Abstract
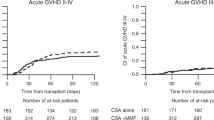
We sought to compare two graft-versus-host disease (GVHD) prophylaxis regimen, cyclosporine and methotrexate (CsA+MTX) with CsA+mycophenolate mofetil (MMF) in 77 acute leukemia patients who underwent hematopoietic stem cell transplant (HSCT) between January 2008 and March 2013. Fifty-three patients received CsA+MTX while 24 received CsA+MMF. The incidence of grade 3–4 mucositis and grade 3–4 diarrhea was 74 and 6 % with CsA+MTX compared to 33 % and 21 % with CsA+MMF (P = 0.001 and 0.09 respectively). Forty-two (79 %) patients in CsA+MTX group required total parenteral nutrition compared to 14 (58 %) in CsA+MMF group (P = 0.09). The incidence of engraftment fever was 17 % with CsA+MTX and 41 % with CsA+MMF (P = 0.02). The median time to neutrophil and platelet engraftment was 14 days and 13 days with CsA+MTX compared to 12 days and 10 days with CsA+MMF (P = 0.003 and 0.08 respectively). The incidence of any grade and grade II–IV acute GVHD was 45 and 13 % with CsA+MTX compared to 42 and 29 % with CsA+MMF (P = NS). Incidence of overall and extensive chronic GVHD was 57 and 38 % with CsA+MTX compared to 42 and 17 % with CsA+MMF (P = NS). Incidence of relapse was 38 % with CsA+MTX compared to 33 % with CsA+MMF (P = NS). TRM was 6 % with CsA+MTX and 21 % with CsA+MMF (P = NS). At 2 years, overall survival (OS) was 64 % in CsA+MTX group compared to 46 % in CsA+MMF group (P = NS). We conclude that CsA+MMF is associated with lesser toxicity, faster myeloid engraftment and similar rates of acute and chronic GVHD, TRM, relapse and OS compared to CsA+MTX in acute leukemia transplant.
Similar content being viewed by others
References
Kolb HJ (2008) Graft-versus-leukemia effects of transplantation and donor lymphocytes. Blood 112:4371–4383
Mohty M, Gaugler B (2008) Inflammatory cytokines and dendritic cells in acute graft-versus-host disease after allogeneic stem cell transplantation. Cytokine Growth Factor Rev 19:53–63
Powles RL, Clink HM, Spence D, Morgenstern G, Watson JG, Selby PJ et al (1980) Cyclosporin A to prevent graft-versus-host disease in man after allogeneic bone-marrow transplantation. Lancet 16:327–329
Storb R, Deeg HJ, Pepe M, Appelbaum F, Anasetti C, Beatty P et al (1989) Methotrexate and cyclosporine versus cyclosporine alone for prophylaxis of graft-versus-host disease in patients given HLA-identical marrow grafts for leukaemia: long-term follow up of a controlled trial. Blood 73:1729–1734
Ratanatharathorn V, Nash RA, Przepiorka D, Devine SM, Klein JL, Weisdorf D et al (1998) Phase III study comparing methotrexate and tacrolimus (prograf, FK506) with methotrexate and cyclosporine for graft-versus-host disease prophylaxis after HLA-identical sibling bone marrow transplantation. Blood 92:2303–2314
Nash RA, Antin JA, Karanes C, Fay JW, Avalos BR, Yeager AM et al (2000) Phase 3 study comparing methotrexate and tacrolimus with methotrexate and cyclosporine for prophylaxis of acute graft-versus-host disease after marrow transplantation. Blood 96:2062–2068
Hiraoka A, Ohashi Y, Okamoto S, Moriyama Y, Nagao T, Kodera Y et al (2001) Phase III study comparing tacrolimus (FK506) with cyclosporine for graft-versus-host disease prophylaxis after allogeneic bone marrow transplantation. Bone Marrow Transplant 28:181–185
Bolwell B, Sobecks R, Pohlman B, Andresen S, Rybicki L, Kuczkowski E et al (2004) A prospective randomized trial comparing cyclosporine and short course methotrexate with cyclosporine and mycophenolate mofetil for GVHD prophylaxis in myeloablative allogeneic bone marrow transplantation. Bone Marrow Transplant 34:621–625
Neumann F, Graef T, Tapprich C, Vaupel M, Steidl U, Germing U et al (2005) Cyclosporine A and mycophenolate mofetil vs cyclosporine A and methotrexate for graft-versus-host disease prophylaxis after stem cell transplantation from HLA-identical siblings. Bone Marrow Transplant 35:1089–1093
Piñana JL, Valcárcel D, Fernández-Avilés F et al (2010) MTX or mycophenolate mofetil with CsA as GVHD prophylaxis after reduced-intensity conditioning PBSCT from HLA-identical siblings. Bone Marrow Transplant 45:1449–1456
