Abstract
Background
Exosomes released from cancer cells can activate normal fibroblasts (NFs) into cancer-associated fibroblasts (CAFs), which promotes cancer development. Our study aims to explore the role and potential mechanisms of breast cancer exosomes-delivered long non-coding RNA (lncRNA) SNHG14 in regulating CAFs transformation.
Methods
Adjacent normal tissues, cancerous and serum specimens were gathered in breast cancer patients. Exosomes and NFs were separated from breast cancer cells (SKBR-3) and normal tissues of patients, respectively. Cell viability and migration were measured with CCK-8 and Transwell assays. CAFs markers, fibroblast activation protein (FAP) and a-smooth muscle actin (α-SMA) were detected for assessing CAFs activation. The interactions between molecules were evaluated using dual luciferase reporter assay, RNA immunoprecipitation and chromatin immunoprecipitation.
Results
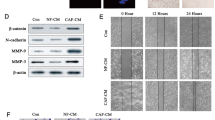
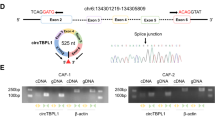
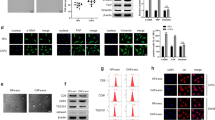
SNHG14 and FAM171A1 were upregulated in breast cancer. Exosomes secreted by SKBR-3 cells induced NFs activation in CAFs, as indicated by upregulating CAFs marker levels and facilitated cell viability and migration. Exosomal SNHG14 silencing in SKBR-3 cells inhibited CAFs activation. SNHG14 positively regulated FAM171A1 expression through EBF1. FAM171A1 overexpression eliminated the inhibition effect of exosomal SNHG14 silencing in CAFs transformation.
Conclusion
Breast cancer-derived exosomal SNHG14 contributed to NFs transformation into CAFs by the EBF1/FAM171A1 axis.





Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- lncRNAs:
-
Long non-coding RNAs
- SNHG14:
-
Small nucleolar RNA host gene 14
- NFs:
-
Normal fibroblasts
- CAFs:
-
Cancer-associated fibroblasts
- EBF1:
-
Early B cell factor 1
- FAM171A1:
-
The family with sequence similarity 171, member A1 protein
- α-SMA:
-
A-smooth muscle actin
- FAP:
-
Fibroblast activation protein
- TFF3:
-
Trefoil factor 3
- KLF5:
-
Kruppel-like factor 5
- LMTK3:
-
Lemur tyrosine kinase-3
- EGR1:
-
Early growth response protein 1
- KLF7:
-
Krüppel-like factor 7
- CTSF:
-
Cathepsin F
- HEY1:
-
YRPW motif 1
- C16orf74:
-
Chromosome 16 open reading frame 74
References
Fahad Ullah M. Breast cancer: current perspectives on the disease status. Adv Exp Med Biol. 2019;1152:51–64.
Yoshimaru T, Nakamura Y, Katagiri T. Functional genomics for breast cancer drug target discovery. J Hum Genet. 2021;66(9):927–35.
Koual M, Tomkiewicz C, Cano-Sancho G, Antignac JP, Bats AS, Coumoul X. Environmental chemicals, breast cancer progression and drug resistance. Environ Health. 2020;19(1):117.
Trayes KP, Cokenakes SEH. Breast cancer treatment. Am Fam Physician. 2021;104(2):171–8.
Xiao Y, Yu D. Tumor microenvironment as a therapeutic target in cancer. Pharmacol Ther. 2021;221: 107753.
Mao X, Xu J, Wang W, Liang C, Hua J, Liu J, Zhang B, Meng Q, Yu X, Shi S. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Mol Cancer. 2021;20(1):131.
Huang L, Xu AM, Liu S, Liu W, Li TJ. Cancer-associated fibroblasts in digestive tumors. World J Gastroenterol. 2014;20(47):17804–18.
Schoepp M, Strose AJ, Haier J. Dysregulation of miRNA expression in cancer associated fibroblasts (CAFs) and its consequences on the tumor microenvironment. Cancers (Basel). 2017;9(6):54.
Ringuette Goulet C, Bernard G, Tremblay S, Chabaud S, Bolduc S, Pouliot F. Exosomes induce fibroblast differentiation into cancer-associated fibroblasts through TGFbeta signaling. Mol Cancer Res. 2018;16(7):1196–204.
Ren Z, Lv M, Yu Q, Bao J, Lou K, Li X. MicroRNA-370-3p shuttled by breast cancer cell-derived extracellular vesicles induces fibroblast activation through the CYLD/Nf-kappaB axis to promote breast cancer progression. FASEB J. 2021;35(3): e21383.
Yang SS, Ma S, Dou H, Liu F, Zhang SY, Jiang C, Xiao M, Huang YX. Breast cancer-derived exosomes regulate cell invasion and metastasis in breast cancer via miR-146a to activate cancer associated fibroblasts in tumor microenvironment. Exp Cell Res. 2020;391(2): 111983.
