Abstract
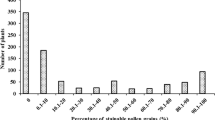
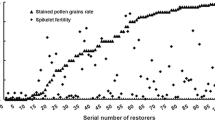
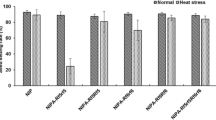
Iso-cytoplasmic restorers possess the same male sterile cytoplasm as the cytoplasmic male sterile (CMS) lines, thereby minimizing the potential cyto-nuclear conflict in the hybrids. Restoration of fertility of the wild abortive CMS is governed by two major genes namely, Rf3 and Rf4. Therefore, assessing the allelic status of these restorer genes in the iso-cytoplasmic restorers using molecular markers will not only help in estimating the efficiency of these genes either alone or in combination, in fertility restoration in the hybrids in different environments, but will also be useful in determining the efficacy of these markers. In the present study, the efficiency of molecular markers in identifying genotypes carrying restorer allele of the gene(s) Rf3 and Rf4, restoring male fertility of WA cytoplasm in rice was assessed in a set of 100 iso-cytoplasmic rice restorers using gene linked as well as candidate gene based markers. In order to validate the efficacy of markers in identifying the restorers, a sub-set of selected 25 iso-cytoplasmic rice restorers were crossed with four different cytoplasmic male sterile lines namely, IR 79156A, IR 58025A, Pusa 6A and RTN 12A, and the pollen and spikelet fertility of the F1s were evaluated at three different locations. Marker analysis showed that Rf4 was the predominant fertility restorer gene in the iso-cytoplasmic restorers and Rf3 had a synergistic effect on fertility restoration. The efficiency of gene based markers, DRCG-RF4-14 and DRRM-RF3-10 for Rf4 (87%) and Rf3 (84%) genes was higher than respective gene-linked SSR markers RM6100 (80%) and RM3873 (82%). It is concluded that the gene based markers can be effectively used in identifying fertility restorer lines obviating the need for making crosses and evaluating the F1s. Though gene based markers are more efficient, there is a need to identify functional polymorphisms which can provide 100% efficiency. Three iso-cytoplasmic restorers namely, PRR 300, PRR 363 and PRR 396 possessing both Rf4 and Rf3 genes and good fertility restoration have been identified which could be used further in hybrid rice breeding.





Similar content being viewed by others
References
Alavi M, Ahamadidhan A, Kamkar B, Kalateh M (2009) Mapping Rf3 locus in rice by SSR and CAPS markers. Int J Genet Mol Biol 7:121–126
Alvarado R (1999) Influence of air temperature on rice population, length of period from sowing to flowering and spikelet sterility. In: Hill JE, Hardy B (eds) Proceedings of the second temperate rice conference. Sacramento, California, pp 63–68
Betran FJ, Ribaut JM, Beck D, De Leon DG (2003) Genetic diversity, specific combining ability, heterosis in tropical maize under stress and non-stress environments. Crop Sci 43:797–806
Chaudhury RC, Virmani SS, Khush GS (1981) CMS lines in rice. Andhra Agric J 38:302–303
Falodun D, Njoku KL, Ogunyebi AL, Akinola MO (2012) Spikelet numbers, filled grains and spikelet fertility potential of NERICA rice (Mecux, Tox and WitA.4) grown in hydrocarbon polluted soils. J Res Environ Sci Toxicol 1(8):201–206
FAO (2014) FAO STAT agricultural data (on line), In: http://www.fao.org/available at http://faostat.fao.org, Rome, Italy: Food and Agriculture Organization (FAO)
Farrell TC, Fox KM, Williams RL, Fukai S (2006) Genotypic variation for cold tolerance during reproductive development in rice: screening with cold air and cold water. Field Crops Res 98:178–194
Ghara AG, Nematzadeh G, Bagheri N, Ebrahimi A, Oladi M (2012) Molecular and cytological evaluation of male sterile and restorer lines in hybrids rice. Int Res J Appl Basic Sci 3(1):183–189
Govinda Raj K, Virmani SS (1988) Genetics of fertility restoration of ‘WA’ type cytoplasmic genic male sterility in rice. Crop Sci 28:787–792
Joshi BK, Subedi LP, Gurung SB, Sharma RC (2007) Pollen and spikelet analysis in F1 rice hybrids and their parents. Nepal Agric Res J 8:120–126
