Abstract
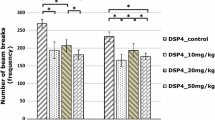
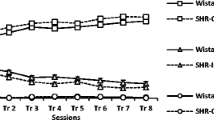
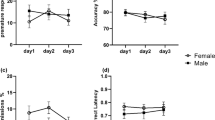
The aim of the present study was to assess the effects of the neurotoxin N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP4), which allows a depletion of noradrenergic terminals in a dose-dependent manner, on attention in rats as measured using the five-choice serial-reaction time task (5CSRTT). In addition, we investigated whether the effects of DSP4 treatment can be reversed by atomoxetine. Atomoxetine is a selective noradrenaline reuptake inhibitor and has been shown to be effective in the treatment of attention deficit hyperactivity disorder. Wistar rats were trained in the 5CSRTT and treated with one of the three doses of DSP4 (10, 20 or 50 mg/kg) or saline. Following DSP4 treatment, rats were injected with three doses of atomoxetine (0.1, 0.5 or 1 mg/kg) or saline and tested in the 5CSRTT. The treatment with DSP4 caused a reduction in activity and a decline of performance in parameters related to attention in the 5CSRTT. Whether or not these impairments are due to attention deficits or changes in explorative behaviour and activity remains to be investigated. The treatment with atomoxetine had no beneficial effect on the rats’ performance regardless of the DSP4 treatment. The present findings support the role of noradrenaline in modulating attentional processes and call for future studies regarding the effects of atomoxetine on attention in rats.

Similar content being viewed by others
Change history
15 July 2017
An erratum to this article has been published.
References
Al Zahrani SS, Al Ruwaitea AS, Ho MY, Bradshaw CM, Szabadi E (1997) Destruction of central noradrenergic neurones with DSP4 impairs the acquisition of temporal discrimination but does not affect memory for duration in a delayed conditional discrimination task. Psychopharmacology 130:166–173
Arnsten AF (1997) Catecholamine regulation of the prefrontal cortex. J Psychopharmacol 11:151–162
Arnsten AF (2000) Genetics of childhood disorders: XVIII. ADHD, Part. 2: norepinephrine has a critical modulatory influence on prefrontal cortical function. J Am Acad Child Adolesc Psychiatry 39:1201–1203
Arnsten AF (2006) Fundamentals of attention-deficit/hyperactivity disorder: circuits and pathways. J Clin Psychiatry 67(Suppl 8):7–12
Arnsten AF (2011) Catecholamine influences on dorsolateral prefrontal cortical networks. Biol Psychiatry 69:e89–e99
Arnsten AF, Dudley AG (2005) Methylphenidate improves prefrontal cortical cognitive function through alpha2 adrenoceptor and dopamine D1 receptor actions: relevance to therapeutic effects in Attention Deficit Hyperactivity Disorder. Behav Brain Funct 1:2
Arnsten AF, Steere JC, Hunt RD (1996) The contribution of alpha 2-noradrenergic mechanisms of prefrontal cortical cognitive function. Potential significance for attention-deficit hyperactivity disorder. Arch Gen Psychiatry 53:448–455
Aston-Jones G, Rajkowski J, Kubiak P, Alexinsky T (1994) Locus coeruleus neurons in monkey are selectively activated by attended cues in a vigilance task. J Neurosci 14:4467–4480
Aston-Jones G, Rajkowski J, Kubiak P (1997) Conditioned responses of monkey locus coeruleus neurons anticipate acquisition of discriminative behavior in a vigilance task. Neuroscience 80:697–715
Bari A, Eagle DM, Mar AC, Robinson ES, Robbins TW (2009) Dissociable effects of noradrenaline, dopamine, and serotonin uptake blockade on stop task performance in rats. Psychopharmacology 205:273–283
Barkley RA (2006) Attention-deficit hyperactivity disorder: a handbook for diagnosis and treatment, vol 3. Guilford Press, New York
Bear MF (1999) Homosynaptic long-term depression: a mechanism for memory? Proc Natl Acad Sci USA 96:9457–9458
Benloucif S, Bennett EL, Rosenzweig MR (1995) Norepinephrine and neural plasticity: the effects of xylamine on experience-induced changes in brain weight, memory, and behavior. Neurobiol Learn Mem 63:33–42
Biederman J, Faraone SV (2005) Attention-deficit hyperactivity disorder. Lancet 366:237–248
