Abstract
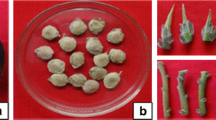
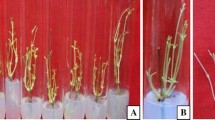
A micropropagation system for Bauhinia racemosa Lam. was developed involving axillary shoot proliferation and ex vitro rooting using nodal explants obtained from mature tree. MS medium with 3.0 mg l−1 BA (6-benzyladenine) was optimum for shoot bud induction. For shoot multiplication, mother explants were transferred repeatedly on medium containing low concentration of BA (0.75 mg l−1). Number of shoots was increased up to two passages and decreased thereafter. Shoot multiplication was further enhanced on MS medium containing 0.25 mg l−1 each of BA and Kin (Kinetin) with 0.1 mg l−1 of NAA (α-naphthalene acetic acid). Addition of 0.004 mg l−1 TDZ (thidiazuron) increased the rate of shoot multiplication and 21.81 ± 1.26 shoots per culture vessel were obtained. In vitro regenerated shoots were rooted under ex vitro conditions treated with 400 mg l−1 IBA (indole-3-butyric acid) for 7 min on sterile soilrite. After successful hardening in greenhouse, ex vitro rooted plants were transferred to the field conditions with ≈85% of survival rate. Micromorphological changes were observed on leaf surface i.e. development of vein density and trichomes and stomatal appearance, when plants were subjected to environmental conditions. This is the first report on in vitro regeneration of B. racemosa from mature tree.



Similar content being viewed by others
Abbreviations
- BA:
-
6-Benzyladenine
- IBA:
-
Indole-3-butyric acid
- Kin:
-
Kinetin (N6-furfuryladenine)
- MS:
-
Murashige and Skoog (1962) medium
- NAA:
-
α-Naphthalene acetic acid
- NOA:
-
2-Naphthoxy acetic acid
- PFD:
-
Photon flux density
- PGRs:
-
Plant growth regulators
- RH:
-
Relative humidity
- TDZ:
-
Thidiazuron
References
Anjaneyulu ASR, Reddy AVR, Reddy DSK, Ward RS, Adhikesavalu D, Cameron TS (1984) Pacharin: a new dibenzo(2,3-6,7)oxepin derivative from Bauhinia racemosa lamk. Tetrahedron 40:4245–4252
Anjaneyulu ASR, Reddy AVR, Reddy DSK, Cameron TS, Roe SP (1986) Racemosol: a novel tetracyclic phenol from Bauhinia racemosa lamk. Tetrahedron 42:2417–2420
Arya V, Shekhawat NS, Singh RP (2003) Micropropagation of Leptadenia reticulata—a medicinal plant. In Vitro Cell Dev Biol Plant 39:180–185
Asthana P, Jaiswal VS, Jaiswal U (2011) Micropropagation of Sapindus trifoliatus L. and assessment of genetic fidelity of micropropagated plants using RAPD analysis. Acta Physiol Plant 33:1821–1822
Bandhopadhyay S (1997) Notes on the distribution of Bauhinia racemosa Lam. (Leguminosae: caesalpinioideae) in India. J Econ Tax Bot 21:662
Benmahioul B, Dorion N, Kaid-Harche M, Daguin F (2012) Micropropagation and ex vitro rooting of Pistachio (Pistacia vera L.). Plant Cell Tissue Organ Cult 41:71–73
Borkowska B (2001) Morphological and physiological characteristics of micropropagated strawberry plants rooted in vitro or ex vitro. Sci Hortic 89:195–206
Goyal P, Kachhawa S, Kothari SL (2012) Micropropagation of Pithecellobium dulce (Roxb.) Benth-a multipurpose leguminous tree and assessment of genetic fidelity of micropropagated plants using molecular markers. Physiol Mol Biol Plants 18:169–176
Gupta M, Mazumdar UK, Kumar RS, Kumar TS (2004a) Antitumor activity and antioxidant role of Bauhinia racemosa against Ehrlich ascites carcinoma in Swiss albino mice. Acta Pharmacol Sin 25:1070–1076
