Abstract
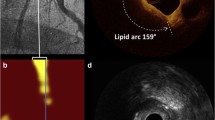
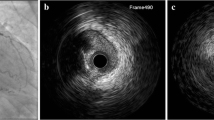
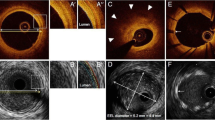
Intracoronary near-infrared spectroscopy (NIRS)-IVUS is a direct coronary imaging catheter for the identification and quantification of lipid core plaque (LCP). NIRS is combined with traditional grayscale IVUS in a hybrid catheter that provides co-registered architectural and compositional plaque characterization data. The novel capability of the NIRS catheter component is its ability to directly identify LCP, which underlies the majority of vulnerable and unstable plaques. NIRS-IVUS has potential to optimize percutaneous coronary intervention (PCI) by delineating precise lesion length, assuring adequate stent expansion and apposition, detection of stent edge dissection, and predicting periprocedural myocardial infarction (PMI). NIRS-IVUS may also be important in the identification of vulnerable plaques at risk to cause future events.










Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Liu X et al. Intravascular ultrasound assessment of the incidence and predictors of edge dissections after drug-eluting stent implantation. JACC Cardiovasc Interv. 2009;2(10):997–1004.
Oemrawsingh RM et al. Near-infrared spectroscopy predicts cardiovascular outcome in patients with coronary artery disease. J Am Coll Cardiol. 2014;64(23):2510–8.
Witzenbichler B et al. Relationship between intravascular ultrasound guidance and clinical outcomes after drug-eluting stents: the assessment of dual antiplatelet therapy with drug-eluting stents (ADAPT-DES) study. Circulation. 2014;129(4):463–70. This is one of the largest studies documenting that IVUS-guidance of PCI leads to changes in the stenting procedure that may lead to improved clinical outcomes.
Zhang Y et al. Comparison of intravascular ultrasound versus angiography-guided drug-eluting stent implantation: a meta-analysis of one randomised trial and ten observational studies involving 19,619 patients. EuroIntervention. 2012;8(7):855–65. This large meta-analysis compiles the available randomized and non-randomized data and shows that IVUS-guidance of PCI results in improved clinical outcomes.
Sakhuja R et al. Residual thrombogenic substrate after rupture of a lipid-rich plaque: possible mechanism of acute stent thrombosis? Circulation. 2010;122(22):2349–50.
Waxman S et al. A case of lipid core plaque progression and rupture at the edge of a coronary stent: elucidating the mechanisms of drug-eluting stent failure. Circ Cardiovasc Interv. 2010;3(2):193–6.
Fujii K et al. Intravascular ultrasound assessment of ulcerated ruptured plaques: a comparison of culprit and nonculprit lesions of patients with acute coronary syndromes and lesions in patients without acute coronary syndromes. Circulation. 2003;108(20):2473–8.
Jang IK et al. In vivo characterization of coronary atherosclerotic plaque by use of optical coherence tomography. Circulation. 2005;111(12):1551–5.
Kubo T et al. Assessment of culprit lesion morphology in acute myocardial infarction: ability of optical coherence tomography compared with intravascular ultrasound and coronary angioscopy. J Am Coll Cardiol. 2007;50(10):933–9.
Stone GW et al. A prospective natural-history study of coronary atherosclerosis. N Engl J Med. 2011;364(3):226–35. This seminal study suggests that there are features of plaque instability assessed by virtual histology IVUS that may predict downstream acute coronary events in the non-culprit arteries in patients presenting with acute myocardial infarction.
Virmani R et al. Pathology of the vulnerable plaque. J Am Coll Cardiol. 2006;47(8 Suppl):C13–8.
Yamagishi M et al. Morphology of vulnerable coronary plaque: insights from follow-up of patients examined by intravascular ultrasound before an acute coronary syndrome. J Am Coll Cardiol. 2000;35(1):106–11.
Goldstein JA et al. Coronary embolization following balloon dilation of lipid-core plaques. JACC Cardiovasc Imaging. 2009;2(12):1420–4.
Goldstein JA et al. Detection of lipid-core plaques by intracoronary near-infrared spectroscopy identifies high risk of periprocedural myocardial infarction. Circ Cardiovasc Interv. 2011;4(5):429–37.
Heusch G et al. Coronary microembolization: from bedside to bench and back to bedside. Circulation. 2009;120(18):1822–36.
Hong YJ et al. Impact of plaque composition on cardiac troponin elevation after percutaneous coronary intervention: an ultrasound analysis. JACC Cardiovasc Imaging. 2009;2(4):458–68.
