Abstract
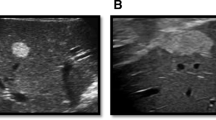
A 52-year-old man with a 9-year history of hepatic hemangioma was treated with the anti-diabetic drug metformin, resulting in complete remission of the tumor. In 2006, a hemangioma with diameter of 20 × 25 mm was detected incidentally in the liver. The results of imaging studies including ultrasound (US), computed tomography (CT) and magnetic resonance imaging (MRI) were all compatible with that of hepatic hemangioma. The patient consequently underwent imaging annually from 2006 to 2015. The tumor size increased slightly, to 30 × 35 mm in 2012; however, the general tumor characteristics in imaging were not changed. Beginning May 2012, metformin (750 mg/day) was administered because of an increase in blood sugar and hemoglobin A1c levels. After the start of metformin treatment, the tumor size on US gradually decreased. Finally, in October 2015, the tumor was no longer detected. Dynamic CT study also demonstrated markedly reduced tumor size, with a decrease of 2–3 mm in diameter. These results indicate that metformin treatment strongly suppressed cell proliferation in liver hemangioma. The anti-angiogenic effect of metformin was indicated as a possible cause of the reduction in tumor size.



Similar content being viewed by others
References
Sacco F, Silvestri A, Posca D, et al. Deep proteomics of breast cancer cells reveals that metformin rewires signaling networks away from a pro-growth state. Cell Syst. 2016;2:159–71.
Calle EE, Rodriguez C, Walker-Thurmond K, et al. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of US adults. N Eng J Med. 2003;348:1625–38.
Rui R, Lou J, Zou L, et al. Excess body mass index and risk of liver cancer: a nonlinear dose-response meta-analysis of prospective studies. PLoS One. 2012;7:e44522.
White DL, Kanwal F, El-Serag HB. Association between non-alcoholic fatty liver disease and risk for hepatocellular cancer, based on systematic review. Clin Gastroenterol Hepatol. 2012;10(1342–59):e2.
El-Serag HB, Hampel H, Javadi F. The association between diabetes and hepatocellular carcinoma: a systematic review of epidemiologic evidence. Clin Gastroenterol Hepatol. 2006;4:369–80.
Fujita K, Iwama H, Miyoshi H, et al. Diabetes mellitus and metformin in hepatocellular carcinoma. Word J Gastroenterol. 2016;22:6100–13.
Sage BP, Laumonier H, Laurent C, et al. Benign and malignant vascular tumors of the liver in adults. Semin Liver Dis. 2008;28:302–14.
Gandolfi L, Leo P, Solmi L, et al. Natural history of hepatic hemangiomas: clinical and ultrasound study. Gut. 1991;32:677–80.
Mezhir JJ, Fourman LT, Do RK, et al. Changes in the management of benign liver tumours: an analysis of 285 patients. HPB (Oxford). 2013;15:156–63.
Lee HL, Chung TS, Chen SW, et al. CT and MRI findings correlate with the time-course of unresectable cavernous haemangioma of the liver after fractionated radiotherapy. Br J Radiol. 2012;85:e49–52.
Medici D, Olsen BR. Rapamycin inhibits proliferation of hemangioma endothelial cells by reducing HIF-1-dependent expression of VEGF. PLoS One. 2012;7:e42913.
Tadakawa M, Takeda T, Li B, et al. The anti-diabetic drug metformin inhibits vascular endothelial growth factor expression via the mammalian target of rapamycin complex 1/hypoxia-inducible factor-1α signaling pathway in ELT-3 cells. Mol Cell Endocrinol. 2015;399:1–8.
Giovannucci E, Harlan DM, Archer MC, et al. Diabetes and cancer: a consensus report. Diabetes Care. 2010;33:1674–85.
Jalving M, Gietema JA, Lefrandt JD, et al. Metformin: taking away the candy for cancer? Eur J Cancer. 2010;46:2369–80.
Bhat M, Sonenberg N, Gores GJ. The mTOR pathway in hepatic malignancies. Hepatology. 2013;58:810–8.
Kelly B, Tannahill GM, Murphy MP, et al. Metformin inhibits the production of reactive oxygen species from NADH: ubiquinone oxidoreductase to limit induction of interleukin-1β (IL-1β) and boosts interleukin-10 (IL-10) in lipopolysaccharide (LPS)-activated macrophage. J Biol Chem. 2015;290:20348–59.
Dowling RJ, Zakikhani M, Fantus IG, et al. Metformin inhibits mammalian target of rapamycin-dependent translation initiation in breast cancer cells. Cancer Res. 2007;7:10804–12.
Gotlieb WH, Saumet J, Beauchamp MC, et al. In vitro metformin anti-neoplastic activity in epithelial ovarian cancer. Gynecol Oncol. 2008;110:246–50.
Zakikhani M, Dowling R, Fantus IG, et al. Metformin is an AMP kinase-dependent growth inhibitor for breast cancer cells. Cancer Res. 2006;66:10269–73.
Kalender A, Selvaraj A, Kim SY, et al. Metformin, independent of AMPK, inhibits mTORC1 in a rag GTPase-dependent manner. Cell Metab. 2010;11:390–401.
Xavier DO, Amaral LS, Gomes MA, et al. Metformin inhibits inflammatory angiogenesis in a murine sponge model. Biomed Pharmacother. 2010;64:220–5.
Ersoy C, Kiyici S, Budak F, et al. The effect of metformin treatment on VEGF and PAI-1 levels in obese type 2 diabetic patients. Diabetes Res Clin Pract. 2008;81:56–60.
Vazquez-MA Oliveras FC, Menendez JA. The antidiabetic drug metformin suppresses HER2 (erbB-2) oncoprotein overexpression via inhibition of the mTOR effector p70S6K1 in human breast carcinoma cells. Cell Cycle. 2009;8:88–96.
Sahra B, Laurent K, Loubat A, et al. The antidiabetic drug metformin exerts an antitumoral effect in vitro and in vivo through a decrease of cyclin D1 level. Oncogene. 2008;27:3576–86.
Sahra B, Regazzetti C, Robert G, et al. Metformin, independent of AMPK, induces mTOR inhibition and cell-cycle arrest through REDD1. Cancer Res. 2011;71:4366–72.
Isakovic A, Harhaji L, Stevanovic D, et al. Dual antiglioma action of metformin: cell cycle arrest and mitochondria-dependent apoptosis. Cell Mol Life Sci. 2007;64:1290–302.
Liu B, Fan Z, Edgerton SM, et al. Metformin induces unique biological and molecular responses in triple negative breast cancer cells. Cell Cycle. 2009;8:2031–40.
Blandino G, Valerio M, Cioce M, et al. Metformin elicits anticancer effects through the sequential modulation of DICER and c-MYC. Nat Commun. 2012;3:865.
Li J, Hernanda PY, Bramer WM, et al. Anti-tumor effects of metformin in animal models of hepatocellular carcinoma: a systematic review and meta-analysis. PLoS One. 2015;10:e0127967.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest:
The authors declare that they have no conflict of interest.
Human Rights:
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008(5).
Informed Consent:
Informed consent was obtained from the patient to be included in the study.
Rights and permissions
About this article
Cite this article
Ono, M., Sawada, K. & Okumura, T. A case of liver hemangioma with markedly reduced tumor size after metformin treatment: a case report. Clin J Gastroenterol 10, 63–67 (2017). https://doi.org/10.1007/s12328-016-0705-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-016-0705-0