Gupta M, Mazumdar UK, Kumar TS, Gomathi P, Kumar RS (2004b) Antioxidant and hepatoprotective effects of Bauhinia racemosa against paracetamol and carbon tetrachloride induced liver damage in rats. Iran J Pharmacol Ther 3:12–20
Huetteman CA, Preece JE (1993) Thidiazuron: a potent cytokinin for woody plant tissue culture. Plant Cell Tissue Organ Cult 33:105–119
Kevers C, Franck T, Strasser RJ, Dommes J, Gaspar T (2004) Hyperhydricity of micropropagated shoots; a typically stress induced change of physiological state. Plant Cell Tissue Organ Cult 77:181–191
Khan D, Zaki MJ, Anis M (2015) Seedling characteristics of jhinjera (Bauhinia racemosa Lamk.). Int J Biol Biotech 12:143–154
Khateeb WA, Bahar E, Lahham J, Schroeder D, Hussein E (2013) Regeneration and assessment of genetic fidelity of the endangered tree Moringa peregrina (Forsk.) Fiori using inter simple sequence repeat (ISSR). Physiol Mol Biol Plant 19:157–164
Klerk GJD (2002) Rooting of microcuttings: theory and practice. In Vitro Cell Dev Biol Plant 38:415–422
Lodha D, Patel A, Shekhawat NS (2015) A high-frequency in vitro multiplication, micromorphological studies and ex vitro rooting of Cadaba fruticosa (L.) Druce (Bahuguni): a multipurpose endangered medicinal shrub. Physiol Mol Biol Plants 21:407–415
Ludwig-Muller J (2000) Indole-3-butyric acid in plant growth and development. Plant Growth Regul 32:219–230
Malik SK, Chaudhury R, Kalia RK (2005) Rapid in vitro multiplication and conservation of Garcinia indica: a tropical medicinal tree species. Sci Hortic 106:539–553
Montenius O (1987) In vitro meristem culture of juvenile and mature Sequoiandendron giagantium. Tree Physiol 3:265–272
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Murthy BNS, Murch SJ, Saxena PK (1998) Thidiazuron: a potent regulator of in vitro plant morphogenesis. In Vitro Cell Dev Biol Plant 34:267–275
Phulwaria M, Rai MK, Harish Gupta AK, Ram K, Shekhawat NS (2012) An improved micropropagation of Terminalia bellirica from nodal explants of mature tree. Acta Physiol Plant 34:299–305
Pospisilova J, Ticha I, Kadleck P, Haisel D, Plzakova S (1999) Acclimatization of micropropagated plants to ex vitro conditions. Biol Plant 42:481–497
Pospisilova J, Synkova H, Rulcova J (2000) Cytokinins and water stress. Biol Plant 43:321–328
Prabhakar P, Ghandhidasan R, Raman PV, Krishnasamy NR, Nanduri S (1994) De-o-methylracemosol: a tetracyclic 2,2-dimethylchroman from the roots of Bauhinia racemosa. Phytochemistry 36:817–818
Prasad P, Nautiyal AR (1996) Physiology of germination in Bauhinia: involvement of seed coat in inhibition of germination in B. racemosa Lam. seeds. Seed Sci Technol 24:305–308
Preece JE, Shutter EG (1991) Acclimatization of micropropagated plants to the greenhouse and field. In: Debergh PC, Zimmerman RH (eds) Micropropagation: technology and application. Kluwer Academic Publishers, Dordrecht, pp 71–94
Prusty KB, Rao JV, Subudhi SK, Reddy PA, Kumar JR (2012) Anti hyperglycemic activity of extracts of leaves of Bauhinia Racemosa Lamk (family-Caesalpiniaceae) on normal and alloxan induced diabetic rats. Int J Pharm Res Allied Sci 1:94–99
