Abstract
Introduction
Despite breakthrough pain (BTP) being one of the most severe forms of pain, there are no definitive data on its prevalence.
Methods
The authors performed a retrospective survey of the prevalence of BTP in consecutive patients in four Italian pain clinics, subsequent to application of an Italian law mandating detailed clinical records on pain characteristics, treatment, and results. Mean pain intensity was assessed with a numerical rating scale from 0 to 10.
Results
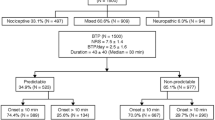
The authors analyzed records of 1,401 patients (58% women, 33.1% patients with cancer). Transient episodes of severe pain or BTP were referred by 790 patients (56.4%), including 58.2% of the men (342 of 588) and 55.1% of the women (448 of 813). Among the 464 patients with cancer, 70.3% reported daily exacerbation of pain. The mean BTP intensity was 8.31 ± 1.58 and 31.1% of patients reported experiencing three episodes per day.
Conclusion
Despite some limitations of the study, the authors show that transient episodes of severe pain or BTP are significantly present both in cancer and other diseases, and that many patients are not yet receiving appropriate opioid therapy. The authors need validated tools at international level for the diagnosis and treatment of BTP in patients with cancer and for transitory and patients with severe non-cancer pain. A survey at national level is needed to estimate the prevalence of BTP in different settings, to plan specific medical education.
Similar content being viewed by others
References
Portenoy RK, Hagen NA. Breakthrough pain: definition and management. Oncology (Williston Park). 1989;3(Suppl.): 25–29.
Portenoy RK, Hagen NA. Breakthrough pain: definition, prevalence and characteristics. Pain. 1990;41:273–281.
Portenoy RK, Payne D, Jacobsen P. Breakthrough pain: characteristics and impact in patients with cancer pain. Pain. 1999;81:129–134.
Haugen DF, Hjermstad MJ, Hagen N, Caraceni A, Kaasa S; European Palliative Care Research Collaborative (EPCRC). Assessment and classification of cancer breakthrough pain: a systematic literature review. Pain. 2010;149:476–482.
Svendsen KB, Andersen S, Arnason S, et al. Breakthrough pain in malignant and non-malignant diseases: a review of prevalence, characteristics and mechanisms. Eur J Pain. 2005;9:195–206.
Mercadante S, Radbruch L, Caraceni A, et al.; Steering Committee of the European Association for Palliative Care (EAPC) Research Network. Episodic (breakthrough) pain: consensus conference of an expert working group of the European Association for Palliative Care. Cancer. 2002;94:832–839.
Zeppetella G. Impact and management of breakthrough pain in cancer. Curr Opin Support Palliat Care. 2009;3:1–6.
Caraceni A, Martini C, Zecca E, et al. Breakthrough pain characteristics and syndromes in patients with cancer pain. An international survey. Palliat Med. 2004;18:177–183.
Mercadante S, Costanzo BV, Fusco F, et al. Breakthrough pain in advanced cancer patients followed at home: a longitudinal study. J Pain Symptom Manage. 2009;38:554–560.
Mercadante S, Zagonel V, Breda E, et al. Breakthrough pain in oncology: a longitudinal study. J Pain Symptom Manage. 2010;40:183–190.
Greco MT, Corli O, Montanari M, et al.; Writing Protocol Committee; Cancer Pain Outcome Research Study Group (CPOR SG) Investigators. Epidemiology and pattern of care of breakthrough cancer pain in a longitudinal sample of cancer patients: results from the Cancer Pain Outcome Research Study Group. Clin J Pain. 2011;27:9–18.
Mercadante S, Villari P, Casuccio A. An Italian survey on the attitudes in treating breakthrough cancer pain in hospice. Support Care Cancer. 2011;19:979–983.
Zeppetella G, O’Doherty CA, Collins S. Prevalence and characteristics of breakthrough pain in patients with non-malignant terminal disease admitted to a hospice. Palliat Med. 2001;15:243–246.
Portenoy RK, Bruns D, Shoemaker B, Shoemaker SA. Breakthrough pain in community-dwelling patients with cancer pain and non cancer pain, part 1: prevalence and characteristics. J Opioid Manag. 2010;6:97–108.
