Abstract
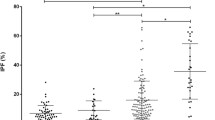
The occurrence of thrombotic microangiopathy (TMA) in pregnancy is an unfortunate emergency condition. Proper diagnosis is mandatory which requires the consideration of two overlapping diagnoses: severe preeclampsia/haemolysis, elevated liver enzymes, and low platelet syndrome (SPE/HELLP) and thrombotic thrombocytopenic purpura (TTP). The long turn-around times of ADAMTS13 testing precludes the timely distinction between the two conditions. We aimed at evaluating schistocyte counts and immature platelet fraction (IPF%), as both increase in TMAs, to discriminate between TTP and SPE/HELLP of pregnancy. IPF% was measured using Sysmex XE-2100 automated hematology analyzer, and schistocyte counts were estimated microscopically as per the International Council for Standardization in Hematology-Schistocyte Working Group guidelines. The study included 30 pregnant patients with SPE/HELLP, 13 pregnant patients with TTP, and 30 women with normal pregnancy. The discrimination between the two patient categories was based on clinical judgment and TTP cases were identified using the PLASMIC score. TTP patients had higher values of IPF% than SPE/HELLP [19.5% (16.9–27.1) vs 13% (9.5–23.25); p < 0.001]; similar results were revealed regarding schistocyte counts [6.5% (3.9–8.6) vs 2.1% (1.6–3.5); p < 0.001]. IPF% and schistocyte counts were able to discriminate between TMA patients and normal pregnant women, and between and SPE/HELLP and TTP patients. Moreover, the discriminatory function of each was improved when the two parameters were used in combination. IPF% analysis should be used in conjunction with manual schistocyte counting in TMA cases to distinguish TTP pregnant patients from patients having SPE/HELLP.

Similar content being viewed by others
References
Pourrat O, Coudroy R, Pierre F (2015) Differentiation between severe HELLP syndrome and thrombotic microangiopathy, thrombotic thrombocytopenic purpura and other imitators. Eur J Obstet Gynecol Reprod Biol 189:68–72. https://doi.org/10.1016/j.ejogrb.2015.03.017
Weinstein L (1982) Syndrome of hemolysis, elevated liver enzymes, and low platelet count: a severe consequence of hypertension in pregnancy. Am J Obstet Gynecol 142(2):159–167. https://doi.org/10.1016/j.ajog.2005.02.113
Schwartz J, Padmanabhan A, Aqui N, Balogun RA, Connelly-Smith L, Delaney M et al (2016) Guidelines on the use of therapeutic apheresis in clinical practice-evidence-based approach from the Writing Committee of the American Society for Apheresis: the seventh special issue. J Clin Apher 31(3):149–162. https://doi.org/10.1002/jca.21470
George JN, Nester CM, McIntosh JJ (2015) Syndromes of thrombotic microangiopathy associated with pregnancy. Hematol Am Soc Hematol Educ Program. 2015:644–648. https://doi.org/10.1182/asheducation-2015.1.644
Ramadan MK, Badrd DA, Hubeisha M, Itania S, Hijazic H, Mogharbile A (2018) HELLP syndrome, thrombotic thrombocytopenic purpura or both: appraising the complex association and proposing a stepwise practical plan for differential diagnosis. J Hematol 7(1):32–37. https://doi.org/10.14740/jh347w
Chandran R, Serra-Serra V, Redman CW (1992) Spontaneous resolution of pre-eclampsia related thrombocytopenia. Br J Obstet Gynaecol 99(11):887–890
Eser B, Guven M, Unal A, Coskun R, Altuntas F, Sungur M et al (2005) The role of plasma exchange in HELLP syndrome. Clin Appl Thromb Hemost 11(2):211–217
Keiser SD, Boyd KW, Rehberg JF, Elkins S, Owens MY, Sunesara I et al (2012) A high LDH to AST ratio helps to differentiate pregnancy-associated thrombotic thrombocytopenic purpura (TTP) from HELLP syndrome. J Matern Fetal Neonatal Med 25(7):1059–1063. https://doi.org/10.3109/14767058.2011.619603
Bendapudi PK, Hurwitz S, Fry A, Marques MB, Waldo SW, Li A et al (2017) Derivation and external validation of the PLASMIC score for rapid assessment of adults with thrombotic microangiopathies: a cohort study. Lancet Haematol 4(4):e157–e164. https://doi.org/10.1016/S2352-3026(17)30026-1
Burns ER, Lou Y, Pathak A (2004) Morphologic diagnosis of thrombotic thrombocytopenic purpura. Am J Hematol 75:18–21. https://doi.org/10.1002/ajh.10450
Lesesve JF, Salignac S, Lecompte T (2007) Laboratory measurement of schistocytes. Int J Lab Hematol 29(2):149–151. https://doi.org/10.1111/j.1751-553x.2006.00829.x
Zini G, D’Onofrio G, Briggs C, Erber W, Jou JM, Lee SH et al (2012) ICSH recommendations for identification, diagnostic value and quantification of schistocytes. Int J Lab Hematol 34:107–116. https://doi.org/10.1111/j.1751-553X.2011.01380.x
Harrison P, Robinson MSC, Mackie IJ, Machin SJ (1997) Reticulated platelets. Platelets 8(6):379–384. https://doi.org/10.1080/09537109777050
