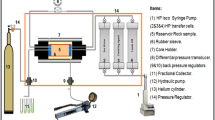
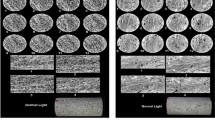
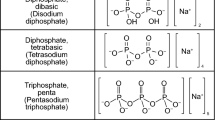
Abstract
Waterflooding through the injection of seawater into petroleum reservoirs is a well-established secondary recovery method to maintain reservoir pressure. The incompatibility between injection and formation water at subsurface requires cost-intensive chemical treatment methods for seawater to reduce scale risk and clogging of pore space. This paper presents an alternative method to improve fluid injectivity by mixing SO42−-enriched seawater with Ba2+-enriched produced water to trigger mineral precipitation. In contrast to the standard procedure of chemical pre-treatment of reservoir-compatible seawater to avoid subsequent scaling in the reservoir, the present technology triggers the precipitation and removal of sulfate minerals prior to injection. Geochemical analyses of major, minor, and trace elements on fluid phases and mineralogical analysis on solid precipitates were coupled with thermodynamic modeling and laboratory experiments in order to define optimum fluid mixing ratios for waterflooding strategies. Geochemical reactive modeling with thermodynamic data revealed the lowest residual SO42− concentrations (< 100 mg/L) in commingled fluids at a mixing ratio of 10:90 between seawater and produced water. A peak removal of 1.1 g of celestite (SrSO4) and 0.5 g barite (BaSO4) is achieved for mixing ratios of 40:60 and 10:90, respectively, per liter of injection fluid water. XRD and ESEM-EDS analyses reconfirmed the formation of homogenous, agglomerated oval-shaped mineral precipitates of 2.5 µmm average size, mainly composed of barite (67 wt%), celestite (10 wt%), and gypsum (14 wt%). As a practical benefit, non-reactive characteristics of the desulfated fluid-mixing product will contribute to minimize scaling of operational wells and to enhance the performance of waterflooding projects. The proposed combination of sulfate removal from seawater and subsequent microfiltration represents a cost-efficient alternative to the energy-intensive nanofiltration technology. The volume and treatment costs of injected seawater will significantly be reduced by the usage of produced water as major component of the total injection fluid mix. Additionally, the enhanced utilization of produced water for waterflooding as a commingled injection fluid component will reduce the amount of produced wastewater and treatment costs.











Similar content being viewed by others
References
Abbasi S, Khamehchi E (2021) Experimental investigation of competitive mechanisms of precipitation and dissolution due to seawater and low salinity water injection in carbonate reservoirs. J Mol Liq 324:114767. https://doi.org/10.1016/j.molliq.2020.114767
Ahmad F, Morris K, Law GTW, Taylor KG, Shaw S (2021) Fate of radium on the discharge of oil and gas produced water to the marine environment. Chemosphere 273:129550. https://doi.org/10.1016/j.chemosphere.2021.129550
Alhseinat E, Sheikholeslami R (2011) An application for new reliable approach to predict the onset of barite, celestite and gypsum scaling during reverse osmosis treatment for produced water. Int J Envir Sci Dev 2(6):454–459. https://doi.org/10.7763/IJESD.2011.V2.168
Al-Roomi YM, Hussain KF (2016) Potential kinetic model for scaling and scale inhibition mechanism. Desalination 393:186–195. https://doi.org/10.1016/j.desal.2015.07.025
Al-Samhan M, Alanezi K, Al-Fadhli J, Al-Attar F, Mukadam S, George J (2020) Evaluating scale deposition and scale tendency of effluent water mix with seawater for compatible injection water. J Petrol Explor Prod Technol 10:2105–2111. https://doi.org/10.1007/s13202-020-00849-w
Al-Shalabi EW, Sepehrnoori K, Pope G (2015) Geochemical interpretation of low salinity water injection in carbonate oil reservoirs. SPE J 20(06):1212–1226. https://doi.org/10.2118/169101-PA
AlSofi AM, Liu JS, Han M (2013) Numerical simulation of surfactant–polymer coreflooding experiments for carbonates. J Petrol Sci Technol 111:184–196. https://doi.org/10.1016/j.petrol.2013.09.009
Austad T, Rezaeidoust A, Puntervold T (2010) Chemical mechanism of low salinity water flooding in sandstone reservoirs. In: SPE-129767-MS. SPE Improved Oil Recovery Symposium, Tulsa, Oklahoma USA. https://doi.org/10.2118/129767-MS
Awolayo A, Sarma H, Alsumaiti AM (2014) A laboratory study of ionic effect of smart water for enhancing oil recovery in carbonate reservoirs. SPE EOR Conference at Oil and Gas West Asia 2014, Muscat, Oman, 31 March - 2 April 2014, SPE 169662-MS, 24 p. https://doi.org/10.2118/169662-MS.
