Abstract
Introduction
Myrtol standardized is a phytomedicine obtained by distillation, consisting of many constituents. In vitro and in vivo, the major monterpenes, d-limonene, 1,8-cineole, and alpha-pinene, are used as biological marker substances. Myrtol standardized has secretolytic, secretomotor, and mucolytic effects in addition to anti-inflammatory and antioxidative actions. The aim of the study was to investigate the effects of different concentrations of Myrtol standardized on in vivo mucociliary clearance in mice and the ciliary beat frequency (CBF) in rat tracheal rings.
Methods
Data regarding the effects of 1,8-cineole and N-acetylcysteine (NAC) were compared. Salbutamol was used as a positive control. CBF was measured using rat tracheal explants and a high-speed video camera linked to a microscope with specific software equipment. Mucociliary clearance was determined using the microdialysis technique, which measured the acceleration of a fluorescent sample in the trachea in vivo.
Results
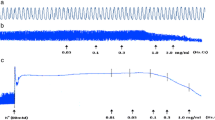
Myrtol standardized accelerated both CBF and mucociliary transport in a concentration-dependent manner. Significant effects were seen at a concentration of 0.01% Myrtol regarding CBF (P < 0.01) and 17.1 mg/kg body weight regarding mucociliary clearance experiments (P < 0.05) according to doses relevant to humans. The 1,8-cineole dosage relative to humans only accelerated the mucociliary clearance in vivo without having an effect on the CBF. Extremely high doses of Myrtol were not able to additionally increase the CBF effect in comparison to salbutamol. Compared to NAC, also used in a dosage relative to humans, Myrtol standardized showed a tendency to be more effective.
Conclusion
In summary, the present data suggest that Myrtol standardized is a pharmacologically important drug which, when used at a dose relative to humans, shows positive effects on both measured parameters, CBF and mucociliary clearance, in vivo.
Similar content being viewed by others
References
Bateman JRM, Pavia D, Sheahan NF, Agnew JE, Clarke SW. Impaired tracheobronchial clearance in patients with mild stable asthma. Thorax. 1983;38:463–7.
Ericsson CH, Juhasz J, Mossberg B, Philipson K, Svartengren M, Camner P. Influence of ambroxol on tracheobronchial clearance in simple chronic bronchitis. Eur J Respir Dis. 1987;70:163–70.
Eliezer N, Sade J, Silberberg A. The role of mucus in transport by cilia. Am Rev Respir Dis. 1970;:48–52.
Sleigh MA, Blake JR, Liron N. The propusion of mucus by cilia. Am Rev Respir Dis. 1988;137:726–41.
Devalia JL, Sapsford RJ, Rusznak C, Toumbis MJ, Davies RJ. The effects of salmeterol and salbutamol on ciliary beat frequency of cultured human bronchial epithelial cells, in vitro. Pulmonary pharmacology. 1992;5:257–63.
Lenders H, Waitzinger J, Zimmerman TH, et al. Suitability of various methods as pharmacodynamic models for the investigation of the efficacy of mucolytic agents on the maxillary sinus, Naunyn-Schmiederberg’s Arch Pharmacol. 1996;353(suppl.):R151.
Lenders H. [Pharmakodynamische Nachweismethoden zur Wirkung von ätherischen Ölen am oberen Respirationstrakt. In Mees K, ed. Die unspezifische Rhino-Sinusitis. 1st edition. Berlin: Springer Verlag. 1996;40–51.
Deitmer T. Physiology and pathology of the mucociliary system, In: Pfaltz CR, ed. Advances in Oto-Rhino-Laryngology. Volume 45. Basel: Karger; 1989:4–18.
Jones N. The nose and paranasal sinuses physiology and anatomy. Adv Drug Deliv Rev. 2001;51:5–19.
Engler H, Szelenyi I. Tracheal phenol red secretion, a new method for screening mucosecretolytic compounds. J Pharmacol Methods. 1984;11:151–7.
App EM. Sekretanalyse und Clearance. Atemw-Lungenkrkh. 1994;20:363–71.
King M. Magnetic Microrheometer. In: Braga PC, Allegra L, eds. Methods in Bronchial Mucology. New York: Raven Press. 1988;73–83.