Khalique MA, Joarder OI, Euus AM (1977) Heterosis and combining ability in a diallel cross of rice (Oryzasativa L.). Bangladesh J Agric Sci 4(2):137–145
Kutka F (2011) Open-pollinated vs. Hybrid maize cultivars. Sustainability 3:1531–1554
Li YC, Yuan LP (1986) Genetic analysis of fertility restoration in male sterile lines of rice. IRRI, Manila, Rice Genetics, pp 617–632
Lin SC, Yuan LP (1980) Hybrid rice breeding in China. In: Innovative approaches to rice breeding. IRRI, Manila, Philippines, pp. 35–51
Nas TMS, Casal CL, Li Z, Virmani SS (2003) Application of molecular markers for identification of restorers. Rice Genet News lett 20:69–71
Pande K, Ratho SN, Patnaik RN, Jachuk PJ (1990) Fertility restoration in cytoplasmic male sterile lines in rice. Oryza 27:232–238
Peng S, Laza RC, Visperas RM, Khush GS, Virk P, Zhu D (2004) Rice: progress in breaking the yield ceiling. “New directions for a diverse planet”. In: Proceedings of the 4th international crop science congress, 26 Sep–1 Oct 2004, Brisbane, Australia Published on CDROM. Web site www.cropscience.org.au
Prakash P (2003) Molecular mapping of fertility restorer gene(s) and validation of Rf-gene linked markers in rice. M.Sc. Dissertation, Indian Agricultural Research Institute, New Delhi
Revathi P, Medoju P, Singh AK, Sundaram RM, Sravan R, Senguttuvel P, Kemparaju KB, Hariprasad AS, Ramesha MS, Neeraja CN, Rani NS, Viraktamath BC (2013) Efficiency of molecular markers in identifying fertility restoration trait of WA-CMS system in rice. Indian J Genet Plant Breed 73(1):89–93
Salgotra RK, Katoch PC, Kaushik RP (2002) Identification of restorers and maintainers for cytoplasmic genic male sterile lines of rice. Oryza 39:55–57
Samanci B (1996) Phenotypic correlations between maize inbreds and their single cross hybrids in short season areas. Euphytica 89:291–296
Sheeba NK, Viraktamath BC, Sivaramakrishnan S, Gangashetti MG, Pawan K, Sundaram RM (2009) Validation of molecular markers linked to fertility restorer gene(s) for WA-CMS lines of rice. Euphytica 167:217–227
Shimono H, Okada M, Inoue M, Nakamura H, Kobayashi K, Hasegawa T (2010) Diurnal and seasonal variations in stomatal conductance of rice at elevated atmospheric CO2 under fully open-air conditions. Plant Cell Environ 33:322–331
Stewart CN Jr, Via LE (1993) A rapid CTAB DNA isolation technique useful for RAPD fingerprinting and other PCR applications. Bio Tech 14(5):748–750
Suresh PB, Srikanth B, Hemanth VK, Rao IS, Vemireddy LR, Dharika N, Sundaram RM, Ramesha MS, Rao KRSS, Viraktamath BC, Neeraja CN (2012) Fine mapping of Rf3 and Rf4 fertility restorer loci of WA-CMS of rice (Oryza sativa L.) and validation of the developed marker system for identification of restorer line. Euphytica 187:421–435
Virmani SS (1987) Hybrid rice breeding. Cytogenetic relationship between two cytoplasmic male sterile lines. Int Rice Res Newsl 12:14
Virmani SS, Aquino RC, Khush GS (1982) Heterosis breeding in rice (Oryza sativa L.). Theor Appl Genet 63:373–380
Xie F, He Z, Esguerra MQ, Qiu F, Ramanathan V (2014) Determination of heterotic groups for tropical indica hybrid rice germplasm. Theor Appl Genet 127:407–417
Yan W, Kang MS (2003) GGE biplot analysis: a graphical tool for breeders, geneticists and agronomists, 1st edn. CRC Press LLC., Boca Raton, p 271
Yao FY, Xu CG, Yu SB, Li JX, Gao YJ, Li XH, Zhang Q (1997) Mapping and Genetic analysis of two fertility restorer loci in the wild abortive cytoplasmic male sterility system of rice (Oryza sativa L.). Euphytica 98:183
Yuan LP, Virmani SS (1988) Status of hybrid rice research and development. In: Hybrid rice. International Rice Research Institute, Manila, Philippines, pp 7–24
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, A., Bhowmick, P.K., Singh, V.J. et al. Marker-assisted identification of restorer gene(s) in iso-cytoplasmic restorer lines of WA cytoplasm in rice and assessment of their fertility restoration potential across environments. Physiol Mol Biol Plants 23, 891–909 (2017). https://doi.org/10.1007/s12298-017-0464-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-017-0464-5