Abstract
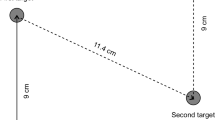
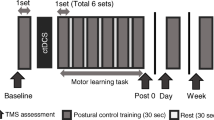
This study aimed to investigate the effects of repetitive transcranial magnetic stimulation (rTMS) of the cerebellum on changes in motor performance during a series of repetitive ballistic-targeting tasks. Twenty-two healthy young adults (n = 12 in the active-rTMS group and n = 10 in the sham rTMS group) participated in this study. The participants sat on a chair in front of a monitor and fixed their right forearms to a manipulandum. They manipulated the handle with the flexion/extension of the wrist to move the bar on the monitor. Immediately after a beep sound was played, the participant moved the bar as quickly as possible to the target line. After the first 10 repetitions of the ballistic-targeting task, active or sham rTMS (1 Hz, 900 pulses) was applied to the right cerebellum. Subsequently, five sets of 100 repetitions of this task were conducted. Participants in the sham rTMS group showed improved reaction time, movement time, maximum velocity of movement, and targeting error after repetition. However, improvements were inhibited in the active-rTMS group. Low-frequency cerebellar rTMS may disrupt motor learning during repetitive ballistic-targeting tasks. This supports the hypothesis that the cerebellum contributes to motor learning and motor-error correction in ballistic-targeting movements.





Similar content being viewed by others
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Manto M, Bower JM, Conforto AB, Delgado-Garcia JM, da Guarda SN, Gerwig M, et al. Consensus paper: roles of the cerebellum in motor control--the diversity of ideas on cerebellar involvement in movement. Cerebellum. 2012;11:457–87. https://doi.org/10.1007/s12311-011-0331-9.
Spampinato DA, Block HJ, Celnik PA. Cerebellar-M1 Connectivity changes associated with motor learning are somatotopic specific. J Neurosci. 2017;37:2377–86. https://doi.org/10.1523/JNEUROSCI.2511-16.2017.
Manto M, Gandini J, Feil K, Strupp M. Cerebellar ataxias: an update. Curr Opin Neurol. 2020;33:150–60. https://doi.org/10.1097/WCO.0000000000000774.
Berardelli A, Hallett M, Rothwell JC, Agostino R, Manfredi M, Thompson PD, Marsden CD. Single-joint rapid arm movements in normal subjects and in patients with motor disorders. Brain. 1996;119(Pt 2):661–74. https://doi.org/10.1093/brain/119.2.661.
Hallett M, Berardelli A, Matheson J, Rothwell J, Marsden CD. Physiological analysis of simple rapid movements in patients with cerebellar deficits. J Neurol Neurosurg Psychiatry. 1991;54:124–33. https://doi.org/10.1136/jnnp.54.2.124.
Lo YL, Fook-Chong S, Chan LL, Ong WY. Cerebellar control of motor activation and cancellation in humans: an electrophysiological study. Cerebellum. 2009;8:302–11. https://doi.org/10.1007/s12311-009-0095-7.
Irlbacher K, Voss M, Meyer BU, Rothwell JC. Influence of ipsilateral transcranial magnetic stimulation on the triphasic EMG pattern accompanying fast ballistic movements in humans. J Physiol. 2006;574:917–28. https://doi.org/10.1113/jphysiol.2006.108563.
Honda T, Mitoma H, Yoshida H, Bando K, Terashi H, Taguchi T, Miyata Y, Kumada S, Hanakawa T, Aizawa H, Yano S, Kondo T, Mizusawa H, Manto M, Kakei S. Assessment and rating of motor cerebellar ataxias with the Kinect v2 depth sensor: extending our appraisal. Front Neurol. 2020;11:179. https://doi.org/10.3389/fneur.2020.00179.
Honda T, Nagao S, Hashimoto Y, Ishikawa K, Yokota T, Mizusawa H, Ito M. Tandem internal models execute motor learning in the cerebellum. Proc Natl Acad Sci U S A. 2018;115:7428–33. https://doi.org/10.1073/pnas.1716489115.