Gota V, Pathak K, Mittal S et al (2014) A limited sampling strategy for therapeutic drug monitoring of mycophenolate mofetil for prophylaxis of acute graft versus host disease in allogeneic stem cell transplantation. Bone Marrow Transplant. 49:S295
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA et al (1974) Clinical manifestations of graft-versus-host disease in human recipients of marrow from HLA-matched sibling donors. Transplantation 18:295–304
Shulman HM, Sullivan KM, Weiden PL, McDonald GB, Striker GE, Sale GE et al (1980) Chronic graft versus-host syndrome in man. A long-term clinicopathologic study of 20 Seattle patients. Am J Med 69:204–217
Storb R, Deeg HJ, Pepe M et al (1989) Graft-versus-host disease prevention by methotrexate combined with cyclosporine compared to methotrexate alone in patients given marrow grafts for severe aplastic anaemia: long-term follow-up of a controlled trial. Br J Haematol 72:567–572
Locatelli F, Bruno B, Zecca M et al (2000) Cyclosporine A and short-term methotrexate versus cyclosporine A as graft versus host disease prophylaxis in patients with severe aplastic anemia given allogeneic bone marrow transplantation from HLA identical sibling: results of a GITMO/EBMT randomized trial. Blood 96:1690–1697
Malard F, Szydlo RM, Brissot E, Chevallier P, Guillaume T, Delaunay J et al (2010) Impact of cyclosporine-A concentration on the incidence of severe acute graft-versus-host disease after allogeneic stem cell transplantation. Biol Blood Marrow Transplant 16:28–34
García Cadenas I, Valcarcel D, Martino R, Piñana JL, Barba P, Novelli S, et al. Impact of cyclosporine levels on the development of acute graft versus host disease after reduced intensity conditioning allogeneic stem cell transplantation. Mediators of Inflamm 2014. http://dx.doi.org/10.1155/2014/620682
Ruutu T, Gratwohl A, de Witte T, Afanasyev B, Apperley J, Bacigalupo A et al (2014) Prophylaxis and treatment of GVHD: EBMT-ELN working group recommendations for a standardized practice. Bone Marrow Transplant 49:168–173
Ruescher TJ, Sodeifi A, Scrivani SJ et al (1998) The impact of mucositis on a´ -hemolytic streptococcal infection in patients undergoing autologous bone marrow transplantation for hematological malignancies. Cancer 82:2275–2281
Bolwell BJ, Kalaycio M, Sobecks R et al (2002) A multivariable analysis of factors influencing mucositis after autologous progenitor cell transplantation. Bone Marrow Transplant 30:587–591
Atkinson K, Downs K (1995) Omission of day 11 methotrexate does not appear to influence the incidence of moderate to severe acute graft-versus-host disease, chronic graft-versus-host disease, relapse rate or survival after HLA-identical sibling bone marrow transplantation. Bone Marrow Transplant 16:755–758
Kumar S, Wolf RC, Chen MG et al (2002) Omission of day +11 methotrexate after allogeneic bone marrow transplantation is associated with increased risk of severe acute graft-versus-host disease. Bone Marrow Transplant 30:161–165
Kanda J, Kaynar L, Kanda Y et al (2013) Pre-engraftment syndrome after myeloablative dual umbilical cord blood transplantation: risk factors and response to treatment. Bone Marrow Transplant 48:926–931
Gondo H, Marada M, Taniguchi S et al (1993) Cyclosporine combined with methylprednisolone or methotrexate in prophylaxis of moderate to severe acute graft-versus-host disease. Bone Marrow Transplant 12:437–441
Acknowledgments
The authors declare that they have not received support in the form of grants and/or equipment and drugs from any source.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Gupta, A., Punatar, S., Mathew, L. et al. Cyclosporine Plus Methotrexate or Cyclosporine Plus Mycophenolate Mofetil as Graft Versus Host Disease Prophylaxis in Acute Leukemia Transplant: Comparison of Toxicity, Engraftment Kinetics and Transplant Outcome. Indian J Hematol Blood Transfus 32, 248–256 (2016). https://doi.org/10.1007/s12288-015-0577-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12288-015-0577-3