Abstract
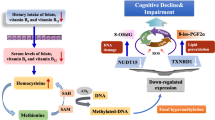
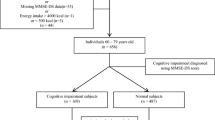
Epigenetic modifications have been implicated in the development of cognitive impairment. However, the relationship between DNA methylation levels and cognitive impairment can be greatly influenced by environmental factors some blood-based nutrition markers. The present study aims to understand the relationship between global DNA methylation levels and cognitive impairment independently and in light of micronutrient status among North Indian adults. This study was conducted among 614 individuals, aged 30 to 79 years from Palwal, Haryana. Cognitive impairment (CI) was assessed using Mini-Mental State Examination (MMSE). Folate, vitamin B12, and homocysteine levels were estimated using chemiluminescence technique. Estimation of global DNA methylation (5mC) levels was performed using the ELISA-based colorimetric technique. Appropriate comparison tests (based on normality distribution) were applied to compare the levels of global DNA methylation in different study groups. Logistic regression models were run to examine association between global DNA methylation and CI. Median 5mC levels of both mild and moderate/severe CI groups were significantly lower than that of the control group. Individuals in the 1st quartile of 5mC, with those in the 4th quartile as the reference, were at a significantly increased risk of both mild and moderate/severe CI. Vitamin B12, but not folate, appeared to mediate global DNA hypomethylation among CI cases. Cognitive impairment may be associated with Global DNA hypomethylation in the studied North Indian population. Vitamin B12 sufficiency may help improve the methylation levels among the cases of cognitive impairment. There is a need to develop population and context-specific epigenetic markers for cognitive impairment.


Similar content being viewed by others
Data Availabilty Statement
The data supporting the findings of this study will be made available by the authors on reasonable request.
References
Janoutová J, Serý O, Hosák L, Janout V. Is mild cognitive impairment a precursor of Alzheimer's disease? Short review. Cent Eur J Public Health. 2015;23(4):365. https://doi.org/10.21101/cejph.a4414
Wortmann M. Dementia: a global health priority-highlights from an ADI and World Health Organization report. Alzheimer’s Res Ther. 2012;4:1–3. https://doi.org/10.1186/alzrt143.
Pais R, Ruano L, Moreira C, Carvalho OP, Barros H. Prevalence and incidence of cognitive impairment in an elder Portuguese population (65–85 years old). BMC Geriatr. 2020;20(1):470.
Das SK, Bose P, Biswas A, Dutt A, Banerjee TK, Hazra A, et al. An epidemiologic study of mild cognitive impairment in Kolkata. India Neurol. 2007;68(23):2019–26. https://doi.org/10.1212/01.wnl.0000264424.76759.e6.
Sengupta P, Benjamin AI, Singh Y, Grover A. Prevalence and correlates of cognitive impairment in a north Indian elderly population. WHO South-East Asia J Public Health. 2014;3(2):135–43. https://doi.org/10.4103/2224-3151.206729.
Khullar S, Kaur G, Dhillon H, Sharma R, Mehta K, Singh M, et al. The prevalence and predictors of cognitive impairment in the type 2 diabetic population of Punjab. India Soc Health Diabetes. 2017;5(1):47–53. https://doi.org/10.4103/2321-0656.193996.
Kaur G, Gaur R, Yadav S, Saraswathy KN. Association of vitamin B12-mediated hyperhomocysteinemia and methylenetetrahydrofolate reductase (C677T) gene polymorphism with cognitive impairment: a population-based study from North India. Psychiatry Res. 2018;270:123–5. https://doi.org/10.1016/j.psychres.2018.08.103.
Singh N, Chaudhary V, Khate K, Bhattacharjee D, Longkumer I, Kataria S, et al. Cognitive impairment viz-a-viz genetic and biochemical variations in the one-carbon metabolic pathway: A population-based study from North India. Gene Rep. 2023;30: 101720. https://doi.org/10.1016/j.genrep.2022.101720.
Khate K, Chaudhary V, Longkumer I, Saraswathy KN, Devi NK. Gender-specific association of blood lipids and reproductive trajectory with cognitive impairment: A community-based cross-sectional study from India. Front Psychol. 2023;14. https://doi.org/10.3389/fpsyg.2023.1107152
Harman MF, Martín MG. Epigenetic mechanisms related to cognitive decline during aging. J Neurosci Res. 2020;98(2):234–46. https://doi.org/10.1002/jnr.24436.
