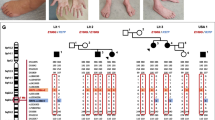
The patient is a compound heterozygous carrying two novel mutations in the GAN gene. The sequence analysis revealed two missense mutations in the Kelch repeats domain. In one allele, a C>T transition was found in exon 9 at the nucleotide position 55393 (g.55393C>T). In the other allele, a transversion G>T in exon 11 at the nucleotide position 67471 (g.67471G>T) was observed. Both of the bioinformatic tools predicted that these amino acid substitutions would have a negative impact on gigaxonin’s function.
Conclusion
This work provides useful information for health professionals and expands the spectrum of disease-causing mutations in the GAN gene and it is the first documented case in Mexican population.




Similar content being viewed by others
References
Asbury AK, Gale MK, Cox SC, Baringer JR, Berg BO. Giant axonal neuropathy--a unique case with segmental neurofilamentous masses. Acta Neuropathol. 1972;20:237–47.
Berg BO, Rosenberg SH, Asbury AK. Giant axonal neuropathy. Pediatrics. 1972;49:894–9.
Johnson-Kerner BL, Roth L, Greene JP, Wichterle H, Sproule DM. Giant axonal neuropathy: an updated perspective on its pathology and pathogenesis. Muscle Nerve. 2014;50:467–76.
Yang Y, Allen E, Ding J, Wang W. Giant axonal neuropathy. Cell Mol Life Sci. 2007;64:601–9.
Donaghy M, King RH, Thomas PK, Workman JM. Abnormalities of the axonal cytoskeleton in giant axonal neuropathy. J Neurocytol. 1988;17:197–208.
Bomont P, Cavalier L, Blondeau F, Ben Hamida C, Belal S, Tazir M, et al. The gene encoding gigaxonin, a new member of the cytoskeletal BTB/kelch repeat family, is mutated in giant axonal neuropathy. Nat Genet. 2000;26:370–4.
Mahammad S, Murthy SN, Didonna A, Grin B, Israeli E, Perrot R, et al. Giant axonal neuropathy-associated gigaxonin mutations impair intermediate filament protein degradation. J Clin Invest. 2013;123:1964–75.
Akagi M, Mohri I, Iwatani Y, Kagitani-Shimono K, Okinaga T, Sakai N, et al. Clinicogenetical features of a japanese patient with giant axonal neuropathy. Brain Dev. 2012;34:156–62.
Abu-Rashid M, Mahajnah M, Jaber L, Kornreich L, Bar-On E, Basel-Vanagaite L, et al. A novel mutation in the GAN gene causes an intermediate form of giant axonal neuropathy in an Arab-Israeli family. Eur J Paediatr Neurol. 2013;17:259–64.
Miyatake S, Tada H, Moriya S, Hirano Y, Hayashi M, Oya Y, et al. Atypical giant axonal neuropathy arising from a homozygous mutation by uniparental isodisomy. Clin Genet. 2014;87:395–7.
Xu M, Da YW, Liu L, Wang F, Jia JP. Giant axonal neuropathy caused by a novel compound heterozygous mutation in the gigaxonin gene. J Child Neurol. 2013;28:1316–9.
Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, et al. A method and server for predicting damaging missense mutations. Nat Methods. 2010;7:248–9.
Thomas PD, Campbell MJ, Kejariwal A, Mi H, Karlak B, Daverman R, et al. PANTHER: a library of protein families and subfamilies indexed by function. Genome Res. 2003;13:2129–41.
Mi H, Lazareva-Ulitsky B, Loo R, Kejariwal A, Vandergriff J, Rabkin S, et al. The PANTHER database of protein families, subfamilies, functions and pathways. Nucleic Acids Res. 2005;33:D284–8.
Mussche S, Devreese B, Nagabhushan Kalburgi S, Bachaboina L, Fox JC, Shih HJ, et al. Restoration of cytoskeleton homeostasis after gigaxonin gene transfer for giant axonal neuropathy. Hum Gene Ther. 2013;24:209–19.
