Abstract
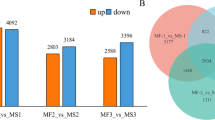
Male sterility is an important factor in improving crop quality and yield through heterosis breeding. In this study, we analyzed the transcriptomes of male fertile (MF) and male sterile (MS) alfalfa flower buds using the Illumina HiSeq™ 4000 platform. A total of 54.05 million clean reads were generated and assembled into 65,777 unigenes with an average length of 874 bp. The differentially expressed genes (DEGs) between the MF and MS flowers at three stages of pollen development were identified, and there were 3832, 5678 and 5925 DEGs respectively in stages 1, 2 and 3. GO and KEGG functional enrichment analysis revealed 12, 12, 6 and 12 key branch-point genes involved in circadian rhythm, transcription factors, pollen development and flavonoid biosynthesis. Our findings provide novel insights into the mechanism of male sterility in alfalfa.








Similar content being viewed by others
References
Ai Y, Zhang Q, Wang W, Zhang C, Cao Z, Bao M et al (2016) Transcriptomic analysis of differentially expressed genes during flower organ development in genetic male sterile and male fertile Tagetes erecta by digital gene-expression profiling. PLoS One 11(3):e0150892
Barr CM, Fishman L (2011) Cytoplasmic male sterility in mimulus hybrids has pleiotropic effects on corolla and pistil traits. Heredity 106:886–893
Bauchan GR (2009) Alfalfa (Medicago sativa ssp. sativa (L.). Genet Resour, Chromosome Eng, Crop Improv 5:11–39
Benjamini Y, Yekutieli D (2001) The control of the false discovery rate in multiple testing under dependency. Ann Stat 29:1165–1188
Cai LY, Shi FL, Chen HL, Li ZY, Li HY, Gao CP et al (2013) (2013) The heterosis of forage yield of cross combinations with alfalfa male sterile lines. Chinese J Grassland 35:24–30
Cecchetti V, Altamura MM, Falasca G, Costantino P, Cardarelli M (2008) Auxin regulates Arabidopsis anther dehiscence, pollen maturation, and filament elongation. Plant Cell 20:1760–1774
Chase CD (2007) Cytoplasmic male sterility: a window to the world of plant mitochondrial-nuclear interactions. Trends Genet 23:81–90
Chen CB, Marcus A, Li WX, Hu Y, Calzada JP, Grossniklaus U et al (2002) The Arabidopsis ATK1 gene is required for spindle morphogenesis in male meiosis. Development 29:2401–2409
Chen CM, Chen GJ, Cao BH, Lei JJ (2015) Transcriptional profiling analysis of genic male sterile-fertile Capsicum annuum reveal candidate genes for pollen development and maturation by RNA-Seq technology. Plant Cell Tissue Organ Cult 122:465–476
Childers WR (1952) Male sterility in Medicago sativa L. Sci Agric (ottawa) 32:351–364
Dong X, Feng H, Xu M, Lee J, Kim YK et al (2013) Comprehensive analysis of genic male sterility-related genes in brassica rapa using a newly developed Br 300K oligomeric chip. PLoS One 8(9):e72178
Fujii S, Kazama T, Yamada M, Toriyama K (2010) Discovery of global genomic re-organization based on comparison of two newly sequenced rice mitochondrial genomes with cytoplasmic male sterility-related genes. BMC Genomics 11:209
Gao CP, Shi FL, Li H (2005) Cyto-morphologyical studies on the stamen development of male sterile line MS-4 of Alfalfa. J Inner Mongolia Univ 36:288–293
Guo JX, Liu YG (2012) Molecular control of male reproductive development and pollen fertility in Rice. J Integr Plant Biol 54:967–978
Higginson T, Li SF, Parish RW (2010) AtMYB103 regulates tapetum and trichome development in Arabidopsis thaliana. Plant J 35:177–192
Hodžić A, Ristanović M, Zorn B, Tulic´C, Maver A, Novakovic´ I, Peterlin B, (2013) Genetic variation in circadian rhythm genes CLOCK and ARNTL as risk factor for male infertility. Plos ONE. 8:e59220