Chouliaras L, Pishva E, Haapakoski R, Zsoldos E, Mahmood A, Filippini N, et al. Peripheral DNA methylation, cognitive decline and brain aging: pilot findings from the Whitehall II imaging study. Epigenomics. 2018;10(5):585–95. https://doi.org/10.2217/epi-2017-0132. ([PubMed PMID: 29723050]).
Sugden K, Caspi A, Elliott ML, Bourassa KJ, Chamarti K, Corcoran DL, et al. Association of pace of aging measured by blood-based DNA methylation with age-related cognitive impairment and dementia. Neurology. 2022 27;99(13):e1402–13. https://doi.org/10.1212/WNL.0000000000200898. [PubMed PMID: 34260763]
Smith AD, Refsum H. Homocysteine, B vitamins, and cognitive impairment. Annu Rev Nutr. 2016;17(36):211–39. https://doi.org/10.1146/annurev-nutr-071715-050947. ([PubMed PMID: 27146033]).
Chaudhary V, Bhattacharjee D, Devi NK, Saraswathy KN. Global DNA Methylation Levels Viz-a-Viz Genetic and Biochemical Variations in One Carbon Metabolic Pathway: An Exploratory Study from North India. Biochem Genet. 2024 Feb 14. https://doi.org/10.1007/s10528-023-10659-4. Epub ahead of print. PMID: 38356009
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–98 ([PubMed PMID: 1202204]).
Miller S, Dykes D, Polesky H. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988;16(3):1215.
Sukla KK, Raman R. Association of MTHFR and RFC1 gene polymorphism with hyperhomocysteinemia and its modulation by vitamin B12 and folic acid in an Indian population. Eur J Clin Nutr. 2012;66(1):111–8. https://doi.org/10.1038/ejcn.2011.152. (Epub 2011 Aug 31 PMID: 21878957).
Li H, Wu TT, Tang L, Liu Q, Mao XZ, Xu JM, et al. Association of global DNA hypomethylation with post-operative cognitive dysfunction in elderly patients undergoing hip surgery. Acta Anaesth Scand. 2020;64(3):354–60. https://doi.org/10.1111/aas.13502.
Yang X, Yuan Y, Lu X, Yang J, Wang L, Song J, et al. The relationship between cognitive impairment and global DNA methylation decrease among aluminum potroom workers. J Occup Environ Med. 2015;57(7):713–7. https://doi.org/10.1097/JOM.0000000000000474. ([PubMed PMID: 26110289]).
Schiepers OJ, Van Boxtel MP, de Groot RH, Jolles J, Kok FJ, Verhoef P, et al. DNA methylation and cognitive functioning in healthy older adults. Br J Nutr. 2012;107(5):744–8. https://doi.org/10.1017/S0007114511003576. ([PubMed PMID: 21967862]).
Gomes MV, Toffoli LV, Arruda DW, Soldera LM, Pelosi GG, Neves-Souza RD, et al. Age-related changes in the global DNA methylation profile of leukocytes are linked to nutrition but are not associated with the MTHFR C677T genotype or to functional capacities. PLoS ONE. 2012;7(12): e52570. https://doi.org/10.1371/journal.pone.0052570. ([PubMed PMID: 23272203]).
Cui D, Xu X. DNA methyltransferases, DNA methylation, and age-associated cognitive function. Int J Mol Sci. 2018;19(5):1315. https://doi.org/10.3390/ijms19051315. ([PubMed PMID: 29710737]).
Kaeberlein M. Time for a new strategy in the war on Alzheimer’s disease. Public Policy Aging Rep. 2019;29(4):119–22. https://doi.org/10.1093/ppar/prz020. ([PubMed PMID: 32189970]).
Ma DK, Jang MH, Guo JU, Kitabatake Y, Chang ML, Pow-Anpongkul N, et al. Neuronal activity–induced Gadd45b promotes epigenetic DNA demethylation and adult neurogenesis. Science. 2009;323(5917):1074–7. https://doi.org/10.1126/science.1166859. ([PubMed PMID: 19229056]).
Lopez-Atalaya JP, Barco A. Can changes in histone acetylation contribute to memory formation? Trends Genet. 2014;30(12):529–39. https://doi.org/10.1016/j.tig.2014.09.003. ([PubMed PMID: 25306222]).
Kaushik A, Chaudhary V, Longkumer I, Saraswathy KN, Jain S. Sex-specific variations in global DNA methylation levels with age: a population-based exploratory study from North India. Front Genet. 2023;14. [PubMed PMID: To be added once available]
Orsitto G. Different components of nutritional status in older inpatients with cognitive impairment. J Nutr Health Aging. 2012;16:468–71. https://doi.org/10.1007/s12603-012-0024-1. ([PubMed PMID: 22689217]).
