Abstract
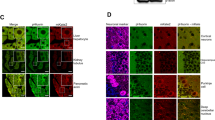
Recent studies using both dissociated and organotypic cell cultures have shown that heterozygous Lurcher (Lc/+) Purkinje cells (PCs) grown in vitro share many of the same survival and morphological characteristics as Lc/+ PCs in vivo. We have used this established tissue culture system as a valuable model for studying cell death mechanisms in a relatively simple system where neurodegeneration is induced by a constitutive cation leak mediated by the Lurcher mutation in the δ2 glutamate receptor (GluRδ2). In this study, Ca++ imaging and immunocytochemistry studies indicate that intracellular levels of Ca++ are chronically increased in Lc/+ PCs and the concentration and/or distribution of the conventional PKCγ isoform is altered in degenerating Lc/+ PCs. To begin to characterize the molecular mechanisms that regulate Lc/+ PC death, the contributions of conventional PKC pathways and of two MAP kinase family members, JNK and p38, were examined in slice cultures from wild-type and Lc/+ mutant mouse cerebellum. Cerebellar slice cultures from P0 pups were treated with either a conventional PKC inhibitor, a JNK inhibitor, or a p38 inhibitor either from 0 to 14 or 7 to 14 DIV. Treatment with either of the three inhibitors from 0 DIV significantly increased wild type and Lc/+ PC survival through 14 DIV, but only Lc/+ PC survival was significantly increased following treatments from 7 to 14 DIV. The results suggest that multiple PC death pathways are induced by the physical trauma of making organotypic slice cultures, naturally-occurring postnatal cell death, and the GluRδ2Lc mutation.






Similar content being viewed by others
References
Vogel MW, Caston J, Yuzaki M, Mariani J. The Lurcher mouse: fresh insights from an old mutant. Brain Res. 2007;1140:4–18.
Yue Z, Horton A, Bravin M, DeJager PL, Selimi F, Heintz N. A novel protein complex linking the delta 2 glutamate receptor and autophagy: implications for neurodegeneration in lurcher mice. Neuron. 2002;35(5):921–33.
Selimi F, Lohof AM, Heitz S, Lalouette A, Jarvis CI, Bailly Y, et al. Lurcher GRID2-induced death and depolarization can be dissociated in cerebellar Purkinje cells. Neuron. 2003;37(5):813–9.
Dumesnil-Bousez N, Sotelo C. Early development of the Lurcher cerebellum: Purkinje cell alterations and impairment of synaptogenesis. J Neurocytol. 1992;21:506–29.
Norman DJ, Feng L, Cheng SS, Gubbay J, Chan E, Heintz N. The lurcher gene induces apoptotic death in cerebellar Purkinje cells. Development. 1995;121(4):1183–93.
Selimi F, Doughty M, Delhaye-Bouchaud N, Mariani J. Target-related and intrinsic neuronal death in Lurcher mutant mice are both mediated by caspase-3 activation. J Neurosci. 2000;20:992–1000.
Wullner U, Loschmann P-A, Weller M, Klockgether T. Apoptotic cell death in the cerebellum of mutant weaver and lurcher mice. Neurosci Lett. 1995;200:109–12.
Cheng SS, Heintz N. Massive loss of mid- and hindbrain neurons during embryonic development of homozygous lurcher mice. J Neurosci. 1997;17(7):2400–7.
Selimi F, Vogel MW, Mariani J. Bax inactivation in Lurcher mutants rescues cerebellar granule cells but not Purkinje cells or inferior olivary neurons. J Neurosci. 2000;20(14):5339–45.
Doughty ML, De Jager PL, Korsmeyer SJ, Heintz N. Neurodegeneration in Lurcher mice occurs via multiple cell death pathways. J Neurosci. 2000;20(10):3687–94.
McFarland R, Blokhin A, Sydnor J, Mariani J, Vogel MW. Oxidative stress, nitric oxide, and the mechanisms of cell death in Lurcher Purkinje cells. Devel Neurobio. 2007;67:1032–46.
Giorgi C, Agnoletto C, Baldini C, Bononi A, Bonora M, Marchi S, et al. Redox control of protein kinase C: cell- and disease-specific aspects. Antioxid Redox Signal. 2010;13(7):1051–85.
Junttila MR, Li SP, Westermarck J. Phosphatase-mediated crosstalk between MAPK signaling pathways in the regulation of cell survival. FASEB J. 2008;22(4):954–65.