App EM. Stellenwert der Mukusclearance für das Bronchialsystem — Pathophysiologie und therapeutische Ansätze, In: Meister R. Entzündliche Erkrankungen des Bronchialsystems. 1st edition. Berlin: Springer Verlag; 2000:27–53.
Graßmann J, Hippeli S, Dornisch K, Rohnert U, Beuscher N, Elstner EF. Antioxidant properties of essential oils. Arzneim-Forsch/Drug Res. 2000;50:135–9.
Hippeli S, Graßmann J, Dornisch K, et al. Freie Radikale in Pathogenese und Therapie von entzündlichen Erkrankungen des Bronchialsystems, In: Meister R. Entzündliche Erkrankungen des Bronchialsystems. 1st edition. Berlin: Springer Verlag; 2000:1–25.
Brigham KL. Oxidant stress and adult respiratory distress syndrome. Eur Respir J. 1990;3:482s–4s.
Behrbohm H, Kaschke O, Sydow K. The influence of a secretolytic drug on mucociliary clearance of the maxillary sinus. J Rol. 1997;4:29–33.
Mercke U, Hakansson CH, Toremalm NG. The influence of temperature on mucociliary activity. Temperature range 20 degrees C-40 degrees C. Acta Otolaryngol. 1974;78:444–50.
Grubb BR, Jones JH, Boucher RC. Mucociliary transport determined by in vivo microdialysis in the airways of normal and CF mice. Am J Physiol Lung Cell Mol Physiol. 2004;286:L588–95.
Wienkötter N, Begrow F, Kinzinger U, Schierstedt D, Verspohl EJ. The effects of thyme extract on β2-receptors and mucociliary clearance. Planta Med. 2007;73:629–35.
Han D, Wang N, Zhang L. The effect of myrtol standardized on human nasal ciliary beat frequency and mucociliary transport time. Am J Rhinol Allergy. 2009;23:610–4.
Shi L, Han D, Zhang L. Effect of ATP on human nasal ciliary beat frequency. Shoudu Yike Daxue Xuebao. 2009;30:57–61.
Alberty J, Stoll W. The effect of antiallergic intransal formulations on ciliary beat frequency of human nasal epithelium in vitro. Allergy. 1998;53:986–7.
Agius AM, Smallman LA, Pahor AL. Age, smoking and nasal ciliary beat frequency, Clin Otolaryngology Allied Sci. 1998;23:227–30.
Jorissen M, Willems T, Van der Schueren B. Nasal ciliary beat frequency is age independent. Laryngoscope. 1998;108:1042–7.
Hafner B, Davris S, Riechelmann H, Mann WJ, Amedee RG. Endonasal sinus surgery improves mucociliary transport in severe chronic sinusitis. Am J Rhinol. 1997;11:271–274.
Zimmermann T, Seiberling M, Thomann P.. Arzneimittelforschung. 1995;45:1198–201. In German.
App EM. Analytischer Abschlussbericht, Bestimmung von Cineol, Limonen und alpha-Pinen in Sputumproben, Myrtol standardisiert Dokumentation. 1997.
Kwok R, Leung R, Tsang KW, et al. The effects of Gelomyrtol forte on human ciliary beat frequncy in vitro. Respirology. 2004;9(suppl.):A16.
Langeneckert A. Untersuchungen zur Pharmakokinetik und relativen Bioverfügbarkeit von α-Pinen, 1,8-cineol und Menthol nach dermaler, inhalativer und peroraler Applikation ätherischcher Öle. Shaker Verlag GmbH. 1999.
Jäger W, Nasel B, Nasel C, et al. Pharmacokinetic studies of the fragrance compound 1,8-cineole in humans during inhalation. Chem Senses. 1996;21:477–80.
Author information
Authors and Affiliations
Corresponding author
Additional information
To view enhanced content go to www.advancesintherapy.com
Rights and permissions
About this article
Cite this article
Begrow, F., Böckenholt, C., Ehmen, M. et al. Effect of Myrtol Standardized and Other Substances on the Respiratory Tract: Ciliary Beat Frequency and Mucociliary Clearance as Parameters. Adv Therapy 29, 350–358 (2012). https://doi.org/10.1007/s12325-012-0014-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-012-0014-z