Abstract
Endomyocardial biopsy with its inherent invasiveness and morbidity calls for the development of noninvasive imaging methods to evaluate heart transplant recipients. While conventional imaging technologies report on anatomical and metabolic changes in heart grafts, macrophage-targeted imaging could allow disease detection before gross anatomical and functional changes have occurred. One important approach in magnetic resonance–based molecular imaging exploits an increased T2/T2* relaxation effect, occurring when phagocytic cells localized in the heart graft take up iron-oxide nanoparticles. This methodology of nanoparticle reporting on immune cell accumulation in the graft combined with precise functional and morphological information of cardiac MRI has potential to supplant endomyocardial biopsy. The use of multifunctional nanoparticles fit for multiple imaging modalities (magnetic, optical, and nuclear) will help improve methods of ex vivo and in vitro imaging of allograft rejection and also further our knowledge of allograft rejection by providing a tool for nondestructive serial in vivo assessment.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Mehra MR, Ventura HO, Chambers R, et al.: Predictive model to assess risk for cardiac allograft vasculopathy: an intravascular ultrasound study. J Am Coll Cardiol 1995, 26:1537–1544.
Yamani MH, Yousufuddin M, Starling RC, et al.: Does acute cellular rejection correlate with cardiac allograft vasculopathy? J Heart Lung Transplant 2004, 23:272–276.
Hathout E, Beeson WL, Kuhn M, et al.: Cardiac allograft vasculopathy in pediatric heart transplant recipients. Transpl Int 2006, 19:184–189.
Rodriguez ER: The pathology of heart transplant biopsy specimens: revisiting the 1990 ISHLT working formulation. J Heart Lung Transplant 2003, 22:3–15.
Tan CD, Baldwin WM 3rd, Rodriguez ER: Update on cardiac transplantation pathology. Arch Pathol Lab Med 2007, 131:1169–1191.
Starling RC, Pham M, Valantine H, et al.: Molecular testing in the management of cardiac transplant recipients: initial clinical experience. J Heart Lung Transplant 2006, 25:1389–1395.
Mondillo S, Maccherini M, Galderisi M: Usefulness and limitations of transthoracic echocardiography in heart transplantation recipients. Cardiovasc Ultrasound 2008, 6:2.
Revel D, Chapelon C, Mathieu D, et al.: Magnetic resonance imaging of human orthotopic heart transplantation: correlation with endomyocardial biopsy. J Heart Transplant 1989, 8:139–146.
Smart FW, Young JB, Weilbaecher D, et al.: Magnetic resonance imaging for assessment of tissue rejection after heterotopic heart transplantation. J Heart Lung Transplant 1993, 12:403–410.
Almenar L, Igual B, Martinez-Dolz L, et al.: Utility of cardiac magnetic resonance imaging for the diagnosis of heart transplant rejection. Transplant Proc 2003, 35:1962–1964.
Marie PY, Carteaux JP, Angioi M, et al.: Detection and prediction of acute heart transplant rejection: preliminary results on the clinical use of a “black blood” magnetic resonance imaging sequence. Transplant Proc 1998, 30:1933–1935.
Marie PY, Angioi M, Carteaux JP, et al.: Detection and prediction of acute heart transplant rejection with the myocardial T2 determination provided by a black-blood magnetic resonance imaging sequence. J Am Coll Cardiol 2001, 37:825–831.
Wisenberg G, Pflugfelder PW, Kostuk WJ, et al.: Diagnostic applicability of magnetic resonance imaging in assessing human cardiac allograft rejection. Am J Cardiol 1987, 60:130–136.
Mousseaux E, Farge D, Guillemain R, et al.: Assessing human cardiac allograft rejection using MRI with Gd-DOTA. J Comput Assist Tomogr 1993, 17:237–244.
Kanno S, Wu YJ, Lee PC, et al.: Macrophage accumulation associated with rat cardiac allograft rejection detected by magnetic resonance imaging with ultrasmall superparamagnetic iron oxide particles. Circulation 2001, 104:934–938.
Romeo G, Houyel L, Angel CY, et al.: Coronary stenosis detection by 16-slice computed tomography in heart transplant patients: comparison with conventional angiography and impact on clinical management. J Am Coll Cardiol 2005, 45:1826–1831.
Weinreb JC, Larson PA, Woodard PK, et al.: American College of Radiology clinical statement on noninvasive cardiac imaging. Radiology 2005, 235:723–727.
Rechavia E, de Silva R, Kushwaha SS, et al.: Enhanced myocardial 18F-2-fluoro-2-deoxyglucose uptake after orthotopic heart transplantation assessed by positron emission tomography. J Am Coll Cardiol 1997, 30:533–538.
Ardehali A, Laks H, Drinkwater DC, et al.: Vascular cell adhesion molecule-1 is induced on vascular endothelia and medial smooth muscle cells in experimental cardiac allograft vasculopathy. Circulation 1995, 92:450–456.
Dietrich H, Hu Y, Zou Y, et al.: Mouse model of transplant arteriosclerosis: role of intercellular adhesion molecule-1. Arterioscler Thromb Vasc Biol 2000, 20:343–352.
Lee S, Willoughby WF, Smallwood CJ, et al.: Heterotopic heart and lung transplantation in the rat. Am J Pathol 1970, 59:279–298.