Scarmeas N, Anastasiou CA, Yannakoulia M. Nutrition and prevention of cognitive impairment. Lancet Neurol. 2018;17(11):1006–15. https://doi.org/10.1016/S1474-4422(18)30338-7. ([PubMed PMID: 30389355]).
Alosco ML, Spitznagel MB, Cohen R, Raz N, Sweet LH, Josephson R, et al. Decreased physical activity predicts cognitive dysfunction and reduced cerebral blood flow in heart failure. J Neurol Sci. 2014;339(1–2):169–75. https://doi.org/10.1016/j.jns.2014.02.008. ([PubMed PMID: 24461454]).
Nakakubo S, Makizako H, Doi T, Tsutsumimoto K, Lee S, Lee S, et al. Impact of poor sleep quality and physical inactivity on cognitive function in community‐dwelling older adults. Geriatr Gerontol Intl. 2017;17(11):1823–8. https://doi.org/10.1111/ggi.12973 [PubMed PMID: 28294469]
Matyas N, Aschenberger FK, Wagner G, Teufer B, Auer S, Gisinger C, et al. Continuing education for the prevention of mild cognitive impairment and Alzheimer’s-type dementia: a systematic review and overview of systematic reviews. BMJ Open. 2019;9(7): e027719. https://doi.org/10.1136/bmjopen-2018-027719. ([PubMed PMID: 31272929]).
Wang AY, Hu HY, Ou YN, Wang ZT, Ma YH, Tan L, et al. Socioeconomic status and risks of cognitive impairment and dementia: a systematic review and meta-analysis of 39 prospective studies. J Prev Alz Dis. 2023;10(1):83–94. https://doi.org/10.14283/jpad.2022.81 [PubMed PMID: To be added once available]
Maugeri A, Barchitta M. How dietary factors affect DNA methylation: lesson from epidemiological studies. Medicina. 2020;56(8):374. https://doi.org/10.3390/medicina56080374. ([PubMed PMID: 32722698]).
Cerutti J, Lussier AA, Zhu Y, Liu J, Dunn EC. Associations between indicators of socioeconomic position and DNA methylation: a scoping review. Cli Epigenetics. 2021;13:1–20. https://doi.org/10.1186/s13148-021-01189-0. ([PubMed PMID: 34945607]).
Van Dongen J, Bonder MJ, Dekkers KF, Nivard MG, van Iterson M, Willemsen G, et al. DNA methylation signatures of educational attainment. NPJ Sci Learn. 2018;3(1):7. https://doi.org/10.1038/s41539-018-0020-2. ([PubMed PMID: 29594135]).
Wang Z, Zhu W, Xing Y, Jia J, Tang Y. B vitamins and prevention of cognitive decline and incident dementia: a systematic review and meta-analysis. Nutr Rev. 2022;80(4):931–49. https://doi.org/10.1093/nutrit/nuab057. ([PubMed PMID: 35001561]).
Acknowledgements
The authors would like to acknowledge all the kind participants for their invaluable time and cooperation in the present study. We also thank the Department of Anthropology, University of Delhi for providing the required facilities to conduct the study.
Funding
The present study was supported by the Department of Science and Technology, Ministry of Science and Technology, Government of India (DST/CSRI/2018/162G) and Indian Council of Medical Research (45/01/2022-ANTHRO/BMS).
Author information
Authors and Affiliations
Contributions
KNS and NB supervised the project and data collection. KNS conceptualised the manuscript. DB and AK performed lab analyses for global DNA methylation. KK performed data analyses and wrote the first draft. VC, GKW, NKD and KNS critically revised the manuscript for intellectual content. All the authors approved the final version of the manuscript for publication.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no competing interests.
Human Ethics
The present study was conducted following the principles of the Declaration of Helsinki for biomedical studies involving human subjects. Moreover, the present study was approved by the Institutional Ethics Committee, Department of Anthropology, University of Delhi (Ref No. Anth/2018/2890/1/28-12-2018). Prior to recruitment and data collection, pre-informed voluntary written consent, transcribed in local language was obtained from each participant.
Consent for publication
All the authors have provided consent for the publication of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khate, K., Chaudhary, V., Bhattacharjee, D. et al. Global DNA Methylation and Cognitive Impairment: A Population-Based Study from Haryana, North India. Ind J Clin Biochem (2024). https://doi.org/10.1007/s12291-024-01205-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12291-024-01205-z