Salvagno GL, Montagnana M, Degan M, Marradi PL, Ricetti MM, Riolfi P et al (2006) Evaluation of platelet turnover by flow cytometry. Platelets 17(3):170–177. https://doi.org/10.1080/09537100500437851
Hong H, Xiao W, Stempak LM, Sandhaus LM, Maitta RW (2015) Absolute immature platelet count dynamics in diagnosing and monitoring the clinical course of thrombotic thrombocytopenic purpura. Transfusion 55(4):756–765. https://doi.org/10.1111/trf.12912
Moraes D, Munhoz TP, Pinheiro da Costa BE, Hentschke MR, Sontag F, Silveira Lucas L et al (2016) Immature platelet fraction in hypertensive pregnancy. Platelets 27(4):333–337. https://doi.org/10.3109/09537104.2015.1101060
American College of Obstetricians and Gynecologists (2013) Hypertension in pregnancy: report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet Gynecol 122:1122–1131. https://doi.org/10.1097/01.AOG.0000437382.03963.88
Magee LA, Pels A, Helewa M, Rey E, von Dadelszen P, SOGC Hypertension Guideline Committee (2014) Diagnosis, evaluation, and management of the hypertensive disorders of pregnancy: executive summary. J Obstet Gynaecol Can 36(5):416–438. https://doi.org/10.1016/s1701-2163(15)30533-8
Joly BS, Coppo P, Veyradier A (2017) Thrombotic thrombocytopenic purpura. Blood 129(21):2836–2846. https://doi.org/10.1182/blood-2016-10-709857
Ko YJ, Hur M, Kim H, Choi SG, Moon HW, Yun YM (2015) Reference interval for immature platelet fraction on Sysmex XN hematology analyser: a comparison study with Sysmex XE-2100. Clin Chem Lab Med 53:1091–1097. https://doi.org/10.1515/cclm-2014-0839
Jajosky R, Floyd M, Thompson T, Shikle J (2017) Validation of the PLASMIC score at a University Medical Center. Transfus Apher Sci 56(4):591–594. https://doi.org/10.1016/j.transci.2017.06.006
Seo A, Yuan D, Daniels S, Yuan S, Gallagher M, Wong E (2015) Reference intervals for immature platelet fraction and immature platelet count. Int J Lab Hematol 37:e12. https://doi.org/10.1111/ijlh.12237
Park SH, Park C, Lee B, Kim M, Han M, Cho Y et al (2016) Establishment of age- and gender-specific reference ranges for 36 routine and 57 cell population data items in a new automated blood cell analyser, Sysmex XN-2000. Ann Lab Med 36:244–249. https://doi.org/10.3343/alm.2016.36.3.244
Imperiali CE, Arbiol-Roca A, Sanchez-Navarro L, Dastis-Arias M, Lopez-Delgado JC, Cortes-Bosch A et al (2018) Reference interval for immature platelet fraction on Sysmex XN haematology analyser in adult population. Biochem Med (Zagreb) 28(1):010708. https://doi.org/10.11613/BM.2018.010708
Ratsch U, Kaiser T, Stepan H, Jank A (2017) Evaluation of bone marrow function with immature platelet fraction in normal pregnancy. Pregnancy Hypertens 10:70–73. https://doi.org/10.1016/j.preghy.2017.06.006
Everett TR, Garner SF, Lees CC, Goodall AH (2014) Immature platelet fraction analysis demonstrates a difference in thrombopoiesis between normotensive and preeclamptic pregnancies. Thromb Haemost 111:1177–1179. https://doi.org/10.1160/TH13-09-0746
Briggs C, Kunka S, Hart D, Oguni S, Machin SJ (2004) Assessment of an immature platelet fraction (IPF) in peripheral thrombocytopenia. Br J Haematol 126(1):93–99. https://doi.org/10.1111/j.1365-2141.2004.04987.x
Huh HJ, Chung JW, Chae SL (2013) Microscopic schistocyte determination according to International Council for Standardization in Hematology recommendation in various diseases. Int J Lab Hematol 35(5):542–547. https://doi.org/10.1111/ijlh.12059
Schapkaitz E, Mezgebe MH (2017) The clinical significance of schistocytes: a prospective evaluation of the international council for standardization in hematology schistocyte guidelines. Turk J Haematol 34(1):59–63. https://doi.org/10.4274/tjh.2016.0359
Lesesve J, Adssi HE, Watine JC, Oosterhuis WP, Régnier F (2013) Evaluation of ICSH schistocytes measurement guidelines in France. Int J Lab Hematol 35(6):601–607. https://doi.org/10.1111/ijlh.12092
Acknowledgements
We acknowledge the Intensive Care Unit (ICU) of Ain Shams Maternity Hospital for supporting patients’ data access. We also thank Prof Sameh Michel for his help in the statistical analysis.
Funding
This work was not supported by any funding and grant-awarding bodies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no declarations of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El-Gamal, R.A., Mekawy, M.A., Abd Elkader, A.M. et al. Combined Immature Platelet Fraction and Schistocyte Count to Differentiate Pregnancy-Associated Thrombotic Thrombocytopenic Purpura from Severe Preeclampsia/Haemolysis, Elevated Liver Enzymes, and Low Platelet Syndrome (SPE/HELLP). Indian J Hematol Blood Transfus 36, 316–323 (2020). https://doi.org/10.1007/s12288-019-01200-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12288-019-01200-y