Sames L, Moore A, Arnold R, Ekins S. Recommendations to enable drug development for inherited neuropathies: Charcot–Marie–Tooth and Giant Axonal Neuropathy. Version 2. F1000Res. 2014;3:83.
Stogios PJ, Privé GG. The BACK domain in BTB-kelch proteins. Trends Biochem Sci. 2004;29:634–7.
Perez-Torrado R, Yamada D, Defossez PA. Born to bind: the BTB protein–protein interaction domain. BioEssays. 2006;28:1194–202.
Ding J, Liu JJ, Kowal AS, Nardine T, Bhattacharya P, Lee A, et al. Microtubule-associated protein 1B: a neuronal binding partner for gigaxonin. J Cell Biol. 2002;158:427–33.
Allen E, Ding J, Wang W, Pramanik S, Chou J, Yau V, et al. Gigaxonin-controlled degradation of MAP1B light chain is critical to neuronal survival. Nature. 2005;438:224–8.
Wang W, Ding J, Allen E, Zhu P, Zhang L, Vogel H, et al. Gigaxonin interacts with tubulin folding cofactor B and controls its degradation through the ubiquitin–proteasome pathway. Curr Biol. 2005;15:2050–5.
Ding J, Allen E, Wang W, Valle A, Wu C, Nardine T, et al. Gene targeting of GAN in mouse causes a toxic accumulation of microtubule-associated protein 8 and impaired retrograde axonal transport. Hum Mol Genet. 2006;15:1451–63.
Koop O, Schirmacher A, Nelis E, Timmerman V, De Jonghe P, Ringelstein B, et al. Genotype-phenotype analysis in patients with giant axonal neuropathy (GAN). Neuromuscul Disord. 2007;17:624–30.
Acknowledgements
This work was supported by the Federal budget of the INR. The authors want to acknowledge the patient and her parents for their collaboration in this study.
Author information
Authors and Affiliations
Contributions
MINM, RM, MMH and NEG contributed to the acquisition and analysis of the clinical data. MVF, LCA, VPLS, LMC and AHB designed and performed the molecular analysis of the patient’ parents and control subjects and carried out the bioinformatic analysis. VJRD carried out the histologic analysis of the biopsy. All the authors contributed with the draft of the manuscript and all of them approved the final version for submission.
Corresponding author
Ethics declarations
Ethical approval
The study was approved by the Ethical Committee of the Institutional Review Board of the Medical Specialties Hospital of Leon, Guanajuato. The individual’s parents in the manuscript had given written informed consent to publish this case details. All data were analyzed anonymously.
Conflict of interest
Authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Amplicons of exon 9 and 11 of the gene GAN. Amplicons of exon 9 (327 bp) and 11 (223 bp) of the gene GAN were size separated in 1.5% agarose gels. Figures show PCR products from the father (lane 1), the mother (lane 2) and three unrelated unaffected individuals (lanes 3-5), along 100 bp size marker. Supplementary material 1 (TIFF 164 kb)
Supplementary Fig. 2
Electropherograms of exons 9 and 11 of the gene GAN from one unrelated unaffected subject. The first row of numbers below the nucleotide sequence corresponds to the numeration of the genomic DNA and the second corresponds to the coding DNA sequence. The amino acid sequence and codon number are shown above the nucleotide sequence. Panel A shows the sequence of exon 9. The underlined G corresponds to the first base of exon 9, the upstream sequence from this point corresponds to the intron 8. The codon in italics represents the p.461Ala (GCG). The subject is homozygous for the C allele at this position (red arrow). Panel B shows the sequence of exon 11. The underlined G corresponds to the first base of exon 11, the upstream sequence from this point corresponds to the intron 10. The codon in italics represents the p.545Arg (CGT). The subject is homozygous for the C allele at this position (red arrow). Neither of these variants were found in 100 chromosomes from control individuals. Supplementary material 2 (TIFF 940 kb)
Rights and permissions
About this article
Cite this article
Normendez-Martínez, M.I., Monterde-Cruz, L., Martínez, R. et al. Two novel mutations in the GAN gene causing giant axonal neuropathy. World J Pediatr 14, 298–304 (2018). https://doi.org/10.1007/s12519-018-0140-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-018-0140-z