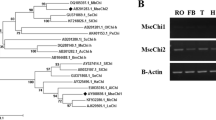
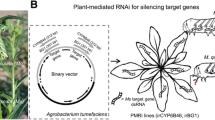
Abstract
Insect pests are one of the major biotic stresses limiting yield in commercially important crops. The lepidopteran polyphagous spotted pod borer, Maruca vitrata causes significant economic losses in legumes including pigeonpea. RNA interference (RNAi)-based gene silencing has emerged as one of the potential biotechnological tools for crop improvement. We report in this paper, RNAi in M. vitrata through exogenous administration of dsRNA with sequence specificity to three functionally important genes, Alpha-amylase (α-amylase), Chymotrypsin-like serine protease (CTLP) and Tropomyosin (TPM) into the larval haemolymph and their host-delivered RNAi in pigeonpea. Significant decline in the expression of selected genes supported by over-expression of DICER and generation of siRNA indicated the occurrence of RNAi in the dsRNA-injected larvae. Additionally, the onset of RNAi in the herbivore was demonstrated in pigeonpea, one of the prominent hosts, by host-delivered dsRNA. Transgenics in pigeonpea (cv. Pusa 992), a highly recalcitrant crop, were developed through a shoot apical meristem-targeted in planta transformation strategy and evaluated. Plant level bioassays in transgenic events characterized and selected at molecular level showed mortality of M. vitrata larvae as well as reduced feeding when compared to wild-type. Furthermore, molecular evidence for down regulation of target genes in the insects that fed on transgenic plants authenticated RNAi. Considering the variability of gene silencing in lepidopteran pests, this study provided corroborative proof for the possibility of gene silencing in M. vitrata through both the strategies.






Similar content being viewed by others
Data availability
The data generated during the current study are available with the corresponding author.
References
Bett B, Gollasch S, Moore A et al (2017) Transgenic cowpeas (Vigna unguiculata L. Walp) expressing Bacillus thuringiensis Vip3Ba protein are protected against the Maruca pod borer (Maruca vitrata). Plant Cell Tiss Organ Cult 131:335–345. https://doi.org/10.1007/s11240-017-1287-3
Cao T, Jin JP (2020) Evolution of fight muscle contractility and energetic efficiency. Front Physiol 11:1038. https://doi.org/10.3389/fphys.2020.01038
Chatterjee M, Yadav J, Rathinam M et al (2021) Exogenous administration of dsRNA for the demonstration of RNAi in Maruca vitrata (lepidoptera: crambidae). Biotech 11:197. https://doi.org/10.1007/s13205-021-02741-8
Chatterjee M, Yadav J, Vennila S, Shashank PR, Jaiswal N, Sreevathsa R, & Rao U (2019) Diversity analysis reveals genetic homogeneity among Indian populations of legume pod borer, Maruca vitrata (F.). 3 Biotech 9(9): 1–8. Doi: https://doi.org/10.1007/s13205-019-1850-1
Chougule NP, Doyle E, Fitches E, Gatehouse JA (2008) Biochemical characterization of midgut digestive proteases from Mamestra brassicae (cabbage moth; Lepidoptera: Noctuidae) and effect of soybean Kunitz inhibitor (SKTI) in feeding assays. J Insect Physiol 54(3):563–572. https://doi.org/10.1016/j.jinsphys.2007.12.005
Christiaens O, Niu J, Nji Tizi Taning C (2020) RNAi in Insects: a revolution in fundamental research and pest control applications. Insects 11(7):415. https://doi.org/10.3390/insects11070415
Chung SH, Feng H, Jander G (2021) Engineering pest tolerance through plant-mediated RNA interference. Curr Opin Plant Biol 60:102029. https://doi.org/10.1016/j.pbi.2021.102029
Cooper AM, Silver K, Zhang J, Park Y, Zhu KY (2019) Molecular mechanisms influencing efficiency of RNA interference in insects. Pest Manag Sci 75(1):18–28. https://doi.org/10.1002/ps.5126
Da Lage JL (2018) The amylases of insects. Int J Insect Sci. https://doi.org/10.1177/1179543318804783
