Abstract
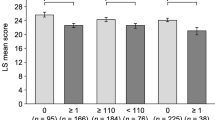
To describe the health-related quality of life (HRQoL) and fatigue burden among adult patients with immune thrombocytopenia (ITP) in China and determine whether they vary with disease phase. This is a cross-sectional, multi-centre observational study of adult ITP patients and the general population. Participants completed the Medical Outcomes Study 36-Item Short Form Health Survey (SF-36) and Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F). We compared HRQoL and fatigue between ITP patients and the general population, overall and by disease phase (newly diagnosed, persistent, and chronic), using propensity score matching. 203 Patients and 269 members of the general population were recruited. Thirty-six ITP patients (17.7%) were newly diagnosed, 46 (22.7%) were persistent, and 121 (59.6%) were chronic. Compared with the general population, ITP patients had impaired HRQoL and greater fatigue burden. The persistent ITP group showed the largest number of SF-36 scales exceeding the minimally important difference: physical functioning [− 10.5; 95% confidence interval (CI) − 24.5 to 3.5; P < 0.001], role physical (− 16.7; 95% CI − 36.4 to 3.0; P < 0.001), social functioning (− 15.6; 95% CI − 34.5 to 3.3; P < 0.001), and role emotional (− 12.1; 95% CI − 26.0 to 1.8; P < 0.001). Chronic ITP patients had the worst FACIT-F scores (36.89 ± 5.21). Higher fatigue severity was associated with lower physical and mental HRQoL. The HRQoL and fatigue burden of Chinese adult patients with ITP vary by disease phase. Persistent ITP patients were the most vulnerable subgroup in terms of HRQoL, while chronic ITP patients bear the heaviest fatigue burden.


Similar content being viewed by others
References
Grace RF, Neunert C (2016) Second-line therapies in immune thrombocytopenia. Hematol Am Soc Hematol Educ Program 2016(1):698–706
Liu XG, Bai XC, Chen FP, Cheng YF, Dai KS, Fang MY, Feng JM, Gong YP, Guo T, Guo XH, Han Y, Hong LJ, Hu Y, Hua BL, Huang RB, Li Y, Peng J, Shu MM, Sun J, Sun PY, Sun YQ, Wang CS, Wang SJ, Wang XM, Wu CM, Wu WM, Yan ZY, Yang FE, Yang LH, Yang RC, Yang TH, Ye X, Zhang GS, Zhang L, Zheng CC, Zhou H, Zhou M, Zhou RF, Zhou ZP, Zhu HL, Zhu TN, Hou M (2018) Chinese guidelines for treatment of adult primary immune thrombocytopenia. Int J Hematol 107(6):615–623
Audia S, Mahévas M, Samson M, Godeau B, Bonnotte B (2017) Pathogenesis of immune thrombocytopenia. Autoimmu Rev 16(6):620–632
Neunert C, Arnold DM (2015) Severe bleeding events in adults and children with primary immune thrombocytopenia: a systematic review: reply. J Thromb Haemost 13(8):1522–1523
Zhou Z, Yang L, Chen Z, Chen X, Guo Y, Wang X, Dong X, Wang T, Zhang L, Qiu Z, Yang R (2010) Health-related quality of life measured by the short form 36 in immune thrombocytopenic purpura: a cross-sectional survey in China. Eur J Haematol 78(6):518–523
Khelif A, Saleh MN, Salama A, Portella MDSO, Duh MS, Ivanova J, Grotzinger K, Roy AN, Bussel JB (2018) Changes in health-related quality of life with long-term eltrombopag treatment in adults with persistent/chronic immune thrombocytopenia: findings from the EXTEND study. Am J Hematol. https://doi.org/10.1002/ajh.25348
McMillan R, Bussel J, George J, Lalla D, Nichol JL (2010) Self-reported health-related quality of life in adults with chronic immune thrombocytopenic purpura. Am J Hematol 83(2):150–154
