Abstract
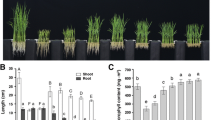
Previously, our work with ginseng hairy root shows that the tissue of low-branching and slow-growing phenotype contains high level of glutamine. In order to check if the high glutamine concentration inhibits the root growth, we applied exogenous glutamine or glutamate into growth medium and check the root growth of Arabidopsis. While glutamine did not affect root growth, over 0.1 mM glutamate inhibited severe root growth. However, when the amino acid solution was adjusted to pH 5.7 and added into medium, Arabidopsis seedlings show normal growth pattern on medium containing glutamate or aspartate. These results demonstrated that inhibition of the root growth by high concentration of exogenous glutamate was a result of the low pH toxicity caused by acidic amino acid, although low concentration (0.05 mM) of glutamate has an inhibitory effect on the primary root growth. The application of exogenous glutamine or glutamate increases glutamine concentration within root tissue about 3- to 4-fold. However, concentration of glutamate is not significantly increased. The KO mutant on each of the Gln1_1, Gln1_2, or Glu2 gene was little effective on the root growth. These results indicate that high concentration of endogenous glutamine observed in root tissue does not affect root growth.





Similar content being viewed by others
References
Casson SA, Lindsey K (2003) Genes and signaling in root development. New Phytol 158:11–38
Coschigano KT, Melo-Oliveira R, Lim J, Coruzzi GM (1998) Arabidopsis gls mutant and distinct Fd-GOGAT genes: implications for photorespiration and primary nitrogen assimilation. Plant Cell 10:741–752
Desnos T (2008) Root branching responses to phosphate and nitrate. Curr Op Plant Sci 11:82–87
Ericsson T (1995) Growth and shoot: root ratio of seedling in relation to nutrient availability. Plant Soil 168–169:205–214
Fawzy H, Overstreet R, Jacobson L (1954) The influence of hydrogen ion concentration on cation absorption by barley roots. Plant Physiol 29:234–237
Filleur S, Walch-Liu P, Gan Y, Forde BG (2005) Nitrate and glutamate sensing by plant roots. Biochem Soc Trans 33:283–286
Forde BG, Lea PJ (2007) Glutamate in plants: metabolism, regulation, and signaling. J Exp Bot 58:2339–2358
Fritz C, Mueller C, Matt P, Feil R, Stitt M (2006) Impact of the C–N status on the amino acid profile in tobacco source leaves. Plant Cell Environ 29:2055–2076
Geiger M, Walch-Liu P, Engels C, Harnecker J, Shulze ED, Ludewig F, Sonnewald U, Scheible WR, Stitt M (1998) Enhanced carbon dioxide leads to a modified diurnal rhythm of nitrate reductase activity in older plants, and a large stimulation of nitrate reductase activity and higher levels of amino acids in young tobacco plants. Plant Cell Environ 21:253–268
Ishiyama K, Inoue E, Watanabe-Takahashi A, Obara M, Yamaya T, Takakashi H (2004) Kinetic properties and ammonium dependent regulation of cytosolic isoenzymes of glutamine synthetase in Arabidopsis. J Biol Chem 279:16598–16605
Jung SM, Kim SW, Ban SH, In DS, Jung JD, Chung HJ, Liu JR, Lim YP, Choi DW (2006) Glutamine accumulation inhibits root growth and lateral root formation in ginseng hairy roots. Plant Sci 170:801–807
Koyama H, Toda T, Yokota S, Zuraida D, Hara T (1995) Effects of aluminum and pH on root growth and cell viability in Arabidopsis thaniana strain Landsberg in hydroponic culture. Plant Cell Physiol 36:201–205
Koyama H, Toda T, Hara T (2001) Brief exposure to low-pH stress causes irreversible damage to the growing root in Arabidopsis thaliana: pectin–Ca interaction may play an important in proton rhizotoxicity. J Exp Bot 52:361–368
Masclaux-Daubresse C, Carrayol E, Valadier MH (2005) The two nitrogen mobilization- and senescence-associated GS1 and GDH genes are controlled by C and N metabolites. Planta 221:580–588
Oliveira IC, Coruzzi GM (1999) Carbon and amino acids reciprocally modulate the expression of glutamine synthetase in Arabidopsis. Plant Physiol 121:301–309
Peterman TK, Goodman HM (1991) The glutamine synthetase gene family of Arabidopsis thaliana: light-regulation and differential expression in leaves, root and seeds. Mol Gen Gent 230:145–154
Scheneidereit J, Hausler RE, Fiene G, Kaiser WM, Weber APM (2006) Antisense repression reveals a crucial role of the plastidic 2-oxoglutarate/malate translocator DiT1 at the interface between carbon and nitrogen metabolism. Plant J 45:206–224
Taiz L, Zeiger E (2002) Plant physiology, 3rd edn. Sinauer, Sunderland
Walch-Liu P, Forde BG (2008) Nitrate signaling mediated by the NRT1.1 nitrate transporter antagonizes l-glutamate-induced changes in root architecture. Plant J 54:820–828
Walch-Liu P, Ivanov II, Filleur S, Gan Y, Remans T, Forde BG (2006a) Nitrogen regulation of root branching. Ann Bot 97:875–881
Walch-Liu P, Liu LH, Remans T, Tester M, Forde BG (2006b) Evidence that l-glutamate can act as an exogenous signal to modulate root growth and branching in Arabidopsis thaliana. Plant Cell Physiol 47:1045–1057
Watanabe A, Takagi N, Hayashi H, Chino M, Watanabe A (1997) Internal Gln/Glu ration as a potential regulatory parameter for the expression of a cytosolic glutamine synthetase of radish in cultured cells. Plant Cell Physiol 38:1000–1006
Zhang H, Forde BG (2000) Regulation of Arabidopsis root development by nitrate availability. J Exp Bot 51:51–59
Zhang H, Jennings A, Barlow PW, Forde BG (1999) Dual pathways for regulation of root branching by nitrate. Proc Natl Acad Sci 96:6529–6534
Zhu B, Coleman GD (2001) The poplar bark storage protein gene (Bspa) promoter is responsive to photoperiod and nitrogen in transgenic poplar and active in floral tissues, immature seeds and germinating seeds of transgenic tobacco. Plant Mol Biol 46:383–394
Acknowledgments
This study was financially supported by Chonnam National University, 2007
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, T.H., Kim, E.C., Kim, S.W. et al. Exogenous Glutamate Inhibits the Root Growth and Increases the Glutamine Content in Arabidopsis thaliana . J. Plant Biol. 53, 45–51 (2010). https://doi.org/10.1007/s12374-009-9084-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-009-9084-0