Bando K, Honda T, Ishikawa K, Takahashi Y, Mizusawa H, Hanakawa T. Impaired adaptive motor learning is correlated with cerebellar hemispheric gray matter atrophy in spinocerebellar ataxia patients: a voxel-based morphometry study. Front Neurol. 2019;10:1183. https://doi.org/10.3389/fneur.2019.01183.
Kagaya K, Patek SN. Feed-forward motor control of ultrafast, ballistic movements. J Exp Biol. 2016;219:319–33. https://doi.org/10.1242/jeb.130518.
Popa LS, Ebner TJ. Cerebellum, predictions and errors. Front Cell Neurosci. 2018;12:524. https://doi.org/10.3389/fncel.2018.00524.
Lundbye-Jensen J, Petersen TH, Rothwell JC, Nielsen JB. Interference in ballistic motor learning: specificity and role of sensory error signals. PLoS ONE. 2011;6: e17451. https://doi.org/10.1371/journal.pone.0017451.
Criscimagna-Hemminger SE, Bastian AJ, Shadmehr R. Size of error affects cerebellar contributions to motor learning. J Neurophysiol. 2010;103:2275–84. https://doi.org/10.1152/jn.00822.2009.
Weissman-Fogel I, Granovsky Y. The, “virtual lesion” approach to transcranial magnetic stimulation: studying the brain-behavioral relationships in experimental pain. Pain Rep. 2019;4: e760. https://doi.org/10.1097/PR9.0000000000000760.
Fleischmann R, Triller P, Brandt SA, Schmidt SH. Human premotor corticospinal projections are engaged in motor preparation at discrete time intervals: a TMS-induced virtual lesion study. Front Neuroergonomics. 2021;2. https://doi.org/10.3389/fnrgo.2021.678906.
Hardwick RM, Lesage E, Miall RC. Cerebellar transcranial magnetic stimulation: the role of coil geometry and tissue depth. Brain Stimul. 2014. https://doi.org/10.1016/j.brs.2014.04.009.
Ugawa Y, Uesaka Y, Terao Y, Hanajima R, Kanazawa I. Magnetic stimulation over the cerebellum in humans. Ann Neurol. 1995;37:703–13. https://doi.org/10.1002/ana.410370603.
Matsugi A, Iwata Y, Mori N, Horino H, Hiraoka K. Long latency electromyographic response induced by transcranial magnetic stimulation over the cerebellum preferentially appears during continuous visually guided manual tracking task. Cerebellum. 2013;12:147–54. https://doi.org/10.1007/s12311-012-0402-6.
Hardwick RM, Lesage E, Miall RC. Cerebellar transcranial magnetic stimulation: the role of coil geometry and tissue depth. Brain Stimul. 2014;7:643–9. https://doi.org/10.1016/j.brs.2014.04.009.
Hiraoka K, Horino K, Yagura A, Matsugi A. Cerebellar TMS evokes a long latency motor response in the hand during a visually guided manual tracking task. Cerebellum. 2010;9:454–60. https://doi.org/10.1007/s12311-010-0187-4.
van Dun K, Bodranghien F, Manto M, Marien P. Targeting the cerebellum by noninvasive neurostimulation: a review. Cerebellum. 2017;16:695–741. https://doi.org/10.1007/s12311-016-0840-7.
Tanaka H, Matsugi A, Okada Y. The effects of imaginary voluntary muscle contraction and relaxation on cerebellar brain inhibition. Neurosci Res. 2017. https://doi.org/10.1016/j.neures.2017.11.004.
Popa T, Russo M, Meunier S. Long-lasting inhibition of cerebellar output. Brain Stimul. 2010;3:161–9. https://doi.org/10.1016/j.brs.2009.10.001.
Fierro B, Giglia G, Palermo A, Pecoraro C, Scalia S, Brighina F. Modulatory effects of 1 Hz rTMS over the cerebellum on motor cortex excitability. Exp Brain Res. 2007;176:440–7. https://doi.org/10.1007/s00221-006-0628-y.
Matsugi A, Yoshida N, Nishishita S, Okada Y, Mori N, Oku K, Douchi S, Hosomi K, Saitoh Y. Cerebellum-mediated trainability of eye and head movements for dynamic gazing. PLoS ONE. 2019;14: e0224458. https://doi.org/10.1371/journal.pone.0224458.
