Abstract
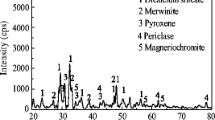
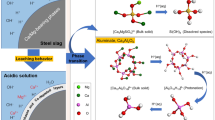
Experimental and modeling studies were conducted to delineate the reaction progress of chromite ore processing residue (COPR) upon hydration and the roles of brownmillerite and calcium aluminum chromium oxide hydrates (CAC) in the scavenging of hexavalent chromium. A kinetic study was conducted by preparing slurry samples with both synthetic brownmillerite and actual COPR samples at ambient temperatures. The hydration reaction of brownmillerite using synthetic brownmillerite was very fast (within 1 hour) and was completed within 2 days. However, the hydration of brownmillerite embedded in COPR to its hydration byproducts was not clearly observed after 7 days of aging. Newly formed Ca4Al2O6(CrO4)·14H2O (CAC-14) was observed after 1 hour of aging in both samples. However, the rate of formation of CAC-14 with synthetic brownmillerite was much faster than the COPR embedded brownmillerite. The reaction progress of synthetic brownmillerite and COPR upon chromate influx was simulated by a reaction path modeling program. The phase transformation of both samples can be predicted by the constructed model. Moreover, the formation of CACs upon chromate addition was predicted by the model, suggesting an effective sink for Cr(VI).
Similar content being viewed by others
References
Allied Signal, 1982, Process Description-Baltimore Works.
Burke, T., Fagliano, J., Goldoft, M., Hazen, R.E., Iglewicz, R., and McKee, T., 1991, Chromite ore processing residue in Hudson County, New Jersey. Environ Health Perspect, 92, 131–137.
Chatterji, S. and Jeffery, J.W., 1962, Studies of early stages of paste hydration of cement compounds. Journal of the American Ceramic Society, 45, 536–543.
Chrysochoou, M., Dermatas, D., Moon, D.H., and Wazne, M., 2005a, Role and properties of calcium aluminum oxide hydrates in chromium waste stabilization. In: Proceedings of the 1st International Conference on Environmental Science and Technology, New Orleans, Louisiana, January 23–26.
Chrysochoou, M., Moon, D.H., Dermatas, D., Wazne, M., Christodoulatos, C., Meng, X., Kaouris, M., Morris, J., French, C., and Sass, B., 2005b, Mineralogical analysis of Chromite Ore Processing Residue by X-ray Powder Diffraction. In: Proceedings of the 8th International In Situ and On-Site Bioremediation Symposium, Baltimore, Maryland, June 6–9.
Darrie, R.G., 2001, Commercial extraction technology and process waste disposal in the manufacture of chromium chemicals from ore. Environmental Geochemistry and Health, 23, 187–193.
Farmer, J.G., Graham, M.C., Thomas, R.P., Licona-Manzur, C., Paterson, E., and Campbell, C.D., 1999, Assessment and modeling of the environmental chemistry and potential for remediative treatment of chromium-contaminated land. Environmental Geochemistry and Health, 21, 331–337.
Geelhoed, J.S., Meeussen, J.C.L., Hillier, S., Lumsdon, D.G., Thomas, R.P., Farmer, J.G., and Paterson, E., 2002, Indentification and geochemical modeling of processes controlling leaching of Cr(VI) and other major elements from chromite ore processing residue. Geochimica et Cosmochimica Acta, 66, 3927–3942.
Graham, M.C., Farmer, J.G., Anderson, P., Paterson, E., Hillier, S., Lumsdon, D.G., and Bewley, R., 2006, Calcium polysulfide remediation of hexavalent chromium contamination from chromite ore processing residue. Science of the Total Environment, 364, 32–44.
Higgins, T.E., Halloran, A.R., Dobbins, M.E., and Pittignano, A.J., 1998, In-situ reduction of hexavalent chromium in alkaline soils enriched with chromite ore processing residue. Air & Waste Management Association, 48, 1100–1106.
Inorganic Crystal Structure Database, 2005, Fachinformationszentrum Karlsruhe, Germany.
International Centre for Diffraction Data (ICDD), 2002, Powder Diffraction File, PDF-2 Database Release.
Jagupilla, S.C., Moon, D.H., Wazne, M., Christodoulatos, C., and Kim, M.G., 2009, Effects of particle size and acid addition on the remediation of chromite ore processing residue using ferrous sulfate. Journal of Hazardous Materials, 168, 121–128.
James, B.R., 1996, The challenge of remediating chromium-contaminated soil. Environmental Science & Technology, 30, 248A–251A.
Jupe, A.C., Cockcroft, J.K., Barnes, P., Colston, S.L., Sankar, G., and Hall, C., 2001, The site occupancy of Mg in the brownmillerite structure and its effect on hydration properties: an X-ray/neutron diffraction and EXAFS study. Journal of Applied Crystallography, 34, 55–61.
Kremser, D.D., Sass, B.M., Clark, G.I., Bhargava, M., and French, C., 2005, Environmental forensics investigation of buried chromite ore processing residue. In: Proceedings of the 8th International In Situ and On-Site Bioremediation Symposium, Baltimore, Maryland, June 6–9.
Material’s Data Inc., 2005, Jade, Version 7.1, California, U.S.A.
Moon, D.H., Chrysochoou, M., Dermatas, D., Christodoulatos, C., Kaouris, M., and Morris, J., 2005a, Investigation of Ettringite Formation in Chrome Ore Processing Residue. In: Proceedings of the 8th International In Situ and On-Site Bioremediation Symposium, Baltimore, Maryland, June 6–9.
Moon, D.H., Dermatas, D., Sanchez, A.M., Chrysochoou, M., Wazne, M., and Grubb, D.G., 2008, Assessment of brownmillerite and periclase hydration in chromite ore processing residue at elevated temperature. In: Proceedings of GeoCongress 2008, New Orleans, Louisiana, March 9–12.
Moon, D.H., Dutko, P., Dermatas, D., Chrysochoou, M., Christodoulatos, C., Kaouris, M., and Morris, J., 2005b, Ettringite-Induced Laboratory Swelling in Chromite Ore Processing Residue. In: Proceedings of the 8th International In Situ and On-Site Biore-mediation Symposium, Baltimore, Maryland, June 6–9.
Rietveld, H.M., 1969, Aprofile refinement method for nuclear and magnetic structures. Journal of Applied Crystallography, 2, 65–71.
Taylor, H.F.W., 1997, The Chemistry of Cement, 2nd edition, Academic Press, London, 418 p.
Wazne, M., Jagupilla, S.C., Moon, D.H., Jagupilla, S.C., Christodoulatos, C., and Lim, M.G., 2007, Assessment of calcium polysulfide for the remediation of hexavalent chromium in chromite ore processing residue (COPR). Journal of Hazardous Materials, 143, 620–628.
Weng, C.H., Huang, C.P., Allen, H.E., Cheng, A.H.D., and Sanders, P.F., 1994, Chromium leaching behaviour in soil derived from chromite ore processing waste. Science of the Total Environment, 154, 71–86.
Wolery, T. and Jarek, R., 2003, EQ3/6 software package, version 8.0.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moon, D.H., Wazne, M. Impact of brownmillerite hydration on Cr(VI) sequestration in chromite ore processing residue. Geosci J 15, 287–296 (2011). https://doi.org/10.1007/s12303-011-0023-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12303-011-0023-y