Abstract
Introduction
Factors affecting the angiographic outcomes of coronary de novo lesions treated with drug-coated balloons (DCBs) have not been well illustrated. The aim of the study is to establish a nomogram for predicting the risk of suboptimal diameter stenosis (DS) at angiographic follow-up.
Methods
A retrospective analysis was performed on a cohort of patients who underwent DCB intervention for coronary de novo lesions with angiographic follow-up data. Multivariable logistic regression analysis was applied to determine the independent predictors of DS ≥ 30% at follow-up, and then a nomogram model was established and validated.
Results
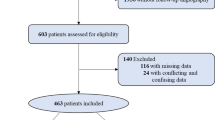
A total of 196 patients (313 lesions) were divided into the suboptimal (DS ≥ 30%) and optimal (DS < 30%) DS groups according to quantitative coronary angiography (QCA) measurements of the target lesions at follow-up. Seven independent factors including calcified lesions, true bifurcation lesions, immediate lumen gain rate (iLG%) < 20%, immediate diameter stenosis (iDS) ≥ 30%, DCB diameter/reference vessel diameter ratio (DCB/RVD) < 1.0, DCB length and mild dissection were identified. The area under the curve (AUC) (95% CI) of the receiver-operating characteristic (ROC) curve of the nomogram was 0.738 (0.683, 0.794). After the internal validation, the AUC (95% CI) was 0.740 (0.685, 0.795). The Hosmer-Lemeshow goodness of fit (GOF) test (χ2 = 6.57, P = 0.766) and the calibration curve suggested a good predictive consistency of the nomogram.
Conclusions
The well-calibrated nomogram could efficiently predict the suboptimal angiographic outcomes at follow-up. This model may be helpful to optimize lesion preparation to achieve optimal outcomes.




Similar content being viewed by others
References
Jeger RV, Eccleshall S, Wan Ahmad WA, Ge J, Poerner TC, Shin E-S, Alfonso F, Latib A, Ong PJ, Rissanen TT, et al. drug-coated balloons for coronary artery disease. JACC Cardiovasc Interv. 2020;13(12):1391–402.
Liu Y, Zhang YJ, Deng LX, Yin ZY, Hu T, Wang Q, Li Y, Li JY, Guo WY, Mou FJ, et al. 12-Month clinical results of drug-coated balloons for de novo coronary lesion in vessels exceeding 3.0 mm. Int J Cardiovasc Imaging. 2019;35(4):579–86.
Lu W, Zhu Y, Han Z, Sun G, Qin X, Wang Z, Liu G, Xi W, Wang X, Pan L, et al. Short-term outcomes from drug-coated balloon for coronary de novo lesions in large vessels. J Cardiol. 2019;73(2):151–5.
Yu X, Ji F, Xu F, Zhang W, Wang X, Lu D, Yang C, Wang F. Treatment of large de novo coronary lesions with paclitaxel-coated balloon only: results from a Chinese institute. Clin Res Cardiol. 2019;108(3):234–43.
Yu X, Wang X, Ji F, Zhang W, Yang C, Xu F, Wang F. A non-inferiority, randomized clinical trial comparing paclitaxel-coated balloon versus new-generation drug-eluting stents on angiographic outcomes for coronary de novo lesions. Cardiovasc Drugs Ther. 2022;36(4):655–64.
Chen Y, Wang J, Liu B, Ji F, Qiu C, Qiao S. Clinical use of drug-coated balloon: recommendations by the Chinese consensus group. Chin J Interv Cardiol. 2016;24(2):61–7.
Huber MSMJ, Madison J, Mooney MR. Use of a morphologic classification to predict clinical outcome after dissection from coronary angioplasty. Am J Cardiol. 1991;68(5):467–71.
Rogers JH, Lasala JM. Coronary artery dissection and perforation complicating percutaneous coronary intervention. J Invasive Cardiol. 2004;16(9):493–9.
Moons KG, Altman DG, Reitsma JB, Ioannidis JP, Macaskill P, Steyerberg EW, Vickers AJ, Ransohoff DF, Collins GS. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): explanation and elaboration. Ann Intern Med. 2015;162(1):W1-73.
Iasonos A, Schrag D, Raj GV, Panageas KS. How to build and interpret a nomogram for cancer prognosis. J Clin Oncol. 2008;26(8):1364–70.
Yamaji K, Kimura T, Morimoto T, Nakagawa Y, Inoue K, Kuramitsu S, Soga Y, Arita T, Shirai S, Ando K, et al. Very long-term (15 to 23 years) outcomes of successful balloon angioplasty compared with bare metal coronary stenting. J Am Heart Assoc. 2012;1(5): e004085.
Kleber FX, Rittger H, Bonaventura K, Zeymer U, Wohrle J, Jeger R, Levenson B, Mobius-Winkler S, Bruch L, Fischer D, et al. Drug-coated balloons for treatment of coronary artery disease: updated recommendations from a consensus group. Clin Res Cardiol. 2013;102(11):785–97.
Her AY, Shin ES, Bang LH, Nuruddin AA, Tang Q, Hsieh IC, Hsu JC, Kiam OT, Qiu C, Qian J, et al. Drug-coated balloon treatment in coronary artery disease: recommendations from an Asia-Pacific Consensus Group. Cardiol J. 2021;28(1):136–49.
Lee HS, Kang J, Park KW, Ki YJ, Chang M, Han JK, Yang HM, Kang HJ, Koo BK, Kim HS. Procedural optimization of drug-coated balloons in the treatment of coronary artery disease. Catheter Cardiovasc Interv. 2021;98(1):E43–52.