Brozoski TJ, Brown RM, Rosvold HE, Goldman PS (1979) Cognitive deficit caused by regional depletion of dopamine in prefrontal cortex of rhesus monkey. Science 205:929–932
Bruno KJ, Freet CS, Twining RC, Egami K, Grigson PS, Hess EJ (2007) Abnormal latent inhibition and impulsivity in coloboma mice, a model of ADHD. Neurobiol Dis 25:206–216
Carli M, Robbins TW, Evenden JL, Everitt BJ (1983) Effects of lesions to ascending noradrenergic neurones on performance of a 5-choice serial reaction task in rats; implications for theories of dorsal noradrenergic bundle function based on selective attention and arousal. Behav Brain Res 9:361–380
Castellanos FX et al (1996) Quantitative brain magnetic resonance imaging in attention-deficit hyperactivity disorder. Arch Gen Psychiatry 53:607–616
Castellanos FX et al (2001) Quantitative brain magnetic resonance imaging in girls with attention-deficit/hyperactivity disorder. Arch Gen Psychiatry 58:289–295
Castellanos FX et al (2002) Developmental trajectories of brain volume abnormalities in children and adolescents with attention-deficit/hyperactivity disorder. JAMA 288:1740–1748
Cheetham SC, Viggers JA, Butler SA, Prow MR, Heal DJ (1996) [3H]nisoxetine–a radioligand for noradrenaline reuptake sites: correlation with inhibition of [3H]noradrenaline uptake and effect of DSP-4 lesioning and antidepressant treatments. Neuropharmacology 35:63–70
Clark CR, Geffen GM, Geffen LB (1987) Catecholamines and attention. I: animal and clinical studies. Neurosci Biobehav Rev 11:341–352
Cole BJ, Robbins TW (1989) Effects of 6-hydroxydopamine lesions of the nucleus accumbens septi on performance of a 5-choice serial reaction time task in rats: implications for theories of selective attention and arousal. Behav Brain Res 33:165–179
Collins P, Roberts AC, Dias R, Everitt BJ, Robbins TW (1998) Perseveration and strategy in a novel spatial self-ordered sequencing task for nonhuman primates: effects of excitotoxic lesions and dopamine depletions of the prefrontal cortex. J Cogn Neurosci 10:332–354
Compton DM, Dietrich KL, Smith JS, Davis BK (1995) Spatial and non-spatial learning in the rat following lesions to the nucleus locus coeruleus. NeuroReport 7:177–182
Coull JT (1994) Pharmacological manipulations of the alpha 2-noradrenergic system. Eff Cognit Drugs Aging 5:116–126
Crofts HS, Dalley JW, Collins P, Van Denderen JC, Everitt BJ, Robbins TW, Roberts AC (2001) Differential effects of 6-OHDA lesions of the frontal cortex and caudate nucleus on the ability to acquire an attentional set. Cereb Cortex 11:1015–1026
Delini-Stula A, Mogilnicka E, Hunn C, Dooley DJ (1984) Novelty-oriented behavior in the rat after selective damage of locus coeruleus projections by DSP-4, a new noradrenergic neurotoxin. Pharmacol Biochem Behav 20:613–618
Deroche V, Marinelli M, Maccari S, Le Moal M, Simon H, Piazza PV (1995) Stress-induced sensitization and glucocorticoids. I. Sensitization of dopamine-dependent locomotor effects of amphetamine and morphine depends on stress-induced corticosterone secretion. J Neurosci 15:7181–7188
Durston S et al (2004) Magnetic resonance imaging of boys with attention-deficit/hyperactivity disorder and their unaffected siblings. J Am Acad Child Adolesc Psychiatry 43:332–340
Everitt BJ, Robbins TW, Selden NR (2009) Functions of the locus coeruleus noradrenergic system: a neurobiological and behavioural synthesis. In: Heal DJ, Marsden CA (eds) The pharmacology of noradrenaline in the central nervous system. Oxford University Press, Oxford, pp 349–378
Faraone SV, Biederman J (1998) Neurobiology of attention-deficit hyperactivity disorder. Biol Psychiatry 44:951–958
Fritschy JM, Grzanna R (1991) Experimentally-induced neuron loss in the locus coeruleus of adult rats. Exp Neurol 111:123–127
Harley CW (1987) A role for norepinephrine in arousal, emotion and learning?: limbic modulation by norepinephrine and the Kety hypothesis. Prog Neuropsychopharmacol Biol Psychiatry 11:419–458
Harro J, Oreland L, Vasar E, Bradwejn J (1995) Impaired exploratory behaviour after DSP-4 treatment in rats: implications for the increased anxiety after noradrenergic denervation. Eur Neuropsychopharmacol 5:447–455