Hill QA, Newland AC (2015) Fatigue in immune thrombocytopenia. Br J Haematol 170(2):141–149
Rodeghiero F, Stasi R, Gernsheimer T, Michel M, Provan D, Arnold DM, Bussel JB, Cines DB, Chong BH, Cooper N, Godeau B, Lechner K, Mazzucconi MG, McMillan R, Sanz MA, Imbach P, Blanchette V, Kühne T, Ruggeri M, George JN (2009) Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood 113(11):2386–2393
Li L, Wang HM, Shen Y (2003) Chinese SF-36 health survey: translation, cultural adaptation, validation, and normalisation. J Epidemiol Commun Health 57(4):259–263
Efficace F, Mandelli F, Fazi P, Santoro C, Gaidano G, Cottone F, Borchiellini A, Carpenedo M, Simula MP, Di Giacomo V, Bergamaschi M, Vincelli ID, Rodeghiero F, Ruggeri M, Scaramucci L, Rambaldi A, Cascavilla N, Forghieri F, Petrungaro A, Ditonno P, Caocci G, Cirrincione S, Mazzucconi MG (2016) Health-related quality of life and burden of fatigue in patients with primary immune thrombocytopenia by phase of disease. Am J Hematol 91(10):995–1001
Signorovitch J, Brainsky A, Grotzinger KM (2011) Validation of the FACIT-fatigue subscale, selected items from FACT-thrombocytopenia, and the SF-36v2 in patients with chronic immune thrombocytopenia. Qual Life Res 20(10):1737–1744
Sloan J, Symonds T, Vargas-Chanes D, Fridley B (2003) Practical guidelines for assessing the clinical significance of health-related quality of life changes within clinical trials. Drug Inf J 37(1):23–31
Valderas JM, Alonso J (2008) Patient reported outcome measures: a model based classification system for research and clinical practice. Qual Life Res 17(9):1125–1135
Snyder CF, Mathias SD, Cella D, Isitt JJ, Wu AW, Young J (2008) Health-related quality of life of immune thrombocytopenic purpura patients: results from a web-based survey. Curr Med Res Opin 24(10):2767–2776
Mathias SD, Bussel JB, George JN, McMillan R, Okano GJ, Nichol JL (2007) A disease-specific measure of health-related quality of life in adults with chronic immune thrombocytopenic purpura: psychometric testing in an open-label clinical trial. Clin Ther 29(5):950–962
Newton JL, Reese JA, Watson SI, Vesely SK, Bolton-Maggs PH, George JN, Terrell DR (2011) Fatigue in adult patients with primary immune thrombocytopenia. Eur J Haematol 86(5):420–429
Efficace F, Baccarani M, Breccia M, Cottone F, Alimena G, Deliliers GL, Baratè C, Specchia G, Di Lorenzo R, Luciano L, Turri D, Martino B, Stagno F, Dabusti M, Bergamaschi M, Leoni P, Simula MP, Levato L, Fava C, Veneri D, Sica S, Rambaldi A, Rosti G, Vignetti M, Mandelli F (2013) Chronic fatigue is the most important factor limiting health-related quality of life of chronic myeloid leukemia patients treated with imatinib. Leukemia 27(7):1511–1519
Acknowledgements
We are grateful to all the participants in this research. We are also grateful to Xiaoqin Yang and Xiaozhu Yang for their kind help and support for this study.
Funding
This work was supported by Opening Foundation of Jiangsu Branch of China Academy of Chinese Medical Sciences (Grant No. FY201702).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Rights and permissions
About this article
Cite this article
Yang, R., Yao, H., Lin, L. et al. Health-Related Quality of Life and Burden of Fatigue in Chinese Patients with Immune Thrombocytopenia: A Cross-Sectional Study. Indian J Hematol Blood Transfus 36, 104–111 (2020). https://doi.org/10.1007/s12288-019-01124-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12288-019-01124-7