Miall RC, Christensen LO. The effect of rTMS over the cerebellum in normal human volunteers on peg-board movement performance. Neurosci Lett. 2004;371:185–9. https://doi.org/10.1016/j.neulet.2004.08.067.
Jenkinson N, Miall RC. Disruption of saccadic adaptation with repetitive transcranial magnetic stimulation of the posterior cerebellum in humans. Cerebellum. 2010;9:548–55. https://doi.org/10.1007/s12311-010-0193-6.
Matsugi A, Kamata N, Tanaka T, Hiraoka K. Long latency fluctuation of the finger movement evoked by cerebellar TMS during visually guided manual tracking task. Indian J Physiol Pharmacol. 2012;56:193–200.
Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods. 2007;39:175–91.
Team J. JASP (Version 0.16) [Computer software]. 2021.
Hadipour-Niktarash A, Lee CK, Desmond JE, Shadmehr R. Impairment of retention but not acquisition of a visuomotor skill through time-dependent disruption of primary motor cortex. J Neurosci. 2007;27:13413–9. https://doi.org/10.1523/JNEUROSCI.2570-07.2007.
Wolkorte R, Kamphuis J, Zijdewind I. Increased reaction times and reduced response preparation already starts at middle age. Front Aging Neurosci. 2014;6:79. https://doi.org/10.3389/fnagi.2014.00079.
Viejo G, Khamassi M, Brovelli A, Girard B. Modeling choice and reaction time during arbitrary visuomotor learning through the coordination of adaptive working memory and reinforcement learning. Front Behav Neurosci. 2015;9:225. https://doi.org/10.3389/fnbeh.2015.00225.
Bares M, Apps R, Avanzino L, Breska A, D’Angelo E, Filip P, Gerwig M, Ivry RB, Lawrenson CL, Louis ED, Lusk NA, Manto M, Meck WH, Mitoma H, Petter EA. Consensus paper: decoding the contributions of the cerebellum as a time machine. From neurons to clinical applications. Cerebellum. 2019;18:266–86. https://doi.org/10.1007/s12311-018-0979-5.
Koziol LF, Budding D, Andreasen N, D’Arrigo S, Bulgheroni S, Imamizu H, Ito M, Manto M, Marvel C, Parker K, Pezzulo G, Ramnani N, Riva D, Schmahmann J, Vandervert L, Yamazaki T. Consensus paper: the cerebellum’s role in movement and cognition. Cerebellum. 2014;13:151–77. https://doi.org/10.1007/s12311-013-0511-x.
Spampinato D, Celnik P. Multiple motor learning processes in humans: defining their neurophysiological bases. Neuroscientist. 2021;27:246–67. https://doi.org/10.1177/1073858420939552.
Bolognini N, Ro T. Transcranial magnetic stimulation: disrupting neural activity to alter and assess brain function. J Neurosci. 2010;30:9647–50. https://doi.org/10.1523/JNEUROSCI.1990-10.2010.
Lannin NA, Horsley SA, Herbert R, McCluskey A, Cusick A. Splinting the hand in the functional position after brain impairment: a randomized, controlled trial. Arch Phys Med Rehabil. 2003;84:297–302. https://doi.org/10.1053/apmr.2003.50031.
Lee JA, Sechachalam S. The effect of wrist position on grip endurance and grip strength. J Hand Surg Am. 2016;41:e367–73. https://doi.org/10.1016/j.jhsa.2016.07.100.
Matsugi A, Mori N, Uehara S, Kamata N, Oku K, Mukai K, Nagano K. Task dependency of the long-latency facilitatory effect on the soleus H-reflex by cerebellar transcranial magnetic stimulation. NeuroReport. 2014;25:1375–80. https://doi.org/10.1097/WNR.0000000000000275.
Matsugi A, Mori N, Uehara S, Kamata N, Oku K, Okada Y, Kikuchi Y, Mukai K, Nagano K. Effect of cerebellar transcranial magnetic stimulation on soleus Ia presynaptic and reciprocal inhibition. NeuroReport. 2015;26:139–43. https://doi.org/10.1097/WNR.0000000000000315.
