Abstract
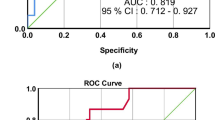
This study was conducted to evaluate whether microalbuminuria on admission and after 24 hrs of admission to intensive care unit (ICU) predicts outcome as well as the Acute Physiology and Chronic Health Evaluation (APACHE) II severity illness score, the current accepted method of doing so. The study was carried out in a 20 bed mixed medical-surgical ICU of a tertiary care hospital. Of 525 consecutive adult patients with ICU stay of more than 24 hrs, 238 were included for the study. Patients with pregnancy, menstruation, anuria, macroscopic hematuria, urinary tract infection, marked proteinuria due to renal and post-renal structural diseases, were excluded. Spot urine samples were collected on admission to ICU and 24 hrs thereafter. Urine albumincreatinine ratio (ACR) was measured on ICU admission (ACR1) and after 24 hrs (ACR2) and expressed in mg/g. Patient demographics were noted on admission. For disease severity scoring, APACHE II scores were calculated. Each patient was followed up throughout their ICU stay for a maximum of 28 days and the following outcome data were obtained: ICU length of stay and ICU mortality. Of the 238 patients, 196 survived while 42 patients died in the ICU. Non-survivors had a significantly higher median ACR2 [162.7 mg/g (IQR 69.5–344.3)] in comparison to the survivors who had a median ACR2 = 54.4 mg/g (IQR 19.0–129.1) (P< 0.0001). The median ACR1 [161.0 mg/g (IQR 29.0–369.3)] of non-survivors was higher than the median ACR1 [80.4 mg/g (IQR 35.1–167.6)] of survivors but failed to reach statistical significance (P= 0.0948). In a receiver operating characteristic curve (ROC) analysis, ACR2 emerged as the best indicator of mortality [(area under curve (AUC) of ACR2 = 0.71 > AUC (ACR1) =0.58 > AUC (ΔACR) =0.55] similar to the currently used APACHE II scores (AUC = 0.78) (P=0.3). At a cutoff of 101 mg/g, ACR2 had a sensitivity of 69%, specificity of 67%, positive predictive value of 31% and a negative predictive value of 91% for predicting mortality in the critically ill patients. Absence of significant microalbuminuria at 24 hrs of ICU admission may help to predict survival in the ICU.
Similar content being viewed by others
References
Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: A severity of disease classification system. Crit Care Med 1985; 13: 818–829.
Lemeshow S, Teres D, Avrunin JS, Pastides H. A comparision of methods to predict mortality of intensive care unit patients. Crit Care Med 1987; 8: 715–722.
Davies MG, Hagen PO. Systemic inflammatory response syndrome. Br J Surg 1997; 84: 920–935.
Aird William C. The role of the endothelium in severe sepsis and multiple organ dysfunction syndrome. Blood 2003; 101(10): 3765–3777.
Fishel RS, Are C, Barbul A. Vessel injury and capillary leak. Crit Care Med 2003; 31: S502–S511.
Jensen JS, Borch-Johnsen K, Jensen G, Feldt-Rasmussen B. Microalbuminuria reflects generalized transvascular albumin leakiness in clinically healthy subjects. Clin Sci 1995; 88: 629–633.
Gosling P. Microalbuminuria: a marker of systemic disease. Br J Hosp Med 1995; 54: 285–290.
Shearman CP, Gosling P, Walker KJ. Is low proteinuria an early predictor of severity of acute pancreatitis? J Clin Pathol 1989; 42: 1132–1135.
Gosling P, Hughes EA, Reynolds TM, Fox JP. Microalbuminuria is an early response following acute myocardial infarction. Eur Heart J 1991; 12: 508–513.
Gosling P, Sanghera K, Dickson G. Generalized vascular permeability and pulmonary function in patients following serious trauma. J Trauma 1994; 36: 477–481.
Roine I. Microalbuminuria: An index of severity in childhood meningitis. Pediatr Infect Dis J 1993; 12: 584–588.
Berton G, Citro T, Palmieri R, Petucco S, De Toni R, Palatini P. Albumin excretion rate increases during acute myocardial infarction and strongly predicts early mortality. Circulation 1997; 96: 3338–3345.
