Abstract
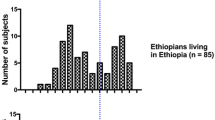
Activities of human hepatic drug metabolizing enzymes N-acetyl transferase (NATS) had earlier been recognized as a cause of inter-individual variation in the metabolism of drugs. Therefore acetylation of many drugs in human exhibit genetic polymorphism. The aim of the study was to investigate if acetylator status predispose diabetic mellitus patients more to the complications of renal disease, One hundred and twenty (120) diabetics consisting of (50) Type 1 (T1) and 70 Type 2 (T2) diabetes mellitus patients and 100 healthy individuals as controls were classified as slow or rapid acetylator using sulphamethazine (SMZ) as an in vivo probe. The percentage acetylation, recovery of SMZ, creatinine clearance and presence of urinary albumin were determined. A significant difference (P < 0.05) was observed in the percentage of SMZ acetylated between slow and rapid acetylators in control, T1 and T2 subjects. The ratios of slow to rapid acetylators for T1, T2 and control subjects were 1:4, 3:2 and 2:3 respectively. No significant differences were observed in the percentage of SMZ recovered in the urine of slow and rapid acetylators that are diabetics. The difference in creatinine clearance of slow and rapid acetylators in T1 and T2 were significant (P < 0.05). 29% out of 120 (24.2%) diabetics (T1 and T2) exhibited albuminuria out of which 25 (86.2%) had slow acetylator status. These findings suggest that slow acetylator status in diabetes mellitus could be a predisposing factor in the development of renal complications. This underscores the need for a rapid pharmacogenetic testing and therapeutic drug monitoring in such patients. However this inference could be further validated with a larger sample size.




Similar content being viewed by others
References
Shenfield GM. Genetic polymorphisms, drug metabolism and drug concentrations. Clin Biochem Rev. 2004;Nov:203–6.
Meyer VA. Pharmacogenetics and adverse drug reactions. Lancet. 2000;356:1667–71.
Stephanovic M, Topic E, Simundic AM. Novel SSCP method for molecular diagnostics of thiopurine S-methyltransferase (TPMP) deficiencies. Clin Chem Lab Med 2004;42(9):A94.
Hein DW. Acetylator genotype and arylamine-induced carcinogenesis. Biochim Biophys Acta. 1988;948:37–66.
Okumura K, Kita T, Chikazewa S, Komada F, Iwakawa S, Tanigawa Y. Genotyping of N-acetylator polymorphism and correlation with procainamide metabolism. Clin Pharmacol Ther. 1997;61:509–17.
Evans DA. Ethnic differences in reactions to drugs and xenobiotics, acetylation. Prog Clin Biol Res. 1986;214:209–42.
El Desoky ES, Abdel Salam YM, Salama RH, El AKkad MA, Atanasova S, Von Ahsen N, et al. NAT 2*5/*5 genotype (341T<C) is a potential risk factor for schistosomiasis-associated bladder cancer in Egyptians. Ther Drug Monit. 2005;27(3):297–304.
Woolhouse WM, Qureshi MM, Bastaki SMA, Patel M, Abdulrazzaq Y, Bayoumi RAL. Polymorphic N-acetyltransferase (NAT2) genotyping of Emiratis. Pharmacogenetics. 1997;7:73–82.
Ait Moussa L, Khassouani CE, Hue B, Jana M, Begaud B, Soulaymani R. Determination of the acetylator phenotype in Moroccan tuberculosis patients using isoniazid as metabolic probe. Crit Care Med. 2002;30:2107–14.
Cranswick N, Mulholland K. Isoniazid treatment of children: can genetics help guide treatment? Arch Dis Child. 2005;90:551–3.
Fawcett IW, Gammon PT. The determination of acetylator phenotype in a Nigerian population. Tubercle. 1975;5:199–201.
Eze LC, Obidoa AO. Acetylator of SMZ in a Nigerian population. Biochem Genet. 1978;16:1073–7.
Jeyakumar LH, French MR. Polymorphic acetylation of sulphamethazine in a Nigerian (Yoruba) population. Xenobiotica. 1981;11:319–21.
Delaney MP, Price CP, Lamb EJ. Kidney function and disease. In: Burtis CA, Ashwood ER, Burns DE, editors. Tiez fundamentals of clinical chemistry. 6th ed. St. Louis, Missouri: Saunders, Elsevier; 2008. p. 638–9.
Evans DA, White TA. Human acetylation polymorphism. J Lab Clin Med. 1964;63:394–403.
Tucker G. Pharmacogenetics—expectations and reality. Editorial. Br Med J. 2004;329:4–5.
Bunn HF. Evaluation of glycosylated haemoglobin in diabetic patients. Diabetes. 1981;30:613–7.
Evans WE. Pharmacogenetics of thiopurine S-methyl transferase and thiopurine. Ther Drug Monit. 2004;26:186–9.
McLaren EH, Burden AC, Moorhead PJ. Acetylator phenotype in diabetic neuropathy. Br Med J. 1977;2:291–3.
Summer Scales JE, Josephy PD. Human acetyl-COA: arylamine N-acetyl transferase variants generated random mutagenesis. Mol Pharmacol. 2004;65:220–6.
Shenfield GM, Gross AS. The cytochrome p450 system and adverse drug reactions. Adv Drug React Bull. 1999;194:739–42.
Chang JB, Anderson RC, Cruz PD. Steven-Johnson syndrome associated with glipizide therapy. Dermatitis. 2006;17(1):36–8.
Schwarz UI. Clinical relevance of genetic polymorphisms in the human CYP 2C9 gene. Eur J Clin Investig. 2003;33(Suppl 2):23–30.
Grant DM, Morike K, Eichelbaum M, Mayer UA. Acetylation, pharmacogenetics; the slow acetylators phenotype is caused by decreased or absent arylamine N-acetyl transferase in human liver. J Clin Investig. 1990;85(3):968–72.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Banjoko, S.O., Akinlade, K.S. Acetylation Pharmacogenetics and Renal Function in Diabetes Mellitus Patients. Indian J Clin Biochem 25, 289–294 (2010). https://doi.org/10.1007/s12291-010-0055-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-010-0055-5