Jentsch JD, Aarde SM, Seu E (2009) Effects of atomoxetine and methylphenidate on performance of a lateralized reaction time task in rats. Psychopharmacology 202:497–504
Jones MD, Hess EJ (2003) Norepinephrine regulates locomotor hyperactivity in the mouse mutant coloboma. Pharmacol Biochem Behav 75:209–216
Jonge AD, van Meel JC, Timmermans PB, van Zwieten PA (1981) A lipophilic, selective alpha1 -adrenoceptor agonist: 2-(2-chloro-5-trifluoromethylphenylimino)imida-zolidine (St 587). Life Sci 28:2009–2016
Koda K, Ago Y, Cong Y, Kita Y, Takuma K, Matsuda T (2010) Effects of acute and chronic administration of atomoxetine and methylphenidate on extracellular levels of noradrenaline, dopamine and serotonin in the prefrontal cortex and striatum of mice. J Neurochem 114:259–270
Koffarnus MN, Katz JL (2011) Response requirement and increases in accuracy produced by stimulant drugs in a 5-choice serial reaction-time task in rats. Psychopharmacology 213:723–733
Lange KW, Reichl S, Lange KM, Tucha L, Tucha O (2010) The history of attention deficit hyperactivity disorder. Atten Defic Hyperact Disord 2:241–255
Langlais PJ, Connor DJ, Thal L (1993) Comparison of the effects of single and combined neurotoxic lesions of the nucleus basalis magnocellularis and dorsal noradrenergic bundle on learning and memory in the rat. Behav Brain Res 54:81–90
Liu YP, Huang TS, Tung CS, Lin CC (2015) Effects of atomoxetine on attention and impulsivity in the five-choice serial reaction time task in rats with lesions of dorsal noradrenergic ascending bundle. Prog Neuropsychopharmacol Biol Psychiatry 56:81–90
McGaugh JL (1989) Involvement of hormonal and neuromodulatory systems in the regulation of memory storage. Annu Rev Neurosci 12:255–287
Mefford IN, Potter WZ (1989) A neuroanatomical and biochemical basis for attention deficit disorder with hyperactivity in children: a defect in tonic adrenaline mediated inhibition of locus coeruleus stimulation. Med Hypotheses 29:33–42
Micheletti G, Warter JM, Marescaux C, Depaulis A, Tranchant C, Rumbach L, Vergnes M (1987) Effects of drugs affecting noradrenergic neurotransmission in rats with spontaneous petit mal-like seizures. Eur J Pharmacol 135:397–402
Navarra R, Graf R, Huang Y, Logue S, Comery T, Hughes Z, Day M (2008) Effects of atomoxetine and methylphenidate on attention and impulsivity in the 5-choice serial reaction time test. Prog Neuropsychopharmacol Biol Psychiatry 32:34–41
Pichler L, Kobinger W (1985) Possible function of alpha 1-adrenoceptors in the CNS in anaesthetized and conscious animals. Eur J Pharmacol 107:305–311
Pliszka SR, McCracken JT, Maas JW (1996) Catecholamines in attention-deficit hyperactivity disorder: current perspectives. J Am Acad Child Adolesc Psychiatry 35:264–272
Pothos EN, Hernandez L, Hoebel BG (1995) Chronic food deprivation decreases extracellular dopamine in the nucleus accumbens: implications for a possible neurochemical link between weight loss and drug abuse. Obes Res 3(Suppl 4):525S–529S
Raskin LA, Shaywitz BA, Anderson GM, Cohen DJ, Teicher MH, Linakis J (1983) Differential effects of selective dopamine, norepinephrine or catecholamine depletion on activity and learning in the developing rat. Pharmacol Biochem Behav 19:743–749
Renner TJ, Gerlach M, Romanos M, Herrmann M, Reif A, Fallgatter AJ, Lesch KP (2008) Neurobiology of attention-deficit hyperactivity disorder. Nervenarzt 79:771–781
Riekkinen P Jr, Lammintausta R, Ekonsalo T, Sirvio J (1993) The effects of alpha 2-adrenoceptor stimulation on neocortical EEG activity in control and 6-hydroxydopamine dorsal noradrenergic bundle-lesioned rats. Eur J Pharmacol 238:263–272
Robbins TW, Roberts AC (2007) Differential regulation of fronto-executive function by the monoamines and acetylcholine. Cereb Cortex 17(Suppl 1):i151–i160
Roberts DC, Price MT, Fibiger HC (1976) The dorsal tegmental noradrenergic projection: an analysis of its role in maze learning. J Comp Physiol Psychol 90:363–372