Bader MSH (2007) Sulfate removal technologies for oil fields seawater injection operations. J Petrol Sci Eng 55:93–110. https://doi.org/10.1016/j.petrol.2006.04.010
Baraka-Lokmane S, Hurtevent C (2012) Chemical incompatibilities between formation water and injection water; comparison between modeling and site observations in fields operated by TOTAL. Abu Dhabi Int Petrol Exhibition & Conference (ADIPEC 2012), SPE-161314-MS, p 10. https://doi.org/10.2118/161314-MS
Bedrikovetsky PG, Mackay EJ, Silva RMP, Patricio FMR, Rosário FF (2009) Produced water re-injection with seawater treated by sulphate reduction plant: injectivity decline, analytical model. J Petrol Sci Eng 68:19–28. https://doi.org/10.1016/j.petrol.2009.05.015
Bedrikovetsky PG (1994) Mathematical theory of oil and gas recovery. Kluwer Academic Publishers, London/Boston
Bethke C (1996) Geochemical reaction modelling. Oxford University Press
Bin Merdah AB, Mohd Yassin AA (2007) Barium sulfate scale formation in oil reservoir during water injection at high-barium formation water. J Appl Sci 7:2393–2403. https://doi.org/10.3923/jas.2007.2393.2403
Bin Merdhah AB, Mohd Yassin AA, Muherei MA (2010) Laboratory and prediction of barium sulfate scaling at high-barium formation water. J Petrol Sci Eng 70:79–88. https://doi.org/10.1016/j.petrol.2009.10.001
Birkle P (2012) Advances in geochemical modeling for geothermal applications. In: Bundschuh J, Zilberbrand M (eds) Geochemical modeling of groundwater, vadose and geothermal systems. Chapter 7, CRC Press, Taylor & Francis Group, London, UK, pp 153–178
Brown R (1986) The content and nature of Arabian Gulf Seawater. Emirates Natural History Group (ENHG), Bulletin 29:5–12. https://www.enhg.org/bulletin/b29/29_05.htm
Canic T, Baur S, Bergfeldt T, Kuhn D (2015) Influences on the barite precipitation from geothermal brines. Proceedings World Geothermal Congress 2015, Melbourne, Australia, pp 1–10
Chen HJ, Burnside CA, Widener M, Hinrichsen CJ (2007) Assessment of barite scaling potentials, sulfate removal options, and chemical treating strategies for the Tombua-Landana development. In: SPE 106480. 2007 Int Symp on Oil Field Chemistry, Houston, Texas, USA, p 13. https://doi.org/10.2118/106480-MS
Daher JS, Gomes JAT, Rosario FF, Bezerra MC, Mackay EJ, Sorbie KS (2005) Evaluation of inorganic scale deposition in an unconsolidated reservoir by numerical simulation. 2005 SPE International Conference on Oilfield Scale, Aberdeen, UK, p 12. https://doi.org/10.2118/95107-MS
Delshad M, Han C, Veedu FK, Pope GA (2013) A simplified model for simulations of alkaline–surfactant–polymer floods. J Petrol Sci Eng 108:1–9. https://doi.org/10.1016/j.petrol.2013.04.006
