Abstract
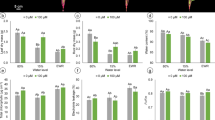
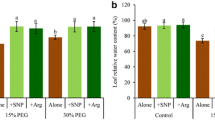
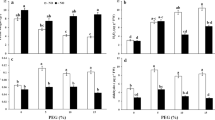
Drought stress is one of the most intensively studied and widespread constraints, and nitric oxide (NO) is a key signaling molecule involved in the mediation of abiotic stresses in plants. We demonstrated that a sprayed solution of NO from donor sodium nitroprusside increased drought stress tolerance responses in both sensitive (Phaseolus vulgaris) and tolerant (Vigna unguiculata) beans. In intact plants subjected to halting irrigation, NO increased the leaf relative water content and stomatal conductance in both species. After cutting leaf discs and washing them, NO induced increased electrolyte leakage, which was more evident in the tolerant species. These leaf discs were then subjected to different water deficits, simulating moderate and severe drought stress conditions through polyethylene glycol solutions. NO supplied at moderate drought stress revealed a reduced membrane injury index in sensitive species. In hydrated discs and at this level of water deficit, NO increased the electron transport rate in both species, and a reduction of these rates was observed at severe stress levels. Taken together, it can be shown that NO has an effective role in ameliorating drought stress effects, activating tolerance responses at moderate water deficit levels and in both bean species which present differential drought tolerance.





Similar content being viewed by others
References
Bajji M, Kinet JM, Lutts S (2001) The use of the electrolyte leakage method for assessing cell membrane stability as a water stress tolerance test in durum wheat. Plant Growth Reg 00:01–10
Beligni MV, Lamattina L (1999a) Is nitric oxide toxic or protective? Trends Plant Sci 4:299–300
Beligni MV, Lamattina L (1999b) Nitric oxide counteracts cytotoxic processes mediated by reactive oxygen species in plant tissues. Planta 208:337–344
Beligni MV, Lamattina L (2001) Nitric oxide in plants: the history is just beginning. Plant Cell Environ 24:267–278
Berkels R, Suerhoff S, Roesen R, Klaus W (2000) Nitric oxide causes a cGMP-independent intracellular calcium rise in porcine endothelial cells-a paradox? Microvasc Res 59:38–44
Borochov-Neori H, Borochov A (1991) Response of melon plants to salt. 1. Growth, morphology and root membrane properties. J Plant Physiol 139:100–105
Cruz de Carvalho MH, Laffray D, Louguet P (1998) Comparison of the physiological responses of Phaseolus vulgaris and Vigna unguiculata cultivars when submitted to drought conditions. Environ Exp Bot 40:197–207
Faria AP, Lemos-Filho JP, Modolo LV, França MGC (2013) Electrolyte leakage and chlorophyll fluorescence among castor bean cultivars under induced water deficit. Acta Physiol Plant 35:119–128
Feng Y, Li X, Boersma L (2006) Roles of osmotic adjustment and turgor potential in leaf area expansion. Physiol Plant 90:1–8
França MGC, Pham-Thi AT, Pimentel C, Rossiello ROP, Zuily-Fodil Y, Laffray D (2000) Differences in growth and water relations among Phaseolus vulgaris cultivars in response to induced drought stress. Environ Exp Bot 43:227–237
França MGC, Zimmer-Prados LM, Lemos-Filho JP, Ranieri BD, Vale FHA (2012) Morphophysiological diferences in leaves of Lavoisiera campos-portoana (Melastomataceae) enhance higher drought tolerance in water shortage events. J Plant Res 125:85–92
Garcia-Mata G, Lamattina L (2001) Nitric oxide induces stomatal closure and enhances the adaptive plant response against drought stress. Plant Physiol 126:1196–1204
Garcia-Mata C, Gay R, Sokolovski S, Hills A, Lamattina L, Blatt MR (2003) Nitric oxide regulates K+ and Cl− channels in guard cells through a subset of abscisic acid-evoked signaling pathways. Proc Natl Acad Sci 100:11116–11121
Genty B, Briantais JM, Vieira da Silva JBV (1987) Effects of drought on primary photosynthetic processes of cotton leaves. Plant Physiol 83:360–364
Hancock JT (2012) NO synthase? generation of nitric oxide in plant. Period Biol 114:19–24
