Abstract
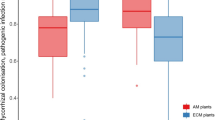
This study aimed to investigate the plant growth-promoting ability of the four most abundant AMF species isolated from the sugarcane rhizosphere, Acaulospora denticulata (ACD), Racocetra fulgida (RAF), Rhizophagus clarus (RHC) and Glomus sp.7 (GLS), on the cultivation of sugarcane under pot conditions. The results showed that GLS and RHC significantly increased root colonization, root morphology (root length, root surface area, root volume), P uptake, plant height and diameter, leaf area, relative water content, photosynthetic rate, stomatal conductance, transpiration rate and total biomass. In contrast, ACD and RAF only affected leaf area, plant height and root qualities. This is the first report of identification of ACD and RAF as the most abundant AMF species in sugarcane rhizosphere soils. Additionally, we are the first to show that these 2 AMF could affect some physiologies of sugarcane. Furthermore, Pearson's correlation analysis suggested that AMF root colonization was the most crucial factor affecting the plant growth parameters of sugarcane. This finding confirmed that inoculation with a specific AMF species could better enhance the growth of sugarcane. The results suggested that GLS and RHC could be used as effective biofertilizers for improving the growth of sugarcane under pot conditions.

Similar content being viewed by others
References
Alexandre, F.S., L.V.D. Flora, I.G. Henrique, D.C. da Silva, A.P. Mercedes, A.C. Silva, A.S. de Oliveira, M.P.B. da Silva, B.P.F. Ronning, D.R. Dreher, B.G. Cano, M.F.L. Andreata, J.B. Filho, E.R. Santos, F.H. Takisawa, R.F. Alfenas, G. Andrade, and M.V.T. Cely. 2021. Arbuscular mycorrhizal fungi (Rhizophagus clarus) and rhizobacteria (Bacillus subtilis) can improve the clonal propagation and development of teak for commercial plantings. Frontiers in Plant Science. https://doi.org/10.3389/fpls.2021.628769.
amf-phylogeny.com. 2018. Glomeromycota SPECIES LIST. http://www.amf-phylogeny.com/. Accessed October 1, 2018.
Baker, A., S.A. Ceasar, A.J. Palmer, J.B. Paterson, W. Qi, S.P. Muench, and S.A. Baldwin. 2015. Replace, reuse, recycle: Improving the sustainable use of phosphorus by plants. Journal of Experimental Botany 66 (12): 3523–3540.
Boonlue, S., W. Surapat, C. Pukahuta, P. Suwanarit, A. Suwanarit, and T. Morinaga. 2012. Diversity and efficiency of arbuscular mycorrhizal fungi in soils from organic chili (Capsicum frutescens) farms. Mycoscience 53 (1): 10–16.
Brandes, E.W. 1952. Botany of Sugarcane. C. van Dillewijn. Waltham, Mass.: Chronica Botanica; New York: Stechert-Hafner, 1952. 371 pp $6.00. Science 116 (3013): 333–333.
Calvet, C., A. Camprubi, A. Pérez-Hernández, and P. Lovato. 2013. Plant growth stimulation and root colonization potential of in vivo versus in vitro arbuscular mycorrhizal inocula. HortScience 48 (7): 897–901.
Chen, J., H. Zhang, X. Zhang, and M. Tang. 2017. Arbuscular mycorrhizal symbiosis alleviates salt stress in black locust through improved photosynthesis, water status, and K+/Na+ homeostasis. Frontiers in Plant Science 8:1739. https://doi.org/10.3389/fpls.2017.01739.
Cordell, D., J.O. Drangert, and S. White. 2009. The story of phosphorus: Global food security and food for thought. Global Environmental Change 19 (2): 292–305.
Daniels, B.A., and H.D. Skipper. 1982. Method for the recovery and quantitative estimation of propagules from soil. In Method and principle of micorrhizal research, ed. N.C. Schenck, 29–36. Minessotta: American Phytopathological Society.
Fors, R.O., O.J.S. Júnior, M.A.C. Carneiro, and R.L.L. Berbara. 2020. Selection of arbuscular mycorrhizal fungi for sugarcane in four soils with the presence of darkseptate endophytes. Acta Scientiarum: Agronomy 42(1): e42477. https://doi.org/10.4025/actasciagron.v42i1.42477.
Gavito, M.E., I. Jakobsen, T.N. Mikkelsen, and F. Mora. 2019. Direct evidence for modulation of photosynthesis by an arbuscular mycorrhiza-induced carbon sink strength. New Phytologist 223: 896–907.
Gerdemann, J.W., and T.H. Nicolson. 1963. Spores of mycorrhizal Endogone species extracted from soil by wet sieving and decanting. Transactions of the British Mycological Society 46 (2): 235–244.
Gilbert, N. 2009. Environment: The disappearing nutrient. Nature 461: 716–718.