Farajzadeh R, Matsuura T, Van Batenburg D, Dijk H (2012) Detailed modeling of the alkali/surfactant/polymer (ASP) process by coupling a multipurpose reservoir simulator to the chemistry package PHREEQC. SPE Reserv Eval Eng 15(4):423–435. https://doi.org/10.2118/143671-PA
Fink J (2021) Petroleum Engineer’s Guide to Oil Field Chemicals and Fluids. Chapter 7 – Scale Inhibitors, 3rd edn. Elsevier, pp 351–391. https://doi.org/10.1016/C2020-0-02705-2
Fu Y, van Berk W, Schulz HM (2012) Hydrogeochemical modelling of fluid-rock interactions triggered by seawater injection into oil reservoirs: Case study Miller field (UK North Sea). Appl Geochem 27:1266–1277
Ghalib H, Almallah I (2017) Scaling simulation resulting from mixing predicted model between Mishrif formation water and different waters injection in Basrah oil feld, southern Iraq. Model Earth Syst Environ 3:1557. https://doi.org/10.1007/s40808-017-0384-y
Ghasemian J, Riahi S (2021) Effects of salinity and ionic composition of smartwater on mineral scaling in carbonate reservoirs during water flooding. Petrol Explor Develop 48(2):421–429. https://doi.org/10.1016/S1876-3804(21)60033-2
Heatherly MW, Howell ME, McElhiney JE (1994) Sulfate removal technology for seawater waterflood injection. 26th Annual OTC, Houston, TX, pp 745–762. https://doi.org/10.4043/7593-MS
Hummel W, Berner U, Curti E, Pearson FJ, Thoenen T. (2002) Nagra/PSI Chemical Thermodynamic Data Base 01/01. Nagra Technical Report NTB 02-16, Nagra, Wettingen, Switzerland, and Universal Publishers/uPublish.com, Parkland, Florida, USA, 585.
Kan AT, Dai Z, Tomson MB (2020) The state of the art in scale inhibitor squeeze treatment. Petrol Sci 17:1579–1601. https://doi.org/10.1007/s12182-020-00497-z
Kharaka YK, Hanor JS (2014) Deep fluids in sedimentary basins. Surface and groundwater, weathering and soils. In: Holland HD, Turekian KK (eds) Treatise on Geochemistry, vol 7, 2nd edn. Elsevier, Oxford, United Kingdom, pp 471–515. https://doi.org/10.1016/B978-0-08-095975-7.00516-7
Mahmoud M, Elkatatny S, Abdelgawad KZ (2017) Using high- and low-salinity seawater injection to maintain the oil reservoir pressure without damage. J Petrol Explor Prod Technol 7:589–596. https://doi.org/10.1007/s13202-016-0279-x
Nowrouzi I, Khaksar Manshad A, Mohammadi AH (2020) The mutual effects of injected fluid and rock during imbibition in the process of low and high salinity carbonated water injection into carbonate oil reservoirs. J Mol Liq 305:112432. https://doi.org/10.1016/j.molliq.2019.112432
Oddo JE, Tomson MB (1994) Why scale forms and how to predict it. SPE Prod Fac 9:47–54. https://doi.org/10.2118/21710-PA
Parkhurst DL, Appelo CAJ (2013) Description of input and examples for PHREEQC Version 3 – a computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. U.S. Geological Survey Techniques and Methods book 6, (Chapter A43), http://pubs.usgs.gov/tm/06/a43/.
