Abstract
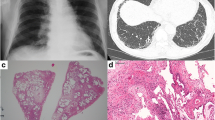
Revised idiopathic pulmonary fibrosis treatment guidelines were published in 2015, and nintedanib was conditionally recommended. Although diarrhea is reported to be a common major adverse event associated with nintedanib, there have been few reports on detailed endoscopic findings of nintedanib-associated enterocolitis. A 74-year-old woman was diagnosed with idiopathic pulmonary fibrosis 4 years ago in May. She was started on nintedanib (300 mg). Three months later, hepatic dysfunction was observed; therefore, the drug was temporarily discontinued and then resumed at a dose reduction of 200 mg. Five months later, the patient developed diarrhea, and the dose was reduced to 150 mg. However, no effect was noted; hence, colonoscopy was performed. Various inflammatory lesions, such as erythema and erosions, were observed continuously at the rectum, which resembled ulcerative colitis. No improvement was observed 2 months after follow-up colonoscopy, and nintedanib-related enterocolitis was suspected. The dose was further reduced to 100 mg. Since the endoscopic findings of nintedanib-associated enterocolitis are similar to those of ulcerative colitis, it is critical to consider patients with diarrhea who are taking nintedanib as having associated enterocolitis and attempt to reduce or discontinue the drug if diarrhea does not improve with antidiarrheal agents.




Similar content being viewed by others
Data availability
No data were generated in the writing of this case report.
References
Richeldi L, du Bois RM, Raghu G, et al. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med. 2014;370:2071–82.
Corte T, Bonella F, Crestani B, et al. Safety, tolerability and appropriate use of nintedanib in idiopathic pulmonary fibrosis. Respir Res. 2015;16:116.
The American Thoracic Society (ATS), The European Respiratory Society (ERS). Idiopathic pulmonary fibrosis: diagnosis and treatment International consensus statement. Am J Respir Crit Care Med. 2000;161:646–64.
Raghu G, Rochwerg B, Zhang Y, et al. An official ATS/ERS/JRS/ALAT clinical practice guideline: treatment of idiopathic pulmonary fibrosis. An update of the 2011 clinical practice guideline. Am J Respir Crit Care Med. 2015;192:3–19.
Richeldi L, Costabel U, Selman M, et al. Efficacy of a tyrosine kinase inhibitor in idiopathic pulmonary fibrosis. N Engl J Med. 2011;365:1079–87.
Fuchs CS, Tomasek J, Yong CJ, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2014;383:31–9.
Anonymous. Nintedanib. Aust Prescr. 2016; 39:62–3.
Amini A, Koury E, Chahla E. Nintedanib-induced colitis treated effectively with budesonide. Cureus. 2020;12: e9489.
Sejben A, Sejben I, Budai A, et al. Inflammatory bowel disease-mimicking colitis associated with nintedanib-based therapy in a lung cancer patient. Int J Surg Pathol. 2023;31:1326–8.
Oda K, Matsunaga T, Sennari K, et al. Colitis associated with nintedanib therapy for idiopathic pulmonary fibrosis (IPF). Intern Med. 2017;56:1267–8.
Chandler RE. Nintedanib and ischemic colitis: signal assessment with the integrated use of two types of real-world evidence, spontaneous reports of suspected adverse drug reactions, and observational data from large health-care databases. Pharmacoepidemiol Drug Saf. 2020;29:951–7.
Peterson J, Haberstroh W, Teslova T. Nintedanib-induced ischemic colitis masquerading as inflammatory bowel disease. Am J Gastroenterol. 2021;116:S805.
Kikuchi H, Sakuraba H, Akemoto Y, et al. A case of nivolumab-associated colitis, which relapsed after mucosal healing and was then successfully treated with mesalazine. Immunol Med. 2019;42:39–44.
Acknowledgements
We thank our patient for her invaluable contributions to this project.
Funding
This report did not receive any external funding.
Author information
Authors and Affiliations
Contributions
Manuscript drafting: KM, RS and KI; endoscopists: KM, YT, and HI; supervision: SS, MN, HN, and HI. The final version of the manuscript was read and approved by all authors.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest regarding the publication of this paper.
Human/animal Rights
This study was conducted in accordance with the Declaration of Helsinki and with approval by the relevant institutional review board.
Informed Consent
The patient provided informed consent for the publication of this report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miyaguchi, K., Tsuzuki, Y., Uemuara, H. et al. Nintedanib-associated enterocolitis with intractable diarrhea: a case report. Clin J Gastroenterol 17, 271–275 (2024). https://doi.org/10.1007/s12328-023-01894-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-023-01894-8