Rai MK, Asthana P, Jaiswal VS, Jaiswal U (2010) Biotechnological advances in guava (Psidium guajava L.): recent developments and prospects for further research. Trees Struct Funct 24:1–12
Rajanna LN, Sharanabasappa G, Seetharam YN, Aravind B, Mallikharjuna PB (2011) In vitro regeneration of cotyledonary node explant of Bauhinia racemosa. Bot Res Intl 4:75–80
Rashed K, Luo M, Zhang L, Zheng Y (2013) Anti-HIV-1 potential of Bauhinia racemosa Lam. (caesalpiniaceae) and phytochemical profile. Topcls J Herb Med 2:95–102
Rathore TS, Deora NS, Shekhawat NS (1992) Cloning of Maytenus emarginata (Willd.) Ding Hou—a tree of the Indian Desert, through tissue culture. Plant Cell Rep 11:449–451
Ravindranath NH, Bhat DM, Swamy VS (2003) Nursery manual for forest tree species. University Press, Hyderabad
Shekhawat MS, Manokari M (2016a) Optimization of in vitro and ex vitro regeneration and micromorphological studies in Basella alba L. Physiol Mol Biol Plants 22:605–612
Shekhawat MS, Manokari M (2016b) In vitro propagation, micromorphological studies and ex vitro rooting of cannon ball tree (Couroupita guianensis aubl.): a multipurpose threatened species. Physiol Mol Biol Plants 22:131–142
Shekhawat MS, Shekhawat NS (2011) Micropropagation of Arnebia hispidissima (Lehm) DC. and production of alkannin from callus and cell suspension culture. Acta Physiol Plant 33:1445–1450
Shekhawat NS, Rathore TS, Singh RP, Deora NS, Rao SR (1993) Factors affecting in vitro cloning of Prosopis cineraria. Plant Growth Regul 12:273–280
Singh V, Pandey RP (1998) Ethnobotany of Rajasthan. Scientific Publisher, Jodhpur
Tripathi M, Kumari N (2010) Micropropagation of a tropical fruit tree Spondias mangifera Willd. through direct organogenesis. Acta Physiol Plant 32:1011–1015
Valverde-Cerdas L, Dufour M, Villalobos VM (1997) In vitro propagation of Pithecellobium saman (Rain tree). In Vitro Cell Dev Biol Plant 33:8–42
Vengadesan G, Pijut PM (2009) In vitro propagation of northern red oak (Quercus rubra L.). In Vitro Cell Dev Biol Plant 45:474–482
Vibha JB, Shekhawat NS, Mehandru P, Dinesh R (2014) Rapid multiplication of Dalbergia sissoo Roxb.: a timber yielding tree legume through axillary shoot proliferation and ex vitro rooting. Physiol Mol Biol Plant 20:81–87
Yan H, Liang C, Yang L, Li Y (2010) In vitro and ex vitro rooting of Siratia grosvenori a traditional medicine plant. Acta Physiol Plant 32:115–120
Zhang A, Wang H, Shao Q, Xu M, Zhang W, Li M (2015) Large scale in vitro propagation of Anoectochilus roxburghii for commercial application: pharmaceutically important and ornamental plant. Ind Crop Prod 70:158–162
Acknowledgement
Udit Sharma gratefully acknowledges to University Grants Commission (UGC), New Delhi for the financial assistance in the form of Junior Research Fellowship (JRF). Vinod Kataria is thankful to the UGC, New Delhi for providing assistance in the form of CAS (Centre of Advance Study) to the Department of Botany, Jai Narain Vyas University, Jodhpur.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Sharma, U., Kataria, V. & Shekhawat, N.S. In vitro propagation, ex vitro rooting and leaf micromorphology of Bauhinia racemosa Lam.: a leguminous tree with medicinal values. Physiol Mol Biol Plants 23, 969–977 (2017). https://doi.org/10.1007/s12298-017-0459-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-017-0459-2