Abstract
Introduction
The action of cortistatin (CST), a novel cyclic neuropeptide, as an anti-inflammatory factor has been studied, but few investigations have explored the immunomodulatory role of CST in transplantation. In the present study, we examined whether CST affects the alloimmune response in a mouse model of skin transplantation and the effects of CST on T lymphocytes.
Methods
BALB/c (H-2Kd) recipient mice (n=70) were divided into seven groups (n=10 per group) and given an intraperitoneal injection of CST or a somatostatin analog, SMS 201-995 (octreotide), on the day of skin transplantation from C57BL/6 (B6) (H-2Kb) donors. Injections were continued for 7 consecutive days. Groups 1-3 received CST at doses of 0.02, 0.2, or 2 mg/kg, respectively. Groups 4–6 received SMS 201–995 at the same doses. Group 7 was a control group and received injections of phosphate buffered saline. Survival of the allografts was recorded. A semiquantitative reverse transcriptase polymerase chain reaction study of Foxp3 expression and a flow cytometry study of CD4 and CD25 markers of T lymphocytes were conducted to determine whether CD4+CD25+ Foxp3high regulatory T cells (Treg) were generated in vivo.
Results
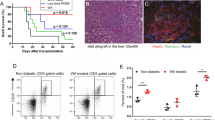
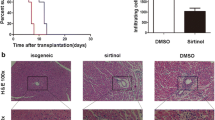
BALB/c mice given CST (0.2 or 2 mg/kg) had prolonged graft survival (median survival time [MST], 13 and 14 days, respectively; P<0.05 compared with controls). SMS 201–995 at the same concentrations did not have a significant effect on allograft survival (MST, 8 days for both groups). We found more than a twofold increase of CD4+CD25+ Treg cells in the CD4+ T-cell population and the expression of Foxp3 was up-regulated in the CST treatment groups, compared with control and SMS 201-995 treatment groups.
Conclusion
In our study, CST induced a significant prolongation in survival time of allogeneic skin grafts and increased the generation of CD4+CD25+ Foxp 3high Treg cells. These results suggest that CST may become a new modality in controlling allograft rejection.
Similar content being viewed by others
References
Sykes M. Immune tolerance: mechanisms and application in clinical transplantation. J Intern Med. 2007;262:288–310.
Kang SM, Tang Q, Bluestone JA. CD4+ CD25+ regulatory T cells in transplantation: progress, challenges and prospects. Am J Transplant. 2007;7:1457–1463.
Lim DG, Joe IY, Park YH, et al. Effect of immunosuppressants on the expansion and function of naturally occurring regulatory T cells. Transpl Immunol. 2007;18:94–100.
Pozo D, Gonzalez-Rey E, Chorny A, Anderson P, Varela N, Delgado M. Tuning immune tolerance with vasoactive intestinal peptide: a new therapeutic approach for immune disorders. Peptides. 2007;28: 1833–1846.
de Lecea L, del Rio JA, Criado JR, et al. Cortistatin is expressed in a distinct subset of cortical interneurons. J Neurosci. 1997;17:5868–5880.
Spier AD, de Lecea L. Cortistatin: a member of the somatostatin neuropeptide family with distinct physiological functions. Brain Res Brain Res Rev. 2000;33:228–241.
Moller LN, Stidsen CE, Hartmann B, Holst JJ. Somatostatin receptors. Biochim Biophys Acta. 2003;1616:1–84.
Broglio F, Papotti M, Muccioli G, Ghigo E. Brain-gut communication: cortistatin, somatostatin and ghrelin. Trends Endocrinol Metab. 2007;18:246–251.
Dalm VA, van Hagen PM, van Koetsveld PM, et al. Cortistatin rather than somatostatin as a potential endogenous ligand for somatostatin receptors in the human immune system. J Clin Endocrinol Metab. 2003;88:270–276.
Gonzalez-Rey E, Chorny A, Del Moral RG, Varela N, Delgado M. Therapeutic effect of cortistatin on experimental arthritis by downregulating inflammatory and Th1 responses. Ann Rheum Dis. 2007;66: 582–588.
Gonzalez-Rey E, Varela N, Sheibanie AF, Chorny A, Ganea D, Delgado M. Cortistatin, an antiinflammatory peptide with therapeutic action in inflammatory bowel disease. Proc Natl Acad Sci U S A. 2006;103:4228–4233.
Pan PY, Ozao J, Zhou Z, Chen SH. Advancements in immune tolerance. Adv Drug Deliv Rev. 2008;60:91–105.
Billingham RE, Medawar PB. The technique of free skin grafting in mammals. J Exp Biol. 1951;28:385–402.
Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3. Science. 2003;299:1057–1061.
Khattri R, Cox T, Yasayko SA, Ramsdell F. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat Immunol. 2003;4:337–342.
Wood KJ, Sakaguchi S. Regulatory T cells in transplantation tolerance. Nat Rev Immunol. 2003;3:199–210.
Thompson C, Powrie F. Regulatory T cells. Current Opin Pharmacol. 2004;4:408–414.
Sakaguchi S, Powrie F. Emerging challenges in regulatory T cell function and biology. Science. 2007;317:627–629.
Salama AD, Najafian N, Clarkson MR, Harmon WE, Sayegh MH. Regulatory CD25+ T cells in human kidney transplant recipients. J Am Soc Nephrol. 2003;14: 1643–1651.
Meloni F, Vitulo P, Bianco AM, et al. Regulatory CD4+CD25+ T cells in the peripheral blood of lung transplant recipients: correlation with transplant outcome. Transplantation. 2004;77:762–766.
Pandiyan P, Zheng L, Ishihara S, Reed J, Lenardo MJ. CD4+CD25+Foxp3+ regulatory T cells induce cytokine deprivationmediated apoptosis of effector CD4+ T cells. Nat Immunol. 2007;8:1353–1362.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, J., Zhao, R., Zhang, F. et al. Control of allograft rejection in mice by applying a novel neuropeptide, cortistatin. Adv Therapy 25, 1331–1341 (2008). https://doi.org/10.1007/s12325-008-0121-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-008-0121-z