Shen HM, Liu ZG. JNK signaling pathway is a key modulator in cell death mediated by reactive oxygen and nitrogen species. Free Radic Biol Med. 2006;40(6):928–39. Epub 2005 Nov 21.
Zuo J, De Jager PL, Takahashi KA, Jiang W, Linden DJ, Heintz N. Neurodegeneration in Lurcher mice caused by mutation in δ2 glutamate receptor. Nature. 1997;388:769–73.
Letellier M, Demais V, Bailly Y, Sherrard RM, Mariani J, Lohof AM. Reinnervation of late post-natal Purkinje cells by climbing fibres: neosynaptogenesis without transient multi-innervation. J Neurosci. 2007;27:5373–83.
Metzger F, Kapfhammer JP. Protein kinase C: its role in activity-dependent Purkinje cell dendritic development and plasticity. Cerebellum. 2003;2(3):206–14.
Zanjani HS, McFarland R, Cavelier P, Blokhin A, Gautheron V, Levenes C, et al. Death and survival of heterozygous Lurcher Purkinje cells in vitro. Dev Neurobiol. 2009;69(8):505–17.
Repici M, Zanjani HS, Gautheron V, Borsello T, Dusart I, Mariani J. Specific JNK inhibition by D-JNKI1 protects Purkinje cells from cell death in Lurcher mutant mouse. Cerebellum. 2008;7(4):534–8.
Lu W, Tsirka SE. Partial rescue of neural apoptosis in the Lurcher mutant mouse through elimination of tissue plasminogen activator. Development. 2002;129(8):2043–50.
Armstrong CL, Duffin CA, McFarland R, Vogel MW. Mechanisms of compartmental Purkinje cell death and survival in the Lurcher mutant mouse. Cerebellum. 2010;10(3):504–14.
Duffin CA, McFarland R, Sarna JR, Vogel MW, Armstrong CL. Heat shock protein 25 expression and preferential Purkinje cell survival in the Lurcher mutant mouse cerebellum. J Comp Neurol. 2010;518:1892–907.
Ghoumari AM, Wehrle R, Bernard O, Sotelo C, Dusart I. Implication of Bcl-2 and caspase-3 in age-related Purkinje cell death in murine organotypic culture: an in vitro model to study apoptosis. Eur J Neurosci. 2000;12(8):2935–49.
Ghoumari AM, Wehrle R, De Zeeuw CI, Sotelo C, Dusart I. Inhibition of protein kinase C prevents Purkinje cell death but does not affect axonal regeneration. J Neurosci. 2002;22(9):3531–42.
Ghoumari AM, Dusart I, El-Etr M, Tronche F, Sotelo C, Schumacher M, et al. Mifepristone (RU486) protects Purkinje cells from cell death in organotypic slice cultures of postnatal rat and mouse cerebellum. Proc Natl Acad Sci U S A. 2003;100(13):7953–8.
Repici M, Wehrle R, Antoniou X, Borsello T, Dusart I. c-Jun N-terminal kinase (JNK) and p38 play different roles in age-related Purkinje cell death in murine organotypic culture. Cerebellum. 2010;10(2):281–90.
Marin-Teva JL, Dusart I, Colin C, Gervais A, van Rooijen N, Mallat M. Microglia promote the death of developing Purkinje cells. Neuron. 2004;41(4):535–47.
Bennett BL, Sasaki DT, Murray BW, O’Leary EC, Sakata ST, Xu W, et al. SP600125, an anthrapyrazolone inhibitor of Jun N-terminal kinase. Proc Natl Acad Sci U S A. 2001;98(24):13681–6.
Bain J, Plater L, Elliott M, Shpiro N, Hastie CJ, McLauchlan H, et al. The selectivity of protein kinase inhibitors: a further update. Biochem J. 2007;408(3):297–315.
Martiny-Baron G, Kazanietz MG, Mischak H, Blumberg PM, Kochs G, Hug H, et al. Selective inhibition of protein kinase C isozymes by the indolocarbazole Go 6976. J Biol Chem. 1993;268(13):9194–7.
Bright R, Mochly-Rosen D. The role of protein kinase C in cerebral ischemic and reperfusion injury. Stroke. 2005;36(12):2781–90. Epub 2005 Oct 27.
Behrens MM, Strasser U, Choi DW. Go 6976 is a potent inhibitor of neurotrophin-receptor intrinsic tyrosine kinase. J Neurochem. 1999;72(3):919–24.