Kok VC, Yu CC. Cancer-derived exosomes: their role in cancer biology and biomarker development. Int J Nanomedicine. 2020;15:8019–36.
Yang X, Li Y, Zou L, Zhu Z. Role of exosomes in crosstalk between cancer-associated fibroblasts and cancer cells. Front Oncol. 2019;9:356.
Alzhrani GN, Alanazi ST, Alsharif SY, Albalawi AM, Alsharif AA, Abdel-Maksoud MS, Elsherbiny N. Exosomes: isolation, characterization, and biomedical applications. Cell Biol Int. 2021;45(9):1807–31.
Fang Z, Xu J, Zhang B, Wang W, Liu J, Liang C, Hua J, Meng Q, Yu X, Shi S. The promising role of noncoding RNAs in cancer-associated fibroblasts: an overview of current status and future perspectives. J Hematol Oncol. 2020;13(1):154.
Choudhari R, Sedano MJ, Harrison AL, Subramani R, Lin KY, Ramos EI, Lakshmanaswamy R, Gadad SS. Long noncoding RNAs in cancer: from discovery to therapeutic targets. Adv Clin Chem. 2020;95:105–47.
Wang Z, Wang X, Zhang T, Su L, Liu B, Zhu Z, Li C. LncRNA MALAT1 promotes gastric cancer progression via inhibiting autophagic flux and inducing fibroblast activation. Cell Death Dis. 2021;12(4):368.
Ren X, Li L, Wu J, Lin K, He Y, Bian L. PDGF-BB regulates the transformation of fibroblasts into cancer-associated fibroblasts via the lncRNA LURAP1L-AS1/LURAP1L/IKK/IkappaB/NF-kappaB signaling pathway. Oncol Lett. 2021;22(1):537.
Tong Y, Yang L, Yu C, Zhu W, Zhou X, Xiong Y, Wang W, Ji F, He D, Cao X. Tumor-secreted exosomal lncRNA POU3F3 promotes cisplatin resistance in ESCC by inducing fibroblast differentiation into CAFs. Mol Ther Oncolytics. 2020;18:1–13.
Hu T, Hu J. Melanoma-derived exosomes induce reprogramming fibroblasts into cancer-associated fibroblasts via Gm26809 delivery. Cell Cycle. 2019;18(22):3085–94.
Shen S, Wang Y, Zhang Y, Dong Z, Xing J. Long non-coding RNA small nucleolar RNA host gene 14, a promising biomarker and therapeutic target in malignancy. Front Cell Dev Biol. 2021;9: 746714.
Zhang D, Ding X, Peng M. LncRNA SNHG14 accelerates breast cancer progression through sponging miR-543 and regulating KLF7 expression. Arch Gynecol Obstet. 2022;305(6):1507–16.
Tang J, Li Y, Sang Y, Yu B, Lv D, Zhang W, Feng H. LncRNA PVT1 regulates triple-negative breast cancer through KLF5/beta-catenin signaling. Oncogene. 2018;37(34):4723–34.
Shuang O, Zhou J, Cai Z, Liao L, Wang Y, Wang W, Xu M. EBF1-mediated up-regulation of lncRNA FGD5-AS1 facilitates osteosarcoma progression by regulating miR-124-3p/G3BP2 axis as a ceRNA. J Orthop Surg Res. 2022;17(1):332.
Fernandez-Jimenez N, Sklias A, Ecsedi S, Cahais V, Degli-Esposti D, Jay A, Ancey PB, Woo HD, Hernandez-Vargas H, Herceg Z. Lowly methylated region analysis identifies EBF1 as a potential epigenetic modifier in breast cancer. Epigenetics. 2017;12(11):964–72.
Sanawar R, Mohan Dan V, Santhoshkumar TR, Kumar R, Pillai MR. Estrogen receptor-alpha regulation of microRNA-590 targets FAM171A1-a modifier of breast cancer invasiveness. Oncogenesis. 2019;8(1):5.
Bhowmick S, Moore JT, Kirschner DL, Drew KL. Arctic ground squirrel hippocampus tolerates oxygen glucose deprivation independent of hibernation season even when not hibernating and after ATP depletion, acidosis, and glutamate efflux. J Neurochem. 2017;142(1):160–70.
Barone I, Gelsomino L, Accattatis FM, Giordano F, Gyorffy B, Panza S, Giuliano M, Veneziani BM, Arpino G, De Angelis C, et al. Analysis of circulating extracellular vesicle derived microRNAs in breast cancer patients with obesity: a potential role for Let-7a. J Transl Med. 2023;21(1):232.
Fu H, Yang H, Zhang X, Xu W. The emerging roles of exosomes in tumor-stroma interaction. J Cancer Res Clin Oncol. 2016;142(9):1897–907.
Hu D, Li Z, Zheng B, Lin X, Pan Y, Gong P, Zhuo W, Hu Y, Chen C, Chen L, et al. Cancer-associated fibroblasts in breast cancer: challenges and opportunities. Cancer Commun (Lond). 2022;42(5):401–34.