Hsiao TC, Acevedo E (1974) Plant responses to water deficits, water-use efficiency, and drought resistance. Agric Meteorol 14:59–84
Huang AX, She XP, Zhang YY, Zhao JL (2013) Cytosolic acidification precedes nitric oxide removal during inhibition of ABA-induced stomatal closure by fusicoccin. Rus J Plant Physiol 60:60–68
Iturbe-Ormaetxe I, Escuredo PR, Arrese-Igor C, Becana M (1998) Oxidative damage in pea plants exposed to water deficit or paraquat. Plant Physiol 116:173–181
Lawson T, Blatt M (2014) Stomatal size, speed and responsiveness impact on photosynthesis and water use efficiency. Plant Physiol 164:1556–1570
Lazalt AM, Beligni VM, Lamattina L (1997) Nitric oxide preserves the level of chlorophyll in potato leaves infected by Phytophthora infestans. Eur J Plant Pathol 103:643–651
Leckie CP, McAinsh MR, Allen GJ, Sanders D, Hetherington AM (1998) Abscisic acid-induced stomatal closure mediated by cyclic ADP-ribose. Proc Natl Acad Sci U S A 95:15837–15842
Loggini B, Scartazza A, Brugnoli E, Navari-Izzo F (1999) Antioxidant defense system, pigment composition, and photosynthetic efficiency in two wheat cultivars subjected to drought. Plant Physiol 119:1091–1100
Magalhaes JR, Monte DC, Durzan D (2000) Nitric oxide and ethylene emission in Arabidopsis thaliana. Physiol Mol Biol Plants 6:117–127
Marciano DPRO, Ramos FT, Alvim MN, Magalhaes JR, França MGC (2010) Nitric oxide reduces the stress effects of aluminum on the process of germination and early root growth of rice. J Plant Nut Soil Sci 173:885–891
Money NP (1989) Osmotic pressure of aqueous polyethylene glycols. Relationship between molecular weight and vapor pressure deficit. Plant Physiol 91:766–769
Mott KA, Buckley TN (2000) Patchy stomatal conductance: emergent collective behaviour of stomata. Trends Plant Sci 5:258–262
Mur LAJ, Mandon J, Persijn S, Cristescu SM, Moshkov IE, Novikova GV, Hall MA, Harren FJM, Hebelstrup KH, Gupta KJ (2012) Nitric oxide in plants: an assessment of the current state of knowledge. AoB Plants 5:1–17
Neill S, Barros R, Bright J, Desikan R, Hancock J, Harrison J, Morris P, Ribeiro D, Wilson Y (2008) Nitric oxide, stomatal closure, and abiotic stress. J Exp Bot 59:165–176
Nilsen ET, Orcutt DM (1996) The physiology of plants under stress: abiotic factors. Wiley, New York
Okamoto M, Tanaka Y, Abrams SR, Kamiya Y, Seki M, Nambara E (2009) High humidity induces abscisic acid 8’-hydroxylase in stomata and vasculature to regulate local and systemic abscisic acid responses in arabidopsis. Plant Physiol 149:825–834
Ribeiro DM, Desikan R, Bright J, Confraria A, Harrison J, Hancock JT, Barros RS, Neill SJ, Wilson ID (2009) Differential requirement for NO during ABA-induced stomatal closure in turgid and wilted leaves. Plant Cell Environ 32:46–57
Sakihama Y, Murakami S, Yamasaki H (2003) Involvement of nitric oxide in the mechanism for stomatal opening in Vicia faba leaves. Biol Plant 46:117–119
Volk T, Mading K, Hensel M, Kox WJ (1997) Nitric oxide induces transient Ca2+ changes in endothelial cells independent of cGMP. J Cell Physiol 172:296–305
Wink DA, Hanbauer I, Krishna MC, DeGraff W, Gamson J, Mitchell JB (1993) Nitric oxide protects against cellular damage and cytotoxicity from reactive oxygen species. Proc Natl Acad Sci 90:9813–9817
Acknowledgments
The authors are grateful to Coordination for the Improvement of Higher Education Personnel (CAPES) and also the Vegetation Biology Post-Graduate Program (PPGBV) of the Federal University of Minas Gerais (UFMG), Brazil. Thanks also to Alistair Hayward for the translation and critical review of the English text.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zimmer-Prados, L.M., Moreira, A.S.F.P., Magalhaes, J.R. et al. Nitric oxide increases tolerance responses to moderate water deficit in leaves of Phaseolus vulgaris and Vigna unguiculata bean species. Physiol Mol Biol Plants 20, 295–301 (2014). https://doi.org/10.1007/s12298-014-0239-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-014-0239-1