Abstract
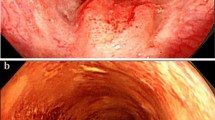
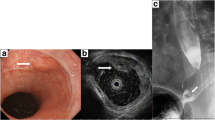
We report on a 53-year-old male with esophageal cancer. He had no evidence of distant metastasis, and received a subtotal esophagectomy. Histopathologically, the tumors were contiguous with Barrett’s epithelium. Undifferentiated carcinoma components existed independently of differentiated adenocarcinoma components. Undifferentiated carcinoma was present proximal to the esophagogastric junction. Both tumors had invaded the submucosa and were associated with a prominent lymphoid stroma. Metastasis from undifferentiated carcinoma was found in the paraesophageal lymph nodes. Immunohistochemically, both components were negative for 34bE12 and positive for CAM5.2 and showed nearly identical staining patterns for p53, indicating that the tumors were derived from Barrett’s epithelium. Because the undifferentiated carcinoma did not express CK20 or carcinoembryonic antigen, the properties of adenocarcinoma had apparently been lost during the process of tumor cell progression. This is the first report of undifferentiated carcinoma associated with Barrett’s esophagus with adenocarcinoma.


Similar content being viewed by others
References
Mori S, Tagaya N, Hamada K, Nakagawa A, Sawada T, Honma K, et al. Undifferentiated carcinoma of the esophagus with rapid growth of lymph node. A case report and review of the literature. Tumori. 2008;94:411–5.
Suwa T, Yoshida M, Kubota K, Kuroda J, Sakuma M, Kitajima M. Successful treatment for undifferentiated carcinoma of the esophagus: a case with complete response to chemoradiotherapy. Esophagus. 2008;5:51–5.
Paraf F, Fléjou JF, Pignon JP, Fékété F, Potet F. Surgical pathology of adenocarcinoma arising in Barrett’s esophagus. Analysis of 67 cases. Am J Surg Pathol. 1995;19:183–91.
Maezato K, Nishimaki T, Oshiro M, Yamashiro T, Sasaki H, Sashida Y. Signet-ring cell carcinoma of the esophagus associated with Barrett’s epithelium: report of a case. Surg Today. 2007;37:1096–101.
Murata S, Akaboshi K, Kumashiro Y, Yao T, Kimura M, Matsui N, et al. Vimentin-positive esophageal undifferentiated carcinoma of the non-small-cell type arising in Barrett’s esophagus, report of a case. Stomach Intest (abstract in English). 2007;42:1543–9.
Zheng Y, Wang L, Zhang JP, Yang JY, Zhao ZM, Zhang XY. Expression of p53, c-erbB-2 and Ki67 in intestinal metaplasia and gastric carcinoma. World J Gastroenterol. 2010;16:339–44.
Moore OS, Foote FW. The relatively favorable prognosis of medullary carcinoma of the breast. Cancer. 1949;2:635–42.
Min KW, Holmquist S, Peiper SC, et al. Poorly differentiated adenocarcinoma with lymphoid stroma (lymphoepithelioma-like carcinomas) of the stomach. Report of three cases with Epstein–Barr virus genome demonstrated by the polymerase chain reaction. Am J Clin Pathol. 1991;96:219–27.
Horiuchi K, Mishima K, Ohsawa M, Aozasa K. Carcinoma of stomach and breast with lymphoid stroma: localisation of Epstein–Barr virus. J Clin Pathol. 1994;47:538–40.
Hasumi K, Sugano H, Sakamoto G, Masubuchi K, Kubo H. Circumscribed carcinoma of the uterine cervix, with marked lymphocytic infiltration. Cancer. 1977;39:2503–7.
Shimizu K, Takiyama W, Mandai K, Tanada M, Kawabuchi Y, Heike Y. Undifferentiated carcinoma with lymphoid infiltration of the esophagus: a case report. Jpn J Clin Oncol. 1999;29:494–7.
Martinet L, Garrido I, Filleron T, Le Guellec S, Bellard E, Fournie JJ, et al. Human solid tumors contain high endothelial venules: association with T- and B-lymphocyte infiltration and favorable prognosis in breast cancer. Cancer Res. 2011;71:5678–87.
Cho YJ, Chang MS, Park SH, Kim HS, Kim WH. In situ hybridization of Epstein–Barr virus in tumor cells and tumor-infiltrating lymphocytes of the gastrointestinal tract. Hum Pathol. 2001;32:297–301.
Jayasurya A, Bay BH, Yap WM, Tan NG. Lymphocytic infiltration in undifferentiated nasopharyngeal cancer. Arch Otolaryngol Head Neck Surg. 2000;126:1329–32.
Chang MS, Kim WH, Kim CW, Kim YI. Epstein-Barr virus in gastric carcinomas with lymphoid stroma. Histopathology. 2000;37:309–15.
Shibata D, Tokunaga M, Uemura Y, Sato E, Tanaka S, Weiss LM. Association of Epstein–Barr virus with undifferentiated gastric carcinomas with intense lymphoid stroma. Am J Pathol. 1991;139:469–74.
Nakamura S, Ueki T, Yao T, Ueyama T, Tsuneyoshi M. Epstein–Barr virus in gastric carcinoma with lymphoid stroma. Special reference to its detection by the polymerase chain reaction and in situ hybridization in 99 tumors, including a morphologic analysis. Cancer. 1994;73:2239–49.
Takubo K, Lambie NK. Barretts adenocarcinoma of the esophagus with lymphoid stroma. J Clin Gastroenterol. 2001;33:141–4.
Nakasono M, Hirokawa M, Suzuki M, Takizawa H, Okitsu H, Okamura S, et al. Lymphoepithelioma-like carcinoma of the esophagus: report of a case with non-progressive behavior. J Gastroenterol Hepatol. 2007;22:2344–7.
Kuwano H, Sumiyoshi K, Sonoda K, Kitamura K, Toh Y, Nakashima H, et al. Pathogenesis of esophageal squamous cell carcinoma with lymphoid stroma. Hepatogastroenterology. 2001;48:458–61.
Saito S, Hosoya Y, Morishima K, Ui T, Haruta H, Kurashina K, et al. A clinicopathological and immunohistochemical study of gastric cancer with squamous cell carcinoma components: a clinically aggressive tumor. J Dig Dis. 2012;13:407–13.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Katsube, O., Saito, S., Hosoya, Y. et al. Barrett’s esophageal carcinoma consisting of undifferentiated carcinoma and differentiated adenocarcinoma: report of case. Clin J Gastroenterol 6, 207–210 (2013). https://doi.org/10.1007/s12328-013-0377-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-013-0377-y