Haanpää M, Attal N, Backonja M, et al. NeuPSIG guidelines on neuropathic pain assessment. Pain. 2011;152:14–27.
Andersson DA, Gentry C, Alenmyr L, et al. TRPA1 mediates spinal antinociception induced by acetaminophen and the cannabinoid Δ(9)-tetrahydrocannabiorcol. Nat Commun. 2011;22,:551.
Caraceni A, Hanks G, Kaasa S, et al.; European Palliative Care Research Collaborative (EPCRC); European Association for Palliative Care (EAPC). Use of opioid analgesics in the treatment of cancer pain: evidence-based recommendations from the EAPC. Lancet Oncol. 2012;13:e58–e68.
Mercadante S. Pharmacotherapy for breakthrough cancer pain. Drugs. 2012;72:181–190.
Fisher A, Watling M, Smith A, Knight A. Pharmacokinetic comparisons of three nasal fentanyl formulations; pectin, chitosan and chitosan-poloxamer 188. Int J Clin Pharmacol Ther. 2010;48:138–145.
Christrup LL, Foster D, Popper LD, Troen T, Upton R. Pharmacokinetics, efficacy, and tolerability of fentanyl following intranasal versus intravenous administration in adults undergoing third-molar extraction: a randomized, double-blind, double-dummy, two-way, crossover study. Clin Ther. 2008;30:469–481.
Lennernäs B, Hedner T, Holmberg M, et al. Pharmacokinetics and tolerability of different doses of fentanyl following sublingual administration of a rapidly dissolving tablet to cancer patients: a new approach to treatment of incident pain. Br J Clin Pharmacol. 2005;59:249–253.
Darwish M, Kirby M, Robertson P Jr, Hellriegel E, Jiang JG. Comparison of equivalent doses of fentanyl buccal tablets and arteriovenous differences in fentanyl pharmacokinetics. Clin Pharmacokinet. 2006;45:843–850.
Watts P, Smith A. PecSys: in situ gelling system for optimised nasal drug delivery. Expert Opin Drug Deliv. 2009;6:543–552.
Davies A, Sitte T, Elsner F, et al. Consistency of efficacy, patient acceptability, and nasal tolerability of fentanyl pectin nasal spray compared with immediate-release morphine sulfate in breakthrough cancer pain. J Pain Symptom Manage. 2011;41:358–366.
Portenoy RK, Burton AW, Gabrail N, Taylor D; Fentanyl Pectin Nasal Spray 043 Study Group. A multicenter, placebo-controlled, double-blind, multiple-crossover study of Fentanyl Pectin Nasal Spray (FPNS) in the treatment of breakthrough cancer pain. Pain. 2010;151:617–624.
Radbruch L, Torres LM, Ellershaw JE, et al. Long-term tolerability, efficacy and acceptability of fentanyl pectin nasal spray for breakthrough cancer pain. Support Care Cancer. 2012;20:565–573.
Taylor D, Galan V, Weinstein SM, et al.; Fentanyl Pectin Nasal Spray 043 Study Group. Fentanyl pectin nasal spray in breakthrough cancer pain. J Support Oncol. 2010;8:184–190.
Mercadante S. Managing breakthrough pain. Curr Pain Headache Rep. 2011;15:244–249.
Mercadante S, Gatti A, Porzio G, et al. Dosing fentanyl buccal tablet for breakthrough cancer pain: dose titration versus proportional doses. Curr Med Res Opin. 2012. Epub ahead of print.
Sabato AF. Idiopathic breakthrough pain. A new hypothesis. Clin Drug Investig. 2010;30:27–29.
Author information
Authors and Affiliations
Corresponding author
Additional information
To view enhanced content go to www.advancesintherapy.com
Rights and permissions
About this article
Cite this article
Gatti, A., Mediati, R.D., Reale, C. et al. Breakthrough Pain in Patients Referred to Pain Clinics: The Italian Pain Network Retrospective Study. Adv Therapy 29, 464–472 (2012). https://doi.org/10.1007/s12325-012-0022-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-012-0022-z