Abstract
Objectives
This study set out to examine the efficacy and tolerability of two innovative implant forms of leuprorelin acetate in men with advanced hormone-dependent prostate cancer in everyday clinical practice.
Methods
Data were collected from 818 patients (from 273 centers across Germany) who were pretreated with slow-release luteinizing hormone-releasing hormone (LHRH) agonist formulations and who were about to be switched to the leuprorelin implants. Patients received three injections of 1- or 3-month leuprorelin implant and physicians were asked to complete a case report form specific to each of the three clinic visits. Documented parameters included laboratory measurements, such as testosterone and prostate-specific antigen (PSA) levels, adverse events, and patient- and physician-rated assessments of the therapy.
Results
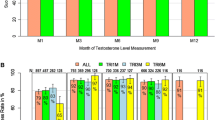
Compared with baseline, a significant decrease in both testosterone and PSA levels were measured after the first and second injections of leuprorelin implant. These results were confirmed for both the 1-month and 3-month implants in separate analyses. Switching, without treatment interruption, from Trenantone® (Takeda Pharma GmBH, Aachen, Germany) to the leuprorelin implant resulted in a significant decrease in the mean serum testosterone concentrations (P < 0.05) and a nonsignificant increase in the proportion of patients reaching castrate testosterone levels, while the number of patients with PSA values ≤4 ng/mL significantly increased (P = 0.045). Similar results were obtained for patients previously treated with goserelin who switched to leuprorelin implant. For 94% of patients, treating physicians rated the efficacy of leuprorelin implant as “very good” or “good.” Treatment with leuprorelin implant was well tolerated, with only 61 adverse events reported in 42 (5.1%) patients. Patients and physicians rated the tolerability of leuprorelin implant as “very good” or “good” in 95% and 91% of cases, respectively.
Conclusions
These results confirm the efficacy, tolerability, and ease of use of the leuprorelin implants among a large population of men with advanced, hormone-dependent prostate cancer treated in a clinical practice setting.
Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global Cancer Statistics. CA Cancer J Clin. 2011;61:69–90.
Ferlay J, Shin H-R, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893–2917.
National Institute for Health and Clinical Excellence. Docetaxel for the treatment of hormone-refractory metastatic prostate cancer. Available at: http://www.nice.org.uk/TA101. Accessed Oct 14 2012.
Heidenreich A, Bastian PJ, Bellmunt, J et al. Guidelines on prostate cancer. Update February 2012. Available at: http://www.uroweb.org/gls/pdf/08%20Prostate%20Cancer_LR%20March%2013th%202012.pdf. Accessed Oct 14 2012.
McLeod DG. Hormonal therapy: historical perspective to future directions. Urology. 2003;61(Suppl. 1):3–7.
Limonta P, Montagnani MM, Moretti RM. LHRH analogues as anticancer agents: pituitary and extrapituitary sites of action. Expert Opin Investig Drugs. 2001;10:709–720.
Geiges G, Schapperer E, Thyroff-Friesinger U, Engert ZV, Gravel P. Clinical development of a new pharmaceutical form of leuprorelin acetate. Ther Adv Urol. 2013;5:3–10.
Thyroff-Friesinger U, Geiges G, Vendel Engert Z. Clinical development of a new pharmaceutical form of leuprorelin acetate for the treatment of advanced prostate cancer: studies of a 1-month implant. J Geriatr Oncol. 2012; 3(Suppl. 1):S86–S87.
Thyroff-Friesinger U, Geiges G, Vendel Engert Z. Clinical development of a new pharmaceutical form of leuprorelin acetate for the treatment of advanced prostate cancer: studies of a 3-month implant. J Geriatr Oncol. 2012;3(Suppl. 1):S86.
Thyroff-Friesinger U, Geiges G, Vendel Engert Z. Clinical development of a new pharmaceutical form of leuprorelin acetate for the treatment of advanced prostate cancer: pharmacokinetics of a 3-month implant. J Geriatr Oncol. 2012; 3(Suppl. 1):S85.
Leuprorelin-Sandoz 5 mg. Summary of Product Characteristics. Holzkirchen, Germany; Sandoz Biopharmaceuticals GmbH.
Leuprorelin-Sandoz 3.6 mg. Summary of Product Characteristics. Holzkirchen, Germany; Sandoz Biopharmaceuticals GmbH.
Zlotta A, Debruyne FMJ. Expert opinion on optimal testosterone control in prostate cancer. Eur Urol Suppl. 2005;4:37–41.
Oefelein MG, Feng A, Scolieri MJ, Ricchiuti D, Resnick MI. Reassessment of the definition of castrate levels of testosterone: implications for clinical decision making. Urology. 2000;56: 1021–1024.
Lucrin PDS 6 month 30 mg, Public Assessment Report of the Medicines Evaluation Board, The Netherlands, 19 April 2009. Available at: http://db.cbg-meb.nl/Pars/h100696.pdf. Accessed on: February 1, 2013.
Tunn UW. A 6-month depot formulation of leuprolide acetate is safe and effective in daily clinical practice: a non-interventional prospective study in 1273 patients. BMC Urology. 2011;11:15.
Thyroff-Friesinger U, Geiges G, Vendel Engert Z. A new pharmaceutical form of leuprorelin acetate for the treatment of advanced prostate cancer: clinical experience from a non-interventional study. J Geriatr Oncol. 2012;3(Suppl. 1):S87.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gravel, P., Samland, D., Löffler, M. et al. Two Innovative Pharmaceutical Forms of Leuprorelin: Results from 818 Patients with Advanced Prostate Cancer. Adv Therapy 30, 271–285 (2013). https://doi.org/10.1007/s12325-013-0010-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-013-0010-y