Abstract
Introduction
Patients with glaucoma or ocular hypertension who do not achieve target intraocular pressure (IOP) using one hypotensive agent are often transitioned to combination therapy. Travoprost 0.004%/timolol 0.5% fixed combination (TTFC) has shown efficacy in patients whose IOP is not controlled with other therapies. The goal of this study was to assess the efficacy and safety of transitioning to TTFC in patients whose IOP was uncontrolled on bimatoprost 0.03%/timolol 0.5%, administered concomitantly or as a fixed combination.
Methods
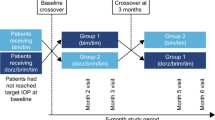
This was a prospective, open-label, multicenter study of patients with open-angle glaucoma or ocular hypertension who transitioned to TTFC from fixed or unfixed bimatoprost/timolol. Patients self-administered TTFC once daily for 8 weeks, and efficacy and safety were assessed at baseline, Week 4, and Week 8. A symptom survey was administered at baseline and Week 8. Both patients and investigators reported their medication preference at Week 8.
Results
A total of 105 patients were enrolled in the study. Mean IOP decreased by 16.5% from baseline after 8 weeks of TTFC therapy in the total population, 15.0% in patients transitioning from fixed-combination therapy, and 20.8% in patients transitioning from unfixed therapy (P<0.001 for all groups). The percentage of patients reaching target IOP (≤18 mmHg) after treatment with TTFC was 69.2% (P<0.001). Patients judged stinging/burning to be less severe with TTFC than with prior therapy (P=0.029); all other symptom frequencies and severities were similar for both treatments. Patients preferred TTFC over bimatoprost/timolol (fixed and unfixed) at a ratio of more than 4:1 (81.4% vs. 18.6%; P<0.001), and investigators reported a nearly five-fold preference for TTFC (83.3% vs. 16.7%; P<0.001). No unexpected safety concerns with TTFC were observed.
Conclusion
Travoprost 0.004%/timolol 0.5% fixed combination produced a significant reduction in IOP, with favorable safety and tolerability profiles. Both patients and investigators strongly preferred TTFC to prior bimatoprost 0.03%/timolol 0.5% therapy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
European Glaucoma Society. Terminology and Guidelines for Glaucoma. 3rd edition. Savona: Editrice Dogma S.r.l.; 2008.
DuoTrav eye drops solution: summary of patient characteristics. Hemel Hempstead: Alcon Laboratories (U.K.) Limited; 2011.
Gross RL, Sullivan EK, Wells DT, Mallick S, Landry TA, Bergamini MVW. Pooled results of two randomized clinical trials comparing the efficacy and safety of travoprost 0.004%/timolol 0.5% in fixed combination versus concomitant travoprost 0.004% and timolol 0.5%. Clin Ophthalmol. 2007;1:317–322.
Centofanti M, Oddone F, Gandolfi S, et al. Comparison of travoprost and bimatoprost plus timolol fixed combinations in open-angle glaucoma patients previously treated with latanoprost plus timolol fixed combination. Am J Ophthalmol. 2010;150:575–580.
Konstas AGP, Mikropoulos AD, Embeslidis TA, et al. 24-h intraocular pressure control with evening-dosed travoprost/timolol, compared with latanoprost/timolol, fixed combinations in exfoliative glaucoma. Eye (Lond). 2010;24:1606–1613.
Teus MA, Miglior S, Laganovska G, et al. C-05-25 Study Group. Efficacy and safety of travoprost/timolol vs. dorzolamide/timolol in patients with open-angle glaucoma or ocular hypertension. Clin Ophthalmol. 2009;3:629–636.
Arend K-O, Raber T. Observational study results in glaucoma patients undergoing a regimen replacement to fixed combination travoprost 0.004%/timolol 0.5% in Germany. J Ocul Pharmacol Ther. 2008;24:414–420.
Pfeiffer N, Scherzer M-L, Maier H, et al. DuoTrav Med Study Group. Safety and efficacy of changing to the travoprost/timolol maleate fixed combination (DuoTrav) from prior mono-or adjunctive therapy. Clin Ophthalmol. 2010;4:459–466.
Rossi GCM, Pasinetti GM, Bracchino M, et al. Switching from concomitant latanoprost 0.005% and timolol 0.5% to a fixed combination of travoprost 0.004%/timolol 0.5% in patients with primary open-angle glaucoma and ocular hypertension: a 6-month, multicenter, cohort study. Expert Opin Pharmacother. 2009;10:1705–1711.
The AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol. 2000;130:429–440.
Chauhan BC, Mikelberg FS, Balaszi AG, LeBlanc RP, Lesk MR, Trope GE. Canadian Glaucoma Study Group. Canadian Glaucoma Study: 2. Risk factors for the progression of open-angle glaucoma. Arch Ophthalmol. 2008;126:1030–1036.
Leske MC, Heijl A, Hussein M, Bengtsson B, Hyman L, Komaroff E. Early Manifest Glaucoma Trial Group. Factors for glaucoma progression and the effect of treatment: the early manifest glaucoma trial. Arch Ophthalmol. 2003;121:48–56.
Holló G. The side effects of the prostaglandin analogues. Expert Opin Drug Saf. 2007;6:45–52.
Bland JM, Altman DG. Regression towards the mean. BMJ. 1994;308:1499.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Scherzer, ML., Liehneova, I., Muñoz Negrete, F.J. et al. Travoprost 0.004%/timolol 0.5% fixed combination in patients transitioning from fixed or unfixed bimatoprost 0.03%/timolol 0.5%. Adv Therapy 28, 661–670 (2011). https://doi.org/10.1007/s12325-011-0043-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-011-0043-z