Franzetti E, Huang ZJ, Shi YX, Xie K, Deng XJ, Li JP et al (2012) Autophagy precedes apoptosis during the remodelling of silkworm larval midgut. Apoptosis 17(3):305–324. https://doi.org/10.1007/s10495-011-0675-0
Guan RB, Li HC, Fan YJ, Hu SR, Christiaens O, Smagghe G, Miao XX (2018) A nuclease specific to lepidopteran insects suppresses RNAi. J Biol Chem 293(16):6011–6021. https://doi.org/10.1074/jbc.RA117.001553
Hada A, Kumari C, Phani V, Singh D, Chinnusamy V, Rao U (2020) Host-induced silencing of FMRF amide-like peptide genes, flp-1 and flp-12, in rice impairs reproductive fitness of the root-knot nematode Meloidogyne graminicola. Front Plant Sci 11:894. https://doi.org/10.3389/fpls.2020.00894
Hasegawa M, Kishino H, Yano T (1985) Dating the human-ape split by a molecular clock of mitochondrial DNA. J Mol Evol 22:160–174
Iwamoto H (2011) Structure, function and evolution of insect flight muscle. Biophysics 7:21–28. https://doi.org/10.2142/biophysics.7.21
Jeanmougin F, Thompson JD, Gouy M, Higgins DG, Gibson TJ (1998) Multiple sequence alignment with Clustal X. Trends Biochem Sci 23(10):403–405. https://doi.org/10.1016/S0968-0004(98)01285-7
Kaur A, Sharma M, Sharma C, Kaur H, Kaur N, Sharma S, et al. (2016) Pod borer resistant transgenic pigeon pea (Cajanus cajan L) expressing cry1Ac transgene generated through simplified Agrobacterium transformation of pricked embryo axes. Plant Cell, Tissue and Organ Culture (PCTOC) 127(3): 717–727 DOI: https://doi.org/10.1007/s11240-016-1055-9.
Kesiraju K, Sreevathsa R (2017) Apical meristem-targeted in planta transformation strategy: an overview on its utility in crop improvement. Agri Res Techol. https://doi.org/10.19080/ARTOAJ.2017.08.555734
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Kumar A, Jaiwal R, Sreevathsa R et al (2021) Transgenic cowpea plants expressing Bacillus thuringiensis Cry2Aa insecticidal protein imparts resistance to Maruca vitrata legume pod borer. Plant Cell Rep 40:583–594. https://doi.org/10.1007/s00299-020-02657-2
Lemaitre B, Miguel-Aliaga I (2013) The digestive tract of Drosophila melanogaster. Ann Rev Gen 47:377–404. https://doi.org/10.1146/annurev-genet-111212-133343
Mahalle RM, Taggar GK (2017) Yield loss assessment and establishment of economic threshold level of Maruca vitrata in pigeonpea. J Food Legumes 31(1):36–44
Naito Y, Yamada T, Matsumiya T, Ui-Tei K, Saigo K, Morishita S (2005) dsCheck: highly sensitive off-target search software for double-stranded RNA-mediated RNA interference. Nucleic Acids Res. Jul 1; 33 (Web Server issue), W589–91. doi: https://doi.org/10.1093/nar/gki419. PMID: 15980542; PMCID: PMC1160180.
Nei M, Kumar S (2000) Molecular evolution and phylogenetics. Oxford University Press, New York
Rathinam M, Marimuthu SK, Tyagi S, Kesiraju K, Prabha AL, Rao U, & Sreevathsa R (2019) Characterization and in planta validation of a CHI4 chitinase from Cajanus platycarpus (Benth.) Maesen for its efficacy against pod borer, Helicoverpa armigera (Hübner). Pest Management Science. https://doi.org/10.1002/ps.6260
Sankara Rao K, Sreevathsa R, Sharma PD et al (2008) In planta transformation of pigeon pea: a method to overcome recalcitrancy of the crop to regeneration in vitro. Physiol Mol Biol Plants 14:321–328. https://doi.org/10.1007/s12298-008-0030-2
Saxena H, Bandi SM, Revanasidda (2020) Sucking pests of pulse crops. In: Omkar (eds) Sucking pests of crops. Springer: Singapore
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108. https://doi.org/10.1038/nprot.2008.73
Scott JG, Michel K, Bartholomay LC, Siegfried BD, Hunter WB, Smagghe G et al (2013) Towards the elements of successful insect RNAi. J Insect Physiol 59(12):1212–1221. https://doi.org/10.1016/j.jinsphys.2013.08.014
Shinde KG, Naik KV, Raut PP, Desai VS, Mehendale SK (2017) Biology of pod borer, Maruca vitrata (Geyer) infesting lablab bean. Int J Curr Microbiol App Sci 6(9):67–74