Magesh P, Thankachan S, Venkatesh T, Suresh PS. Breast cancer fibroblasts and cross-talk. Clin Chim Acta. 2021;521:158–69.
Ahn YH, Kim JS. Long non-coding RNAs as regulators of interactions between cancer-associated fibroblasts and cancer cells in the tumor microenvironment. Int J Mol Sci. 2020;21(20):7484.
Li K, Liu T, Chen J, Ni H, Li W. Survivin in breast cancer-derived exosomes activates fibroblasts by up-regulating SOD1, whose feedback promotes cancer proliferation and metastasis. J Biol Chem. 2020;295(40):13737–52.
Scognamiglio I, Cocca L, Puoti I, Palma F, Ingenito F, Quintavalle C, Affinito A, Roscigno G, Nuzzo S, Chianese RV, et al. Exosomal microRNAs synergistically trigger stromal fibroblasts in breast cancer. Mol Ther Nucleic Acids. 2022;28:17–31.
Xie SD, Qin C, Jin LD, Wang QC, Shen J, Zhou JC, Chen YX, Huang AH, Zhao WH, Wang LB. Long noncoding RNA SNHG14 promotes breast cancer cell proliferation and invasion via sponging miR-193a-3p. Eur Rev Med Pharmacol Sci. 2020;24(14):7543.
Hua Z, White J, Zhou J. Cancer stem cells in TNBC. Semin Cancer Biol. 2022;82:26–34.
Melone V, Salvati A, Brusco N, Alexandrova E, D’Agostino Y, Palumbo D, Palo L, Terenzi I, Nassa G, Rizzo F, et al. Functional relationships between long non-coding RNAs and estrogen receptor alpha: a new frontier in hormone-responsive breast cancer management. Int J Mol Sci. 2023;24(2):1145.
Long Y, Wang X, Youmans DT, Cech TR. How do lncRNAs regulate transcription? Sci Adv. 2017;3(9): eaao2110.
Zheng L, Cao J, Liu L, Xu H, Chen L, Kang L, Gao L. Long noncoding RNA LINC00982 upregulates CTSF expression to inhibit gastric cancer progression via the transcription factor HEY1. Am J Physiol Gastrointest Liver Physiol. 2021;320(5):G816–28.
Gong J, Fan H, Deng J, Zhang Q. LncRNA HAND2-AS1 represses cervical cancer progression by interaction with transcription factor E2F4 at the promoter of C16orf74. J Cell Mol Med. 2020;24(11):6015–27.
Santuario-Facio SK, Cardona-Huerta S, Perez-Paramo YX, Trevino V, Hernandez-Cabrera F, Rojas-Martinez A, Uscanga-Perales G, Martinez-Rodriguez JL, Martinez-Jacobo L, Padilla-Rivas G, et al. A new gene expression signature for triple negative breast cancer using frozen fresh tissue before neoadjuvant chemotherapy. Mol Med. 2017;23:101–11.
Fu XD, Liu CY, Liu YL, Su DW, Chi NN, Zhang JL, Wei WW. LINC00261 regulates EBF1 to suppress malignant progression of thyroid cancer. Eur Rev Med Pharmacol Sci. 2021;25(24):7626–34.
Luo H, Yang L, Liu C, Wang X, Dong Q, Liu L, Wei Q. TMPO-AS1/miR-98-5p/EBF1 feedback loop contributes to the progression of bladder cancer. Int J Biochem Cell Biol. 2020;122: 105702.
Liu GM, Lu TC, Sun ML, Ji X, Zhao YA, Jia WY, Luo YG. RP11–874J12.4 promotes oral squamous cell carcinoma tumorigenesis via the miR-19a-5p/EBF1 axis. J Oral Pathol Med. 2020;49(7):645–54.
Shen A, Chen Y, Liu L, Huang Y, Chen H, Qi F, Lin J, Shen Z, Wu X, Wu M, et al. EBF1-mediated upregulation of ribosome assembly factor PNO1 contributes to cancer progression by negatively regulating the p53 signaling pathway. Cancer Res. 2019;79(9):2257–70.
Qiu K, Zheng Z, Huang Y. Long intergenic noncoding RNA 00844 promotes apoptosis and represses proliferation of prostate cancer cells through upregulating GSTP1 by recruiting EBF1. J Cell Physiol. 2020;235(11):8472–85.
Acknowledgements
Not applicable.
Funding
This work was supported by The Key Research and Development Program of Hainan Province (ZDYF2021SHFZ055) and Hainan Provincial Natural Science Foundation of China (822CXTD535) and National Science Foundation of China (81960475).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval and consent to participate
All participants signed informed consent for specimen collection. The research was approved by the Research Ethics Committee in Hainan General Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Dong, H., Yang, C., Chen, X. et al. Breast cancer-derived exosomal lncRNA SNHG14 induces normal fibroblast activation to cancer-associated fibroblasts via the EBF1/FAM171A1 axis. Breast Cancer 30, 1028–1040 (2023). https://doi.org/10.1007/s12282-023-01496-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-023-01496-9