Jung KH, Han MJ, Lee YS, Kim YW, Hwang I, Kim MJ et al (2005) Rice Undeveloped Tapetum1 is a major regulator of early tapetum development. Plant Cell 17:2705–2722
Kang BH, Rancour DM, Bednarek SY (2010) The dynamin-like protein ADL1C is essential for plasma membrane maintenance during pollen maturation. Plant J 35:1–15
Kobayashi A, Sakamoto A, Kubo K, Rybka Z, Kanno Y, Takatsuji H (1998) Seven zinc-finger transcription factors are expressed sequentially during the development of anthers in petunia. Plant J 13:571–576
Lee SI, Muthusamy M, Nawaz MA, Hong JK, Lim MH, Kim JA, Jeong MJ (2019) Genome-wide analysis of spatiotemporal gene expression patterns during floral organ development in Brassica rapa. Mol Genet Genomics 294:1403–1420
Li H, Pinot F, Sauveplane V, Werck-Reichhart D, Diehl P, Schreiber L et al (2010) Cytochrome P450 family member CYP704B2 catalyzes the ω-hydroxylation of fatty acids and is required for anther cutin biosynthesis and pollen exine formation in rice. Plant Cell 22:173–190
Li JJ, Han SH, Ding XL, He TT, Yang SP, Gai JY (2015) Comparative transcriptome analysis between the cytoplasmic male sterile line NJCMS1A and its maintainer NJCMS1B in Soybean (Glycine max (L.) Merr.). PloS One 10(5):e0126771
Li ZQ, Ding B, Zhou XP, Wang GL (2017) The rice dynamin-related protein OsDRP1E negatively regulates programmed cell death by controlling the release of cytochrome c from mitochondria. PLoS Pathog 13:e1006157
Lin S, Su SW, Jin LB, Peng RY, Sun D, Ji H et al (2020) Identification of microRNAs and their targets in inflorescences of an Ogura-type cytoplasmic male-sterile line and its maintainer fertile line of turnip (Brassica rapa ssp. rapifera) via high-throughput sequencing and degradome analysis. PLoS One 15(7):e0236829
Liu XD, Huang J, Parameswaran S, Ito T, Seubert B, Auer M et al (2009) The SPOROCYTELESS/NOZZLE gene is involved in controlling stamen identity in Arabidopsis. Plant Physiol 151:1401–1411
Liu C, Ma N, Wang PY, Fu N, Shen HL (2013) Transcriptome sequencing and De novo analysis of a cytoplasmic male sterile line and its near-isogenic restorer line in Chili Pepper (Capsicum annuum L.). PloS One 8:e65209
Liu Z, Cai X, Seiler GJ, Jan CC (2014) Interspecific amphiploid-derived alloplasmic male sterility with defective anthers, narrow disc florets and small ray flowers in sunflower. Plant Breeding 133:742–747
Ma H (2005) Molecular genetic analyses of microsporogenesis and micro- gametogenesis in flowering plants. Annu Rev Plant Biol 56:393–434
Martens S, Preuss A, Matern U (2010) Multifunctional flavonoid dioxygenases: flavonol and anthocyanin biosynthesis in Arabidopsis thaliana L. Phytochemistry 71:1040–1049
Mayr E (1986) Joseph gottlieb kolreuter’s contributions to Biology. Osiris 2:135–176
Mehdi M, Anwar A (2009) Role of genetically engineered system of male sterility in hybrid production of vegetables. J Phytol 1:448–460
Mei SY, Liu TM, Wang ZW (2015) Comparative transcriptome profile of the cytoplasmic male sterile and fertile floral buds of radish (Raphanus sativus L). Int J Mol Sci. 17:42
Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B (2008) Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5:621–628
Rhee SJ, Seo M, Jang YJ, Cho S, Lee GP (2015) Transcriptome profiling of differentially expressed genes in floral buds and flowers of male sterile and fertile lines in watermelon. BMC Genomics 16:914
Rhee SJ, Kwon T, Seo M, Jang YJ, Sim TY, Cho S et al (2017) De novo-based transcriptome profiling of male-sterile and fertile watermelon lines. PLoS One 12(11):e0187147
