Abstract
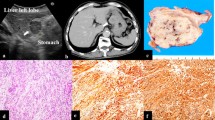
Gastric glomus tumors are extremely rare, constituting approximately 1% of gastric mesenchymal tumors. We report the case of a 45-year-old female patient in whom upper gastrointestinal endoscopy revealed a 3-cm submucosal tumor with a bridging fold in the posterior wall of the antrum of the stomach. Contrast-enhanced computed tomography revealed the tumor to be hypervascular in the arterial phase and exhibit continuous enhancement in the post-venous phase. Dynamic contrast-enhanced magnetic resonance imaging showed that the tumor was hypervascular in the early phase and persistently enhanced in the late phase. Endoscopic ultrasonography revealed a mosaic echo pattern. Endoscopic ultrasound-guided fine-needle aspiration biopsy was performed. Immunohistochemical examination revealed that the tumor was positive for α-smooth muscle actin and synaptophysin, but negative for CD56, chromogranin A and C-kit. Furthermore, 1–2% of the tumor cell nuclei were Ki-67-positive. Thus, the preoperative diagnosis was a benign glomus tumor. The patient then underwent non-exposed endoscopic wall-inversion surgery (NEWS), and the tumor was completely resected. In conclusion, NEWS is an effective method for the treatment of gastric glomus tumors.





Similar content being viewed by others
References
Devanathan M, Jamuna Rani S, Chandramohan SM, et al. A rare submucosal tumour of stomach-glomus tumour: a case report. Int J Surg Case Rep. 2015;11:64–7.
Fang HQ, Yang J, Zhang FF, et al. Clinicopathological features of gastric glomus tumor. World J Gastroenterol. 2010;16:4616–20.
Orellana F, Onetto C, Balbontin P, et al. Gastric glomus tumor: report of one case and review. Endoscopy. 2011;43(Suppl 2 UCTN):E71–2.
Miettinen M, Paal E, Lasota J, et al. Gastrointestinal glomus tumors: a clinicopathologic, immunohistochemical, and molecular genetic study of 32 cases. Am J Surg Pathol. 2002;26:301–11.
Wang ZB, Yuan J, Shi HY. Features of gastric glomus tumor: a clinicopathologic, immunohistochemical and molecular retrospective study. Int J Clin Exp Pathol. 2014;7:1438–48.
Folpe AL, Fanburg-Smith JC, Miettinen M, et al. Atypical and malignant glomus tumors: analysis of 52 cases, with a proposal for the reclassification of glomus tumors. Am J Surg Pathol. 2001;25:1–12.
Zaidi S, Arafah M. Malignant gastric glomus tumor: a case report and literature review of a rare entity. Oman Med J. 2016;31:60–4.
Song SE, Lee CH, Kim KA, et al. Malignant glomus tumor of the stomach with multiorgan metastases: report of a case. Surg Today. 2010;40:662–7.
Teng TH, Huang SH, Liang CW. Malignant gastric glomus tumour mimicking GIST. Pathology. 2012;44:261–3.
Hiki N, Yamamoto Y, Fukunaga T, et al. Laparoscopic and endoscopic cooperative surgery for gastrointestinal stromal tumor dissection. Surg Endosc. 2008;22:1729–35.
Goto O, Takeuchi H, Kawakubo H, et al. Feasibility of non-exposed endoscopic wall-inversion surgery with sentinel node basin dissection as a new surgical method for early gastric cancer: a porcine survival study. Gastric Cancer. 2015;18:440–5.
Nakajo K, Chonan A, Tsuboi R, et al. A case of a glomus tumor of the stomach resected by laparoscopy endoscopy cooperative surgery. Nihon Shokakibyo Gakkai Zasshi. 2016;113:1557–63.
Thambi R, Sheeja S, Joesph CP, et al. Gastric glomus tumor: a brief report. Indian J Pathol Microbiol. 2014;57:509–10.
Ando N, Goto H, Niwa Y, et al. The diagnosis of GI stromal tumors with EUS-guided fine needle aspiration with immunohistochemical analysis. Gastrointest Endosc. 2002;55:37–43.