Shivakumara TN, Sreevathsa R, Dash PK et al (2017) Overexpression of Pea DNA Helicase 45 (PDH45) imparts tolerance to multiple abiotic stresses in chilli (Capsicum annuum L.). Sci Rep 7:2760. https://doi.org/10.1038/s41598-017-02589-0
Singh S, Kumar NR, Maniraj R et al (2018) Expression of Cry2Aa, a Bacillus thuringiensis insecticidal protein in transgenic pigeon pea confers resistance to gram pod borer. Helicoverpa Armigera Sci 8:8820. https://doi.org/10.1038/s41598-018-26358-9
Srinivasan A, Chougule NP, Giri AP, Gatehouse JA, Gupta VS (2005) Pod borer (Helicoverpa armigera Hübn.) does not show specific adaptations in gut proteinases to dietary Cicer arietinum Kunitz proteinase inhibitor. J Insect Physiol 51(11):1268–1276. https://doi.org/10.1016/j.jinsphys.2005.07.005
Sujayanand GK, Chandra A, Pandey S and Bhatt S (2021) Seasonal Abundance of Spotted Pod Borer, Maruca vitrata Fabricius in Early Pigeonpea [Cajanus cajan (L.) Millsp.] and its Management through Farm scaping in Uttar Pradesh. Legume Research: An International Journal 44(2)
Tamura K, Nei M (1993) Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol Biol Evol 10:512–526
Ulrich J, Majumdar U, Schmitt-Engel C, Schwirz J, Schultheis D, Ströhlein N et al (2015) Large scale RNAi screen in Tribolium reveals novel target genes for pest control and the proteasome as prime target. BMC Genom 16(1):1–9. https://doi.org/10.1186/s12864-015-1880-y
Valencia-Jimenez A, Arboleda JW, Lopez Avila A, Grossi-de-Sa MF (2008) Digestive α-amylases from Tecia solanivora larvae (Lepidoptera: Gelechiidae): response to pH, temperature and plant amylase inhibitors. Bull Entomol Res 98:575–579
Vogel E, Santos D, Mingels L, Verdonckt TW, Broeck JV (2019) RNA interference in insects: protecting beneficials and controlling pests. Front Physiol 9:1912. https://doi.org/10.3389/fphys.2018.01912
Whyard S, Singh AD, Wong S (2009) Ingested double-stranded RNAs can act as species-specific insecticides. Insect Biochem Mol Biol 39(11):824–832. https://doi.org/10.1016/j.ibmb.2009.09.007
Xu J, Wang XF, Chen P, Liu FT, Zheng SC, Ye H, Mo MH (2016) RNA interference in moths: mechanisms, applications, and progress. Genes 7(10):88. https://doi.org/10.3390/genes7100088
Yang B, Huang W, Zhang J, Xu Q, Zhu S, Zhang Q et al (2016) Analysis of gene expression in the midgut of Bombyx mori during the larval molting stage. BMC Genom 17(1):1–16. https://doi.org/10.1186/s12864-016-3162-8
Zhang C, Zhou D, Zheng S, Liu L, Tao S, Yang L et al (2010) A chymotrypsin-like serine protease cDNA involved in food protein digestion in the common cutworm, Spodoptera litura: cloning, characterization, developmental and induced expression patterns, and localization. J Insect Physiol 56(7):788–799
Acknowledgements
We are thankful to Department of Biotechnology, Government of India (Grant No. BT/IC-2/ISCB/Phase-IV/Pigeonpea) for funding. We acknowledge Technical Officer, Mr. Purshotam Podar for helping us in insect collection from the field and maintenance of plants in greenhouse. We thank Dr. Pradeep Kumar Papolu for helping us during Gateway construct preparation. MC acknowledges School of Biotechnology, KIIT University, Bhubaneswar, Odisha for Ph.D registration.
Author information
Authors and Affiliations
Contributions
UR conceived and supervised the experiments. MC and JY performed the experiments. MC, MR and KK interpreted, analysed and arranged the experimental data. RS, UR and GC reviewed and finalized the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chatterjee, M., Yadav, J., Rathinam, M. et al. Amenability of Maruca vitrata (Lepidoptera: Crambidae) to gene silencing through exogenous administration and host-delivered dsRNA in pigeonpea (Cajanus cajan L.). Physiol Mol Biol Plants 28, 189–202 (2022). https://doi.org/10.1007/s12298-022-01133-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-022-01133-3