Schuster SC (2008) Next-generation sequencing transforms today’s biology. Nat Methods 5:16–18
Scott RJ, Spielman M, Dickinson HG (2004) Stamen structure and function. Plant Cell 16(Suppl):S46–S60
Thingnaes E, Torre S, Ernstsen A, Moe R (2003) Day and night temperature responses in Arabidopsis: effects on gibberellin and auxin content, cell size, morphology and flowering time. Ann Bot 92:601–612
Ujino-Ihara T, Ueno S, Uchiyama K, Futamura N (2018) Comprehensive analysis of small RNAs expressed in developing male strobili of Cryptomeria japonica. PLoS One 13(3):e0193665
Wang J, Dang ZH, Zhang HR, Zheng LL, Borjigin T, Wang YC (2016) Gene transcript profiles in the desert plant Nitraria tangutorum during fruit development and ripening. Mol Genet Genomics 291:383–398
Wei MM, Song MZ, Fan SL, Yu SX (2013) Transcriptomic analysis of differentially expressed genes during anther development in genetic male sterile and wild type cotton by digital gene-expression profiling. BMC Genomics 14:97
Wu YF, Wei L, Wu RY, Duan QX (1986) A study on flower bud differentiation, the microspore development and the reason of male sterility in alfalafa. Grassl China 6:23–27
Wu YF, Temue B, Xu ZY, Luo HN (1998) Use of male sterile line in Alfalfa hybrid seed production. Grassl China 5:9–12
Yang CY, Vizcay-Barrena G, Conner K, Wilson ZA (2007) Male Sterility1 is required for tapetal development and pollen wall biosynthesis. Plant Cell 19:3530–3548
Zahn LM, Ma X, Altman NS, Zhang Q, Wall PK, Tian DL et al (2010) Comparative transcriptomics among floral organs of the basal eudicot Eschscholzia californica as reference for floral evolutionary developmental studies. Genome Biol 11:R101
Zhang W, Sun YJ, Timofejeva L, Chen CB, Grossniklaus U, Ma H (2006) Regulation of Arabidopsis tapetum development and function by Dysfynctional Tapetum1 (DYT1) encoding a putative bHLH transcription factor. Development 133:3085–3095
Zhang ZB, Zhu J, Gao JF, Wang C, Li H, Li H et al (2007) Transcription factor AtMYB103 is required for anther development by regulating tapetum development, callose dissolution and exine formation in Arabidopsis. Plant J 52:528–538
Zhang XM, Zhao L, Larson-Rabin Z, Li DZ, Guo ZH (2012) De Novo sequencing and characterization of the floral transcriptome of Dendrocalamus latiflorus (Poaceae: Bambusoideae). PloS One 7:e42082
Zhu J, Yang ZN (2013) The research progress of pollen wall development. Chin J Nat 35:112–117
Zhu QD, Song YL, Zhang GS, Ju L, Zhang J, Yu YG et al (2015) De novo assembly and transcriptome analysis of wheat with male sterility induced by the chemical hybridizing agent SQ-1. PLoS One 10(4):e0123556
Acknowledgements
The authors express their appreciation to the reviewers for their valuable comments on this manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (NO. 31260576), the Inner Mongolia Natural Science Foundation Project (NO. 2018LH03028), and the Doctor foundation of Baotou Medical College (NO. BSJJ201810).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: JW and FLS. Performed RNA extraction and qRT-PCR: JW and BX. Collected and analyzed the data: JW, FT, and BX. Wrote the paper JW and FLS. Edited the manuscript: FT, CPG, and XG. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflict of interest.
Consent for publication
All authors agree to publish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, J., Tang, F., Gao, C. et al. Comparative transcriptome between male fertile and male sterile alfalfa (Medicago varia). Physiol Mol Biol Plants 27, 1487–1498 (2021). https://doi.org/10.1007/s12298-021-01026-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-021-01026-x