Kawamoto T et al. The relationship between coronary plaque characteristics and small embolic particles during coronary stent implantation. J Am Coll Cardiol. 2007;50(17):1635–40.
Kotani J et al. Plaque gruel of atheromatous coronary lesion may contribute to the no-reflow phenomenon in patients with acute coronary syndrome. Circulation. 2002;106(13):1672–7.
Mauri L, Rogers C, Baim DS. Devices for distal protection during percutaneous coronary revascularization. Circulation. 2006;113(22):2651–6.
Mehran R et al. Atherosclerotic plaque burden and CK-MB enzyme elevation after coronary interventions: intravascular ultrasound study of 2256 patients. Circulation. 2000;101(6):604–10.
Selvanayagam JB et al. Troponin elevation after percutaneous coronary intervention directly represents the extent of irreversible myocardial injury: insights from cardiovascular magnetic resonance imaging. Circulation. 2005;111(8):1027–32.
Brugaletta S et al. NIRS and IVUS for characterization of atherosclerosis in patients undergoing coronary angiography. JACC Cardiovasc Imaging. 2011;4(6):647–55.
Gardner CM et al. Detection of lipid core coronary plaques in autopsy specimens with a novel catheter-based near-infrared spectroscopy system. JACC Cardiovasc Imaging. 2008;1(5):638–48. This study validates NIRS for the detection of coronary artery cholesterol in vivo.
Madder RD et al. Detection by near-infrared spectroscopy of large lipid core plaques at culprit sites in patients with acute ST-segment elevation myocardial infarction. JACC Cardiovasc Interv. 2013;6(8):838–46. This study suggests that lipid core plaque is the precusor to nearly every case of ST-segment elevation myocardial infarction.
Madder RD et al. Composition of target lesions by near-infrared spectroscopy in patients with acute coronary syndrome versus stable angina. Circ Cardiovasc Interv. 2012;5(1):55–61.
Schultz CJ et al. First-in-man clinical use of combined near-infrared spectroscopy and intravascular ultrasound: a potential key to predict distal embolization and no-reflow? J Am Coll Cardiol. 2010;56(4):314.
Waxman S et al. In vivo validation of a catheter-based near-infrared spectroscopy system for detection of lipid core coronary plaques: initial results of the SPECTACL study. JACC Cardiovasc Imaging. 2009;2(7):858–68.
Oemrawsingh PV et al. Intravascular ultrasound guidance improves angiographic and clinical outcome of stent implantation for long coronary artery stenoses: final results of a randomized comparison with angiographic guidance (TULIP Study). Circulation. 2003;107(1):62–7.
Dixon SR et al. Analysis of target lesion length before coronary artery stenting using angiography and near-infrared spectroscopy versus angiography alone. Am J Cardiol. 2012;109(1):60–6.
Goldstein JA et al. Multiple complex coronary plaques in patients with acute myocardial infarction. N Engl J Med. 2000;343(13):915–22.
Madder RD, Wohns DH, Muller JE. Detection by intracoronary near-infrared spectroscopy of lipid core plaque at culprit sites in survivors of cardiac arrest. J Invasive Cardiol. 2014;26(2):78–9.
Boden WE et al. Optimal medical therapy with or without PCI for stable coronary disease. N Engl J Med. 2007;356(15):1503–16.
Shaw LJ et al. Optimal medical therapy with or without percutaneous coronary intervention to reduce ischemic burden: results from the Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation (COURAGE) trial nuclear substudy. Circulation. 2008;117(10):1283–91.
Kini AS et al. Changes in plaque lipid content after short-term intensive versus standard statin therapy: the YELLOW trial (reduction in yellow plaque by aggressive lipid-lowering therapy). J Am Coll Cardiol. 2013;62(1):21–9.
Compliance with Ethics Guidelines
Conflict of Interest
JA Goldstein has served as a consultant, received travel reimbursement, and owns stock in InfraReDx, Inc. JE Muller has served as Chairman of the Board and Chief Medical Officer, received travel reimbursement, and owns stock in InfraReDx, Inc. ID Hanson, SR Dixon, AE Abbas, and RD Safian all declare no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
James A. Goldstein, M.D. is a consultant for and owner of equity in InfraReDx, Inc.
James E. Muller is a current employee of InfraReDx, Inc.
This article is part of the Topical Collection on Intravascular Imaging
Rights and permissions
About this article
Cite this article
Hanson, I.D., Goldstein, J.A., Dixon, S.R. et al. Present Status and Future Direction of NIRS-IVUS Multimodality Direct Coronary Imaging. Curr Cardiovasc Imaging Rep 8, 25 (2015). https://doi.org/10.1007/s12410-015-9342-0
Published:
DOI: https://doi.org/10.1007/s12410-015-9342-0