Scheller B. Editorial: Rotational atherectomy followed by drug-coated balloons in calcified coronary de novo lesions—an alternative to stent implantation? Cardiovasc Revasc Med. 2020;21(5):654–6.
Iwasaki Y, Koike J, Ko T, Funatsu A, Kobayashi T, Ikeda T, Nakamura S. Comparison of drug-eluting stents vs. drug-coated balloon after rotational atherectomy for severely calcified lesions of nonsmall vessels. Heart Vessels. 2021;36(2):189–99.
Fanelli F, Cannavale A, Gazzetti M, Lucatelli P, Wlderk A, Cirelli C, d’Adamo A, Salvatori FM. Calcium burden assessment and impact on drug-eluting balloons in peripheral arterial disease. Cardiovasc Interv Radiol. 2014;37(4):898–907.
Nagai T, Mizobuchi M, Funatsu A, Kobayashi T, Nakamura S. Acute and mid-term outcomes of drug-coated balloon following rotational atherectomy. Cardiovasc Interv Ther. 2020;35(3):242–9.
Onishi T, Onishi Y, Kobayashi I, Sato Y. Late lumen enlargement after drug-coated balloon angioplasty for de novo coronary artery disease. Cardiovasc Interv Ther. 2021;36(3):311–8.
Funatsu A, Kobayashi T, Mizobuchi M, Nakamura S. Clinical and angiographic outcomes of coronary dissection after paclitaxel-coated balloon angioplasty for small vessel coronary artery disease. Cardiovasc Interv Ther. 2019;34(4):317–24.
Creel CJ, Lovich MA, Edelman ER. Arterial paclitaxel distribution and deposition. Circ Res. 2000;86(8):879–84.
Yamamoto T, Sawada T, Uzu K, Takaya T, Kawai H, Yasaka Y. Possible mechanism of late lumen enlargement after treatment for de novo coronary lesions with drug-coated balloon. Int J Cardiol. 2020;321:30–7.
Hui L, Shin ES, Jun EJ, Bhak Y, Garg S, Kim TH, Sohn CB, Choi BJ, Kun L, Yuan SL, et al. Impact of dissection after drug-coated balloon treatment of de novo coronary lesions: angiographic and clinical outcomes. Yonsei Med J. 2020;61(12):1004–12.
Worthley S, Hendriks R, Worthley M, Whelan A, Walters DL, Whitbourn R, Meredith I. Paclitaxel-eluting balloon and everolimus-eluting stent for provisional stenting of coronary bifurcations: 12-month results of the multicenter BIOLUX-I study. Cardiovasc Revasc Med. 2015;16(7):413–7.
Kleber FX, Rittger H, Ludwig J, Schulz A, Mathey DG, Boxberger M, Degenhardt R, Scheller B, Strasser RH. Drug eluting balloons as stand alone procedure for coronary bifurcational lesions: results of the randomized multicenter PEPCAD-BIF trial. Clin Res Cardiol. 2016;105(7):613–21.
Stella PR, Belkacemi A, Dubois C, Nathoe H, Dens J, Naber C, Adriaenssens T, van Belle E, Doevendans P, Agostoni P. A multicenter randomized comparison of drug-eluting balloon plus bare-metal stent versus bare-metal stent versus drug-eluting stent in bifurcation lesions treated with a single-stenting technique: six-month angiographic and 12-month clinical results of the drug-eluting balloon in bifurcations trial. Catheter Cardiovasc Interv. 2012;80(7):1138–46.
Schulz A, Hauschild T, Kleber FX. Treatment of coronary de novo bifurcation lesions with DCB only strategy. Clin Res Cardiol. 2014;103(6):451–6.
Zuin M, Rigatelli G. Treatment of de novo coronary artery bifurcation lesions with drug coated balloons: a reappraisal according to the available scientific data. Cardiovasc Revasc Med. 2018;19(1 Pt A):57–64.
Cortese B, Piraino D, Buccheri D, Alfonso F. Treatment of bifurcation lesions with drug-coated balloons: a review of currently available scientific data. Int J Cardiol. 2016;220:589–94.
Acknowledgements
We thank the participants of the study.
Funding
This study was funded by the Research Fund for clinical and translational medicine, Chinese Academy of Medical Sciences (grant number 2020-I2M-C&T-B-092), the Scientific Research Project of Central Health Committee (grant number 2020YB08) and the National High Level Hospital Clinical Research Funding (grant number BJ-2022–124). The journal’s Rapid Service Fee was funded by the authors.
Medical Writing, Editorial and Other Assistance
Medical writing/editorial support was provided by Cassie J., the Senior Editor of American Journal Experts, and funded by the authors.
Author Contributions
Conception and design: XY, FJ; Acquisition of data: XY, WZ, XW, CY, PL; Analysis and interpretation of the data: XY, YW, NJ, YZ; Drafting and critical revision of the article for important intellectual content: FJ, FX, XY.
Disclosures
Xue Yu, Yijia Wang, Wenduo Zhang, Xinyue Wang, Na Jia, Yanan Zhang, Chenguang Yang, Peng Li, Feng Xu and Fusui Ji have nothing to disclose.
Compliance with Ethics Guidelines
This study was conducted in accordance with the ethical principles of the Declaration of Helsinki, Good Clinical Practice, the International Committee on Harmonisation guidelines and applicable laws and regulations. The protocol was approved by the ethics committee of Beijing Hospital (ID: 2016BJYYEC-067–01) and all subjects provided written informed consent.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, X., Wang, Y., Zhang, W. et al. Establishment of a Nomogram for Predicting the Suboptimal Angiographic Outcomes of Coronary De Novo Lesions Treated with Drug-Coated Balloons. Adv Ther 40, 975–989 (2023). https://doi.org/10.1007/s12325-022-02400-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-022-02400-1