De Zeeuw CI, Hansel C, Bian F, Koekkoek SK, van Alphen AM, Linden DJ, et al. Expression of a protein kinase C inhibitor in Purkinje cells blocks cerebellar LTD and adaptation of the vestibulo-ocular reflex. Neuron. 1998;20(3):495–508.
Hirono M, Sugiyama T, Kishimoto Y, Sakai I, Miyazawa T, Kishio M, et al. Phospholipase Cbeta4 and protein kinase Calpha and/or protein kinase CbetaI are involved in the induction of long term depression in cerebellar Purkinje cells. J Biol Chem. 2001;276(48):45236–42.
Naik MU, Benedikz E, Hernandez I, Libien J, Hrabe J, Valsamis M, et al. Distribution of protein kinase Mzeta and the complete protein kinase C isoform family in rat brain. J Comp Neurol. 2000;426(2):243–58.
Leitges M, Kovac J, Plomann M, Linden DJ. A unique PDZ ligand in PKCalpha confers induction of cerebellar long-term synaptic depression. Neuron. 2004;44(4):585–94.
Metzger F, Kapfhammer JP. Protein kinase C activity modulates dendritic differentiation of rat Purkinje cells in cerebellar slice cultures. Eur J Neurosci. 2000;12(6):1993–2005.
Kano M, Hashimoto K, Chen C, Abeliovich A, Aiba A, Kurihara H, et al. Impaired synapse elimination during cerebellar development in PKC gamma mutant mice. Cell. 1995;83:1223–31.
Schrenk K, Kapfhammer JP, Metzger F. Altered dendritic development of cerebellar Purkinje cells in slice cultures from protein kinase Cgamma-deficient mice. Neuroscience. 2002;110(4):675–89.
Alkon DL, Sun MK, Nelson TJ. PKC signaling deficits: a mechanistic hypothesis for the origins of Alzheimer’s disease. Trends Pharmacol Sci. 2007;28(2):51–60.
Reyland ME. Protein kinase Cdelta and apoptosis. Biochem Soc Trans. 2007;35:1001–4.
Steinberg SF. Structural basis of protein kinase C isoform function. Physiol Rev. 2008;88(4):1341–78.
Lin D, Takemoto DJ. Oxidative activation of protein kinase Cgamma through the C1 domain. Effects on gap junctions. J Biol Chem. 2005;280(14):13682–93.
Knapp LT, Kanterewicz BI, Hayes EL, Klann E. Peroxynitrite-induced tyrosine nitration and inhibition of protein kinase C. Biochem Biophys Res Commun. 2001;286(4):764–70.
Chakraborti T, Das S, Chakraborti S. Proteolytic activation of protein kinase Calpha by peroxynitrite in stimulating cytosolic phospholipase A2 in pulmonary endothelium: involvement of a pertussis toxin sensitive protein. Biochemistry. 2005;44(13):5246–57.
Nishiyama J, Matsuda K, Kakegawa W, Yamada N, Motohashi J, Mizushima N, et al. Reevaluation of neurodegeneration in lurcher mice: constitutive ion fluxes cause cell death with, not by, autophagy. J Neurosci. 2010;30(6):2177–87.
Brown GC. Nitric oxide and neuronal death. Nitric Oxide. 2010;23(3):153–65.
Zanjani H, Rondi-Reig L, Vogel M, Martinou JC, Delhaye-Bouchaud N, Mariani J. Overexpression of a Hu-bcl-2 transgene in Lurcher mutant mice delays Purkinje cell death. C R Acad Sci III. 1998;321(8):633–40.
Acknowledgments
This work was supported by a NIH grant NS 34309 to M.W.V. and J.M. and by funds from “Centre National de la Recherche Scientifique” and Université Pierre et Marie Curie; Laboratoire Internationaux Associé grant from UPMC. MV was recipient of a visiting professorship grant from UPMC. We thank Fabrice Machulka for his assistance in animal breeding. We gratefully acknowledge the considerable contribution of Dr. Pauline Cavelier in data acquisition. We thank the Cell Imaging and Flow Cytometry facility of the IFR83 (Paris, France) for access and technical support in microscopy.
Conflicts of Interest
The authors certify that there is no conflict of interest concerning the work presented in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zanjani, H.S., Lohof, A.M., McFarland, R. et al. Enhanced Survival of Wild-Type and Lurcher Purkinje Cells In Vitro Following Inhibition of Conventional PKCs or Stress-Activated MAP Kinase Pathways. Cerebellum 12, 377–389 (2013). https://doi.org/10.1007/s12311-012-0427-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-012-0427-x