Philips OM (1991) Flow and reactions in porous media. Cambridge University Press
Plummer L, Parkhurst D, Fleming G, Dunkle S (1988) A computer program incorporating Pitzer’s equations for calculation of geochemical reactions in brines. U.S. Geological Survey, Water - Resources Investigations Report 88-4153, Reston, Virginia, p 309
Puntervold T, Austad T (2008) Injection of seawater and mixtures with produced water into North Sea chalk formation: impact of fluid–rock interactions on wettability and scale formation. J Petrol Sci Eng 63:23–33. https://doi.org/10.1016/j.petrol.2008.07.010.
Scogings A (2014) Drilling grade barite: the global outlook. Executive summary to IM’ drilling grade barite report. https://www.csaglobal.com/wp-content/uploads/2015/03/Drilling-grade-barite-the-global-outlook-IM-Mag-January-2015.pdf.
Sorbie KS, Mackay EJ (2000) Mixing of injected, connate and aquifer brines in water flooding and its relevance to oilfield scaling. J Petrol Sci Eng 27:85–106. https://doi.org/10.1016/S0920-4105(00)00050-4
Templeton CC (1960) Solubility of barium sulfate in sodium chloride solutions from 25° to 95° C. J Chem Eng Data 5(4):514–516
van Berk W, Fu Y, Schulz H-M (2015) Temporal and spatial development of scaling in reservoir aquifers triggered by seawater injection: three-dimensional reactive mass transport modeling of water–rock–gas interactions. J Petrol Sci Technol 135:206–217. https://doi.org/10.1016/j.petrol.2015.09.014
Vu VK, Hurtevent C, Davis RA (2000) Eliminating the need for scale inhibition treatments for Elf Exploration Angola’s Girassol field. International Symposium on Oilfield Scale, Aberdeen, UK. https://doi.org/10.2118/60220-MS
Yousef AA, Al-Saleh S, Al-Jawfi M (2012) Improved/enhanced oil recovery from carbonate reservoirs by tuning injection water salinity and ionic content. SPE-154076-MS, Eighteenth SPE Improved Oil Recovery Symposium, Tulsa, Oklahoma, p 18. https://doi.org/10.2118/154076-MS
Yuan M, Todd AC, Sorbie KS (1994) Sulphate scale precipitation arising from seawater injection: a prediction study. Mar Petrol Geol 11:24–30. https://doi.org/10.1016/0264-8172(94)90006-X
Woods AW, Harker G (2003) Barium sulphate precipitation in porous rock through dispersive mixing. SPE paper 80401. SPE-80401-MS, SPE 5th International Symposium on Oilfield Scale, Aberdeen, UK. https://doi.org/10.2118/80401-MS
Zara K, Verbeek PHJ (2003) Water to value - produced water management for sustainable field development of mature and green fields. J Petrol Technol 55:26–28. https://doi.org/10.2118/73853-MS
Zielinski RA, Budahn JR (2007) Mode of occurrence and environmental mobility of oil-field radioactive material at US Geological Survey research site B, Osage-Skiatook Project, northeastern Oklahoma. Appl Geochem 22(10):2125–2137. https://doi.org/10.1016/j.apgeochem.2007.04.014
Zularisam AW, Ismail AF, Sakinah M (2010) Application and challenges of membrane in surface water treatment. J Appl Sci 10:380–390. https://doi.org/10.3923/jas.2010.380.390
Acknowledgements
The authors acknowledge the performance of geochemical analysis of water samples by the Chemical Analysis (R & DC: Saeed H. Shahrani, Ali M. Tawfiq, and Nada S. Alghamdi) and thank Ibrahim Z. Atwah (EXPEC ARC) for reviewing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author(s) declare that they have no competing interests.
Additional information
Responsible Editor: Broder J. Merkel
Yunjiao Fu and Shouwen Shen are no longer with Saudi Aramco.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Birkle, P., Fu, Y., Al-ShaikhAli, A.H. et al. Scale avoidance during waterflooding by optimized effluent mixing. Arab J Geosci 16, 426 (2023). https://doi.org/10.1007/s12517-023-11495-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12517-023-11495-x