Yew WS, Pal SK. Correlation of microalbuminuria and outcome in patients with extensive burns. Br J Anaesth 2006; 97(4): 499–502.
De Gaudio AR, Spina R, Di Filippo A, Feri M. Glomerular permeability and trauma: A correlation between microalbuminuria and Injury Severity Score. Crit Care Med 1999; 27: 2105–2108.
Szakmany T, Molnar Z. Increased glomerular permeability and pulmonary dysfunction following major surgery: Correlation of microalbuminuria and PaO2/FiO2 ratio. Acta Anaesthesiol Scand 2004; 48: 704–710.
Evans G, Greaves I. Microalbuminuria as a predictor of outcome: Shows promise but large prospective trials are needed. BMJ 1998; 318: 207–208.
MacKinnon KL, Molnar Z, Lowe D, Watson ID, Shearer E. Use of microalbuminuria as a predictor of outcome in critically ill patients. Br J Anaesth 2000; 84: 239–241.
Abid O, Sun Q, Sugimoto K, Mercan D, Vincent JL. Predictive value of microalbuminuria in medical ICU patients: Results of a pilot study. Chest 2001; 120: 1984–1988.
Gosling P, Brudney S, McGrath L, Riseboro S, Manji M. Mortality prediction at admission to intensive care: A comparison of microalbuminuria with acute physiology scores after 24 hours. Crit Care Med 2003; 31: 98–103.
Thorevska N, Sabahi R, Upadya A, Manthous C, Amoateng-Adjepong Y. Microalbuminuria in critically ill medical patients: Prevalence, predictors, and prognostic significance. Crit Care Med 2003; 31: 1075–1081.
Gosling P, Czyz J, Nightingale P, Manji M. Microalbuminuria in the intensive care unit: Clinical correlates and association with outcomes in 431 patients. Crit Care Med 2006; 34(8): 2158–2166.
Gopal S, Carr C, Nelson P. Does microalbuminuria predict illness severity in critically ill patients on the intensive care unit? A systematic review. Crit Care Med 2006; 34(6): 1805–1810.
Terao Y, Takada M, Fujinaga A, Fukusaki M, Sumikawa K. Microalbuminuria is a prognostic predictor in aneurysmal subarachnoid hemorrhage. Intensive Care Med 2007; 33(6): 1000–1006.
Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001; 345: 1368–1377.
Bossuyt PM, Reitsma JE, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM, et al. Towards Complete and Accurate Reporting of Studies of Diagnostic Accuracy: The STARD Initiative. Clin Chem 2003; 49(1): 1–18.
Members of the American College of Chest Physicians, Society of Critical Care Medicine. American College of Chest Physicians-Society of Critical Care Medicine Consensus Conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med 1992; 20: 864–875.
Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, et al. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 2003; 31(4): 1250–1256.
Dziedzic T, Slowik A, Szczudlik A. Urine albumin excretion in acute ischaemic stroke is related to serum interleukin-6. Clin Chem Lab Med 2004; 42(2): 182–185.
Bakker AJ. Detection of microalbuminuria. Receiver operating characteristic curve analysis favours microalbuminuria-to-creatinine ratio over albumin concentration. Diabetes Care 1999; 22: 307–313.
Molnar Z, Szakmany T, Heigl P. Microalbuminuria does not reflect increased systemic capillary permeability in septic shock. Intensive Care Med 2003; 29: 391–395.
Winocour PH, Marshall SM. Microalbuminuria: Biochemistry, Epidemiology, and Clinical Practice. Cambridge, England: Cambridge University Press; 1998.
Jones CA, Francis ME, Eberhardt MS, Chavers B, Coresh J, Engelgau M, et al. Microalbuminuria in the US population: Third National Health and Nutrition Examination Survey. Am J Kidney Dis 2002; 39(3): 445–459.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Basu, S., Chaudhuri, S., Bhattacharyya, M. et al. Microalbuminuria: An inexpensive, non invasive bedside tool to predict outcome in critically ill patients. Indian J Clin Biochem 25, 146–152 (2010). https://doi.org/10.1007/s12291-010-0027-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-010-0027-9