Roberts AC, De Salvia MA, Wilkinson LS, Collins P, Muir JL, Everitt BJ, Robbins TW (1994) 6-Hydroxydopamine lesions of the prefrontal cortex in monkeys enhance performance on an analog of the Wisconsin Card Sort Test: possible interactions with subcortical dopamine. J Neurosci 14:2531–2544
Robinson ES et al (2008) Similar effects of the selective noradrenaline reuptake inhibitor atomoxetine on three distinct forms of impulsivity in the rat. Neuropsychopharmacology 33:1028–1037
Rosa Neto P, Lou H, Cumming P, Pryds O, Gjedde A (2002) Methylphenidate-evoked potentiation of extracellular dopamine in the brain of adolescents with premature birth: correlation with attentional deficit. Ann NY Acad Sci 965:434–439
Ross SB, Stenfors C (2015) DSP4, a selective neurotoxin for the locus coeruleus noradrenergic system. A review of its mode of action. Neurotox Res 27:15–30
Ruotsalainen S, Haapalinna A, Riekkinen PJ Sr, Sirvio J (1997) Dexmedetomidine reduces response tendency, but not accuracy of rats in attention and short-term memory tasks. Pharmacol Biochem Behav 56:31–40
Skrebuhhova T, Allikmets L, Matto V (1999) [3H]-ketanserin binding and elevated plus-maze behavior after chronic antidepressant treatment in DSP-4 and P-CPA pretreated rats: evidence for partial involvement of 5-HT2A receptors. Methods Find Exp Clin Pharmacol 21:483–490
Solanto MV (1998) Neuropsychopharmacological mechanisms of stimulant drug action in attention-deficit hyperactivity disorder: a review and integration. Behav Brain Res 94:127–152
Sontag TA, Hauser J, Kaunzinger I, Gerlach M, Tucha O, Lange KW (2008) Effects of the noradrenergic neurotoxin DSP4 on spatial memory in the rat. J Neural Transm (Vienna) 115:299–303
Sontag TA, Tucha O, Walitza S, Lange KW (2010) Animal models of attention deficit/hyperactivity disorder (ADHD): a critical review. Atten Defic Hyperact Disord 2:1–20
Sontag TA, Hauser J, Tucha O, Lange KW (2011) Effects of DSP4 and methylphenidate on spatial memory performance in rats. Atten Defic Hyperact Disord 3:351–358
Swanson JM et al (2007) Etiologic subtypes of attention-deficit/hyperactivity disorder: brain imaging, molecular genetic and environmental factors and the dopamine hypothesis. Neuropsychol Rev 17:39–59
Teicher MH, Barber NI, Reichheld JH, Baldessarini RJ, Finklestein SP (1986) Selective depletion of cerebral norepinephrine with 6-hydroxydopamine and GBR-12909 in neonatal rat. Brain Res 395:124–128
Thapar A, Cooper M (2016) Attention deficit hyperactivity disorder. Lancet 387:1240–1250. doi:10.1016/S0140-6736(15)00238-X
Tripp G, Wickens JR (2009) Neurobiology of ADHD. Neuropharmacology 57:579–589
Volkow ND et al (2001) Therapeutic doses of oral methylphenidate significantly increase extracellular dopamine in the human brain. J Neurosci 21:RC121
Wankerl B, Hauser J, Makulska-Gertruda E, Reissmann A, Sontag TA, Tucha O, Lange KW (2014) Neurobiology of attention deficit hyperactivity disorder. Fortschr Neurol Psychiatr 82:9–29
Wenk G, Hughey D, Boundy V, Kim A, Walker L, Olton D (1987) Neurotransmitters and memory: role of cholinergic, serotonergic, and noradrenergic systems. Behav Neurosci 101:325–332
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed. All experiments were performed in accordance with the national laws (German law on Protection of Animals) and the principles of laboratory animal care (NIH publication No. 86- 23, revised 1996). The experimental protocol was approved by the District Government of the Oberpfalz/Bavaria (Permit number 54-2531.1-14/07). The rats were handled according to the guidelines of the Federation for European Laboratory Animal Science Associations (FELASA).
Additional information
An erratum to this article is available at https://doi.org/10.1007/s12402-017-0235-8.
Rights and permissions
About this article
Cite this article
Hauser, J., Reissmann, A., Sontag, TA. et al. Effects of atomoxetine on attention in Wistar rats treated with the neurotoxin N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP4). ADHD Atten Def Hyp Disord 9, 253–262 (2017). https://doi.org/10.1007/s12402-017-0225-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12402-017-0225-x