Goetten, L.C., G. Moretto, and S.L. Stürmer. 2016. Influence of arbuscular mycorrhizal fungi inoculum produced on-farm and phosphorus on growth and nutrition of native woody plant species from Brazil. Acta Botanica Brasilica 30 (1): 9–16.
Hart, M., and R. Reader. 2002. Taxonomic basis for variation in the colonization strategy of arbuscular mycorrhizal fungi. New Phytologist 153 (2): 335–344.
Hoagland, D.R. and D.I. Arnon. 1950. The water-culture method for growing plants without soil. California Agricultural Experiment Station, Circular-347.
Jangpromma, N., P. Songsri, S. Thammasirirak, and P. Jaisil. 2010. Rapid assessment of chlorophyll content in sugarcane using a SPAD chlorophyll meter across different water stress conditions. Asian Journal of Plant Sciences 9: 368–374.
Jung, S.C., A. Martinez-Medina, J.A. Lopez-Raez, and M.J. Pozo. 2012. Mycorrhiza-induced resistance and priming of plant defenses. Journal of Chemical Ecology 38 (6): 651–664.
Juntahum, S., N. Jongrungklang, W. Kaewpradit, S. Lumyong, and S. Boonlue. 2020. Impact of arbuscular mycorrhizal fungi on growth and productivity of sugarcane under field conditions. Sugar Tech 22 (3): 451–459.
Kapoor, R., D. Sharma, and A.K. Bhatnagar. 2008. Arbuscular mycorrhizae in micropropagation systems and their potential applications. Scientia Horticulturae 116 (3): 227–239.
Kingston, G. 2014. Mineral nutrition of sugarcane. In Sugarcane: Physiology, biochemistry, and functional biology, ed. P.H. Moore and F.C. Botha, 85–120. Oxford: John Wiley & Sons.
Koske, R.E., and J.N. Gemma. 1989. A modified procedure for staining roots to detect VA mycorrhizas. Mycological Research 92: 486–488.
Kumalawati, Z., Y. Musa, N. Amin, L. Asrul, and I. Ridwan. 2014. Exploration of arbuscular mycorrhizal fungi from sugarcane rhizosphere in south Sulawesi. International Journal of Scientific & Technology Research 3 (1): 201–203.
Kusumi, K., S. Hirotsuka, T. Kumamaru, and K. Iba. 2012. Increased leaf photosynthesis caused by elevated stomatal conductance in a rice mutant deficient in SLAC1, a guard cell anion channel protein. Journal of Experimental Botany 63 (15): 5635–5644.
Lambers, H., C. Mougel, B. Jaillard, and P. Hinsinger. 2009. Plant-microbe-soil interactions in the rhizosphere: An evolutionary perspective. Plant and Soil 321: 83–115.
Matin, M.A., J.H. Brown, and H. Fergunson. 1989. Leaf water potential, relative water content, and diffusive resistance as screening techniques for drought resistance in barley. Agronomy Journal 81: 100–105.
Middleton, E.L., S. Richardson, L. Koziol, C.E. Palmer, Z. Yermakov, J.A. Henning, P.A. Schultz, and J.D. Bever. 2015. Locally adapted arbuscular mycorrhizal fungi improve vigor and resistance to herbivory of native prairie plant species. Ecosphere. https://doi.org/10.1890/ES15-00152.1.
Miller, R.M., S.P. Miller, J.D. Jastrow, and C.B. Rivetta. 2002. Mycorrhizal mediated feedbacks influence net carbon gain and nutrient uptake in Andropogon gerardii. New Phytologist 155: 149–162.
Mishra, B.S., M. Singh, and A. Laxmi. 2009. Glucose and auxin signaling interaction in controlling Arabidopsis thaliana seedlings root growth and development. PLoS ONE 4(2): e4502. https://doi.org/10.1371/journal.pone.0004502.
Muniyamma, M., B.K. Barti, and C.N. Reddy. 2000. Effect of VAM on root induction in vitro sugarcane (Saccharum officinarum L.) seedlings-a new technologue. Mycorrhiza News 12 (1): 13–16.
Nimasow, O.D., and R.K. Singh. 2020. Effect of arbuscular mycorrhizal inoculation on growth of chili plant (Capsicum frutescens L.) in nitrogens amended soil. Plant Cell Biotechnology and Molecular Biology 21 (41–42): 35–50.
Pontes, J.S., F. Oehl, F. Marinho, D. Coyne, and D.K.A.da Silva, A.M. Yano-Melo, and L.C. Maia,. 2017. Diversity of arbuscular mycorrhizal fungi in Brazil’s Caatinga and experimental agroecosystems. Biotropica 49 (3): 413–427.
Ramírez, J.G., L. Osorno, and N.W. Osorio. 2016. Presence of mycorrhizal fungi and a fluorescent Pseudomonas sp. in the rhizosphere of cacao in two agroecosystems and their effects on cacao seedling growth. Agronomía Colombiana 34 (3): 385–392.