Matsugi A, Okada Y. Cerebellar transcranial direct current stimulation modulates the effect of cerebellar transcranial magnetic stimulation on the excitability of spinal reflex. Neurosci Res. 2020;150:37–43. https://doi.org/10.1016/j.neures.2019.01.012.
Matsugi A. Do changes in spinal reflex excitability elicited by transcranial magnetic stimulation differ based on the site of cerebellar stimulation? Somatosens Mot Res. 2018;35:1–6. https://doi.org/10.1080/08990220.2018.1465403.
McCall AA, Miller DM, Yates BJ. Descending influences on vestibulospinal and vestibulosympathetic reflexes. Front Neurol. 2017;8:112. https://doi.org/10.3389/fneur.2017.00112.
Riddle CN, Edgley SA, Baker SN. Direct and indirect connections with upper limb motoneurons from the primate reticulospinal tract. J Neurosci. 2009;29:4993–9. https://doi.org/10.1523/JNEUROSCI.3720-08.2009.
Ebner TJ, Bloedel JR, Vitek JL, Schwartz AB. The effects of cerebellar stimulation on the stretch reflex in the spastic monkey. Brain. 1982;105(Pt 3):425–42. https://doi.org/10.1093/brain/105.3.425.
Mukaiyama K, Irie K, Takeda M, Yamashita R, Uemura S, Kanazawa S, Nagai-Tanima M, Aoyama T. Load distribution and forearm muscle activity during cylinder grip at various grip strength values. Hand Surg Rehabil. 2022. https://doi.org/10.1016/j.hansur.2021.12.010.
Markanday A, Messner J, Thier P. A loss of a velocity-duration trade-off impairs movement precision in patients with cerebellar degeneration. Eur J Neurosci. 2018;48:1976–89. https://doi.org/10.1111/ejn.14062.
Klomjai W, Katz R, Lackmy-Vallee A. Basic principles of transcranial magnetic stimulation (TMS) and repetitive TMS (rTMS). Ann Phys Rehabil Med. 2015;58:208–13. https://doi.org/10.1016/j.rehab.2015.05.005.
Acknowledgements
We thank Akari Hayashi, Yuna Fujii, Keigo Maehara, and Ayumu Watanabe of Shijonawate Gakuen University, who assisted with the many experiments conducted for the purposes of this research. We thank all the volunteers for their participation in this experiment and Editage (www.editage.jp) for providing English language editing.
Funding
This study was funded by JSPS KAKENHI (grant number 20K11298). This study was partially supported by the Japan Agency for Medical Research and Development (21 dm0307007).
Author information
Authors and Affiliations
Contributions
Matsugi A, Nishishita S, and Yoshida N conceptualized and designed this study, and other authors revised the experimental protocols. The experimental equipment for magnetic stimulation was provided by Mori N, Hosomi K, and Saitoh Y, whereas Matsugi A, Nishishita S, and Yoshida N provided other equipment. The experiments were conducted by Matsugi A and Mori N. Nishishita S and Yoshida N analyzed all angle data. Tanaka H and Douchi S analyzed all EMG data. The formal analysis was conducted mainly by Matsugi, Nishishita, and Yoshida, and other authors advised all analyses. Honda T and Odagaki M supervised the study. Funding acquisition was conducted by Matsugi A, Bando K, Kikuchi Y, Nakano H, Okada Y, Mori N, Hosomi K, and Saitoh Y. The original draft of the manuscript was written by Matsugi A, and all authors reviewed, edited, and approved the manuscript prior to journal submission.
Corresponding author
Ethics declarations
Ethics Approval
This study was approved by the Ethics Committee of Shijonawate Gakuen University (approval code: 19–9).
Consent to Publication
All participants were briefed on the publication plans and signed a consent form before participation.
Informed Consent
All participants were briefed on the experiment and signed a consent form before participation.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Matsugi, A., Nishishita, S., Yoshida, N. et al. Impact of Repetitive Transcranial Magnetic Stimulation to the Cerebellum on Performance of a Ballistic Targeting Movement. Cerebellum 22, 680–697 (2023). https://doi.org/10.1007/s12311-022-01438-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-022-01438-9