Okubo K, Yamao K, Nakamura T, et al. Endoscopic ultrasound-guided fine-needle aspiration biopsy for the diagnosis of gastrointestinal stromal tumors in the stomach. J Gastroenterol. 2004;39:747–53.
Chatzipantelis P, Salla C, Konstantinou P, et al. Endoscopic ultrasound-guided fine-needle aspiration cytology of pancreatic neuroendocrine tumors: a study of 48 cases. Cancer. 2008;114:255–62.
Kato S, Kikuchi K, Chinen K, et al. Diagnostic utility of endoscopic ultrasound-guided fine-needle aspiration biopsy for glomus tumor of the stomach. World J Gastroenterol. 2015;21:7052–8.
Haque S, Modlin IM, West AB. Multiple glomus tumors of the stomach with intravascular spread. Am J Surg Pathol. 1992;16:291–9.
Lin YM, Chiu NC, Li AF, et al. Unusual gastric tumors and tumor-like lesions: radiological with pathological correlation and literature review. World J Gastroenterol. 2017;23:2493–504.
Baek YH, Choi SR, Lee BE, et al. Gastric glomus tumor: analysis of endosonographic characteristics and computed tomographic findings. Dig Endosc. 2013;25:80–3.
Cha SH, Cho SB, Kim YW, et al. Helical CT appearance of glomus tumor of the stomach. Eur Radiol. 2000;10:671–3.
Tang M, Hou J, Wu D, et al. Glomus tumor in the stomach: computed tomography and endoscopic ultrasound findings. World J Gastroenterol. 2013;19:1327–9.
Patel TH, Horton KM, Hruban RH, et al. Glomus tumor of the stomach: depiction by multidetector CT and three-dimensional volume rendering imaging. Case Rep Med. 2010;2010:126095.
Matsuda T, Nunobe S, Ohashi M, et al. Laparoscopic endoscopic cooperative surgery (LECS) for the upper gastrointestinal tract. Transl Gastroenterol Hepatol. 2017;2:40.
Kikuchi S, Nishizaki M, Kuroda S, et al. Nonexposure laparoscopic and endoscopic cooperative surgery (closed laparoscopic and endoscopic cooperative surgery) for gastric submucosal tumor. Gastric Cancer. 2017;20:553–7.
Nunobe S, Hiki N, Gotoda T, et al. Successful application of laparoscopic and endoscopic cooperative surgery (LECS) for a lateral-spreading mucosal gastric cancer. Gastric Cancer. 2012;15:338–42.
Goto O, Takeuchi H, Kawakubo H, et al. First case of non-exposed endoscopic wall-inversion surgery with sentinel node basin dissection for early gastric cancer. Gastric Cancer. 2015;18:434–9.
Inoue H, Ikeda H, Hosoya T, et al. Endoscopic mucosal resection, endoscopic submucosal dissection, and beyond: full-layer resection for gastric cancer with nonexposure technique (CLEAN-NET). Surg Oncol Clin N Am. 2012;21:129–40.
Goto O, Mitsui T, Fujishiro M, et al. New method of endoscopic full-thickness resection: a pilot study of non-exposed endoscopic wall-inversion surgery in an ex vivo porcine model. Gastric Cancer. 2011;14:183–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Masahide Ebi, Tomoya Sugiyama, Kazuhiro Yamamoto, Takuya Saito, Tadahisa Inoue, Yoshiharu Yamaguchi, Yasuhiro Tamura, Shinya Izawa, Yasutaka Hijikata, Yasushi Funaki, Naotaka Ogasawara, Makoto Sasaki, and Kunio Kasugai declare that they have no conflict of interest.
Human rights
All procedures followed have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was obtained from this case report.
Rights and permissions
About this article
Cite this article
Ebi, M., Sugiyama, T., Yamamoto, K. et al. A gastric glomus tumor resected using non-exposed endoscopic wall-inversion surgery. Clin J Gastroenterol 10, 508–513 (2017). https://doi.org/10.1007/s12328-017-0782-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-017-0782-8