Rokni, N., and E.M. Goltapeh. 2011. Diversity of arbuscular mycorrhizal fungi associated with common sugarcane varieties in Iran. International Journal of Agricultural Technology 7 (4): 1017–1022.
Rouphael, Y., P. Franken, C. Schneider, D. Schwarz, M. Giovannetti, and M. Agnolucci. 2015. Arbuscular mycorrhizal fungi act as bio-stimulants in horticultural crops. Scientia Horticulturae 196: 91–108.
Schenck, N.C., and Y. Pérez. 1990. Manual for the identification of VA mycorrhizal fungi. Florida, USA: Gainesville.
Shahbandeh M. 2021a. Global sugar production by leading country 2020/2021a. https://www.statista.com/statistics/495973/sugar-production-worldwide/. Accessed October 1, 2021a.
Shahbandeh M. 2021b. Sugar: exports of major countries 2020/2021b. https://www.statista.com/statistics/273437/exported-amount-of-sugar-in-leading-countries/. Accessed October 1, 2021b.
Sheng, M., M. Tang, H. Chan, B. Yang, F. Zhang, and Y. Huang. 2008. Influence of arbuscular mycorrhizae on photosynthesis and water status of maize plants under salt stress. Mycorrhiza 18: 287–296.
Smith, S.E., and D.J. Read. 2008. Mycorrhizal symbiosis, 3rd ed. London: Academic Press.
Sulistiono, W., B. Brahmantyo, S. Hartanto, H.B. Aji, and H.K. Bima. 2020. Effect of arbuscular mycorrhizal fungi and NPK fertilizer on roots growth and nitrate reductase activity of coconut. Journal of Agronomy 19: 46–53.
Sun, Z., J. Song, X. Xin, X. Xie, and B. Zhao. 2018. Arbuscular mycorrhizal fungal proteins 14–3-3- are involved in arbuscule formation and responses to abiotic stresses during AM symbiosis. Frontiers in Microbiology 9: 91. https://doi.org/10.3389/fmicb.2018.00091.
Tommerup, I.C., and L.K. Abbott. 1981. Prolonged survival and viability of VA mycorrhizal hyphae after root death. Soil Biology and Biochemistry 13: 431–433.
Tong, Z., G. Quan, L. Wan, F. He, and X. Li. 2019. The effect of fertilizers on biomass and biodiversity on a semi-arid grassland of Northern China. Sustainability 11(10): 2854. https://doi.org/10.3390/su11102854.
Trouvelot, A., J. Kough, and V. Gianinazzi-Pearson. 1986. Mesure d’un système radiculaire. Recherche de méthodes d’estimation ayant une signification fonctionnelle. In Physiological and genetical aspects of mycorrhizae, ed. V. Gianinazzi-Pearson and S. Gianinazzi, 217–221. Paris: INRA Press.
USDA. 2021. Sugar: World markets and trade. Usda, 1–8. Retrieved from https://www.fas.usda.gov/data/sugar-world-markets-and-trade. Accessed October 1, 2021.
Usuda, H. 2002. Evaluation of the effect of photosynthesis on biomass production with simultaneous analysis of growth and continuous monitoring of CO2 exchange in the whole plants of Radish, cv Kosena under ambient and elevated CO2. Plant Production Science 7 (4): 386–396.
Walan, P., S. Davidsson, S. Johansson, and M. Höök. 2014. Phosphate rock prodution and depletion: Regional disaggregated modeling and global implications. Resources, Conservation and Recycling 93: 178–187.
Wu, Q.S., A.K. Srivastava, and Y. Li. 2015. Effects of mycorrhizal symbiosis on growth behavior and carbohydrate metabolism of trifoliate orange under different substrate P levels. Journal of Plant Growth Regulation 34 (3): 495–508.
Yildiz, A. 2010. A native Glomus sp. from fields in Aydın province and effects of native and commercial mycorrhizal fungi inoculants on the growth of some vegetables. Turkish Journal of Biology 34: 447–452.
Acknowledgements
This research was financially supported by the Research and Researcher for Industry (RRI) with project code PHD60I0056, under funding agency of Thailand Science Research and Innovation (TSRI). We also thank the Northeast Thailand Cane and Sugar Research Center, Salt-tolerant Rice Research Group Khon Kaen University, Thailand, and Centre of Excellence on Biodiversity (BDC), Office of Higher Education Commission under project code BDC-PG1-163002 for partial support of financial, implements and instruments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Juntahum, S., Ekprasert, J. & Boonlue, S. Efficiency of Arbuscular Mycorrhizal Fungi for the Growth Promotion of Sugarcane Under Pot Conditions. Sugar Tech 24, 1738–1747 (2022). https://doi.org/10.1007/s12355-022-01129-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-022-01129-z