Corry RJ, Winn HJ, Russell PS: Primarily vascularized allografts of hearts in mice. The role of H-2D, H-2K, and non-H-2 antigens in rejection. Transplantation 1973, 16:343–350.
Shimizu K, Schonbeck U, Mach F, et al.: Host CD40 ligand deficiency induces long-term allograft survival and donor-specific tolerance in mouse cardiac transplantation but does not prevent graft arteriosclerosis. J Immunol 2000, 165:3506–3518.
• Hasegawa T, Visovatti SH, Hyman MC, et al.: Heterotopic vascularized murine cardiac transplantation to study graft arteriopathy. Nat Protoc 2007, 2:471–480. This very comprehensive review of the procedure protocol of heterotopic vascularized murine cardiac transplantation includes extensive advice for troubleshooting.
Figueiredo JL, Nahrendorf M, Sosnovik DE, Weissleder R: MRI of a novel murine working heart transplant model. Circ Heart Fail 2009, 2:272–274.
Villanueva FS: Molecular imaging of cardiovascular disease using ultrasound. J Nucl Cardiol 2008, 15:576–586.
Ho C, Hitchens TK: A non-invasive approach to detecting organ rejection by MRI: monitoring the accumulation of immune cells at the transplanted organ. Curr Pharm Biotechnol 2004, 5:551–566.
Wu YL, Ye Q, Foley LM, et al.: In situ labeling of immune cells with iron oxide particles: an approach to detect organ rejection by cellular MRI. Proc Natl Acad Sci U S A 2006, 103:1852–1857.
Ye Q, Wu YL, Foley LM, et al.: Longitudinal tracking of recipient macrophages in a rat chronic cardiac allograft rejection model with noninvasive magnetic resonance imaging using micrometer-sized paramagnetic iron oxide particles. Circulation 2008, 118:149–156.
Chang HH, Moura JM, Wu YL, Ho C: Automatic detection of regional heart rejection in USPIO-enhanced MRI. IEEE Trans Med Imaging 2008, 27:1095–1106.
Wu YJ, Sato K, Ye Q, Ho C: MRI investigations of graft rejection following organ transplantation using rodent models. Methods Enzymol 2004, 386:73–105.
• Wu YL, Ye Q, Sato K, et al.: Noninvasive evaluation of cardiac allograft rejection by cellular and functional cardiac magnetic resonance. JACC Cardiovasc Imaging 2009, 2:731–741. This article illustrates the promise of MRI for cardiac allograft rejection by combining detection of immune cell infiltration by iron-oxide nanoparticles with conventional cardiac magnetic resonance techniques.
Penno E, Johansson L, Ahlstrom H, Johnsson C: Ultrasmall iron oxide particle contrast agent and MRI can be used to monitor the effect of anti-rejection treatment. Transplantation 2007, 84:374–379.
Johansson L, Johnsson C, Penno E, et al.: Acute cardiac transplant rejection: detection and grading with MR imaging with a blood pool contrast agent-experimental study in the rat. Radiology 2002, 225:97–103.
Tanaka M, Swijnenburg RJ, Gunawan F, et al.: In vivo visualization of cardiac allograft rejection and trafficking passenger leukocytes using bioluminescence imaging. Circulation 2005, 112:I105–I110.
Ma L, Xiang Z, Sherrill TP, et al.: Bioluminescence imaging visualizes activation of nuclear factor-kappaB in mouse cardiac transplantation. Transplantation 2008, 85:903–910.
• Christen T, Nahrendorf M, Wildgruber M, et al.: Molecular imaging of innate immune cell function in transplant rejection. Circulation 2009, 119:1925–1932. This article illustrates the importance of multifunctional nanoparticles fit for multiple imaging modalities and how this versatility facilitates ex vivo validation of imaging signals of cardiac allograft rejection.
Nahrendorf M, Keliher E, Panizzi P, et al.: 18F-4V for PET-CT imaging of VCAM-1 expression in atherosclerosis. JACC Cardiovasc Imaging 2009, 2:1213–1222.
Acknowledgments
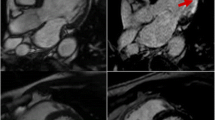
This work was supported by the Donald W. Reynolds Foundation, by grants from the National Institutes of Health (HL-80472 to Dr. Libby, and HL-80731 to Dr. Libby and Prof. Ralph Weissleder), by an American Society of Transplantation Faculty Development grant (to Dr. Shimizu), and by an American Heart Association Scientist Development grant (to Dr. Shimizu). We acknowledge the Center for Systems Biology and the Center for Molecular Imaging Research from Massachusetts General Hospital, directed by Prof. Ralph Weissleder, for support in obtaining data presented in the figure (Matthias Nahrendorf, MD, PhD and Peter Waterman, BS in FRI; Nicolai Sergeyev, PhD in probe preparation; and Yoshi Iwamoto, BS in histology). We thank Sara Karwacki for editing the manuscript.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Christen, T., Shimizu, K. & Libby, P. Advances in Imaging of Cardiac Allograft Rejection. curr cardiovasc imaging rep 3, 99–105 (2010). https://doi.org/10.1007/s12410-010-9011-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12410-010-9011-2