Abstract
Introduction
Large postmarketing surveillance (PMS) studies have demonstrated the efficacy and tolerability of frovatriptan for treatment of acute migraine in patients attending general practitioners (GPs). The aim of the ALADIN (Allegro® Anwendung durch interessierte Neurologen [Allegro® application by interested neurologists]) PMS study was to evaluate frovatriptan in patients attending neurologists or pain therapists.
Methods
Patients fulfilling International Headache Society criteria for migraine, with or without aura, were enrolled. Patients completed an attack diary, including details of the attack, time to onset of action of frovatriptan, and recurrence of headache. Physicians completed a case report form detailing prior and actual migraine treatment. Frovatriptan 2.5 mg was administered for up to three consecutive attacks.
Results
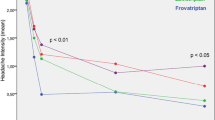
In total, 2160 patients were enrolled and data were obtained for 5831 attacks. Patients attending neurologists had more frequent attacks and longer history of migraine compared with those attending GPs. Median time to frovatriptan onset of action was 40 min and time to freedom from headache 70 min. An average of 1.2 frovatriptan tablets was required per attack, and mean additional analgesic use was 0.13. Recurrent headache occurred in 13.6%–15.5% of patients. Physicians as well as patients judged frovatriptan onset and duration of action as at least “good” in approximately 80% of attacks. A similar number judged the efficacy of frovatriptan against headache as “better” than previous treatment. The study drug was generally well tolerated. The frequency of adverse events was 0.6%.
Conclusion
Frovatriptan, with fast onset of action and low rate of headache recurrence, was efficacious and well accepted by migraineurs attending neurologists or pain therapists. Approximately 80% of patients wished to continue migraine treatment with frovatriptan.
Similar content being viewed by others
References
Rasmussen, BK, Jensen R, Schroll M, Olesen J. Epidemiology of headache in a general population — a prevalence study. J Clin Epidemiol. 1991;44:1147–1157.
Stovner L, Hagen K, Jensen R, et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia. 2007;27:193–210.
Pfaffenrath, V, Fendrich K, Vennemann M, et al. Regional variations in the prevalence of migraine and tension-type headache applying the new IHS criteria: the German DMKG Headache Study. Cephalalgia. 2009;29:48–57.
Brandes JL. Global trends in migraine care: results from the MAZE survey. CNS Drugs. 2002;16(suppl. 1):13–18.
Silberstein SD. Practice parameter: evidence-based guidelines for migraine headache (an evidencebased review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2000;55:754–762.
Evers S, Afra J, Frese A, et al. EFNS guideline on the drug treatment of migraine — report of an EFNS task force. Eur J Neurol. 2009;16:968–981.
Evers S, May A. Acute therapy and prophylaxis of migraine — guideline of the German Migraine and Headache Society and the German Society of Neurology. Nervenheilkunde. 2008;27:933–949.
Lipton RB, Stewart WF, Cady R, et al. 2000 Wolfe Award. Sumatriptan for the range of headaches in migraine sufferers: results of the Spectrum Study. Headache. 2000;40:783–791.
Ryan R, Geraud G, Goldstein J, Cady R, Keywood C. Clinical efficacy of frovatriptan: placebo-controlled studies. Headache. 2002;42(suppl. 2):S84–S92.
Miner JR, Smith SW, Moore J, Biros M. Sumatriptan for the treatment of undifferentiated primary headaches in the ED. Am J Emerg Med. 2007;25:60–64.
Buzzi MG, Moskowitz MA. The antimigraine drug, sumatriptan (GR43175), selectively blocks neurogenic plasma extravasation from blood vessels in dura mater. Br J Pharmacol. 1990;99:202–206.
Goadsby PJ, Akerman S, Storer RJ. Evidence for postjunctional serotonin (5-HT1) receptors in the trigeminocervical complex. Ann Neurol. 2001;50:804–807.
Geraud G, Keywood C, Senard JM. Migraine headache recurrence: relationship to clinical, pharmacological, and pharmacokinetic properties of triptans. Headache. 2003;43:376–388.
Pfaffenrath V, Spierings EH. Acute treatment of migraine attacks — frovatriptan 2.5 mg as an effective and economic alternative. Nervenheilkunde. 2004;23:545–548.
Diener HC, Pfaffenrath V. Efficacy and tolerability of frovatriptan in general practice — results of two PMS-studies with 16,737 migraineurs. Nervenheilkunde. 2007;26:180–185.
Goebel H. Erfolgreich gegen Kopfschmerzen und äne (Successful against headache and migraine). Heidelberg: Springer Verlag; 2004.
MIGARD 2.5 mg (frovatriptan) summary of product characteristics. A. Menarini Pharma. Electronic Medicines Compendium. July 2008. Available at: http://emc.medicines.org.uk/medicine/15216/spc/migard. Accessed October 23, 2008.
Lipton RB, Stewart WF, Stone AM, et al. Stratified care vs step care strategies for migraine: the Disability in Strategies of Care (DISC) Study: a randomized trial. JAMA. 2000;284:2599–2605.
Cady R, Elkind A, Goldstein J, Keywood C. Randomized, placebo-controlled comparison of early use of frovatriptan in a migraine attack versus dosing after the headache has become moderate or severe. Curr Med Res Opin. 2004;20:1465–1472.
Cady R, Martin V, Mauskop A, et al. Efficacy of rizatriptan 10 mg administered early in a migraine attack. Headache. 2006;46:914–924.
Dowson AJ, Mathew NT, Pascual J. Review of clinical trials using early acute intervention with oral triptans for migraine management. Int J Clin Pract. 2006;60:698–706.
Zwart JA, Dyb G, Hagen K, Svebak S, Holmen J. Analgesic use: a predictor of chronic pain and medication overuse headache: the Head-HUNT Study. Neurology. 2003;61:160–164.
Meskunas CA, Tepper SJ, Rapoport AM, Sheftell FD, Bigal ME. Medications associated with probable medication overuse headache reported in a tertiary care headache center over a 15-year period. Headache. 2006;46:766–772.
Katsarava Z, Zwarg T. Medication overuse as a risk factor for the chronification of headache. Nervenheilkunde. 2010. In press.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wallasch, TM. Frovatriptan in the practice of office-based neurologists/pain therapists: Results of postmarketing surveillance study ALADIN. Adv Therapy 27, 56–62 (2010). https://doi.org/10.1007/s12325-010-0001-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-010-0001-1