Abstract
Background
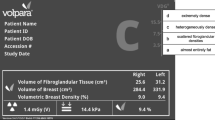
The volumetric measurement system for mammographic breast density is a high-precision objective method for evaluating the percentage of fibroglandular tissue volume (FG%). Nonetheless, FG% does not precisely correlate with subjective visual estimation (SVE) and shows poor evaluation performance regarding masking risk in patients with comparatively thin compressed breast thickness (CBT), commonly found in Japanese women. We considered that the mean compressed fibroglandular tissue thickness (mCGT), which incorporates the CBT element into the evaluation of breast density, may better predict masking risk.
Methods
Volumetric measurements and SVEs were performed on mammograms of 108 breast cancer patients from our center. mCGT was calculated as the product of CBT and FG%. SVE was classified using the Breast Imaging-Reporting and Data System classification, 5th edition. Subsequently, the performance of mCGT, SVE, and FG% in predicting masking risk was estimated using the AUC.
Results
The AUC values of mCGT and SVE were 0.84 (95% confidence interval, 0.71–0.92) and 0.78 (0.66–0.86), respectively (P = 0.16). The AUC of the FG% was 0.65 (0.52–0.77), which was significantly lower than that of mCGT (P < 0.001). The sensitivity and specificity of mCGT in predicting negative detection were 89% and 71%, respectively; of SVE 83% and 61% (versus 72% and 57% with FG%), suggesting that mCGT was superior to FG% in both sensitivity and specificity, and comparable with SVE.
Conclusions
Objective mCGT calculated from the volumetric measurement system will highly likely be useful in evaluating breast density and supporting visual assessment for masking risk stratification.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AI:
-
Artificial intelligence
- BA:
-
Breast area
- BV:
-
Breast volume
- CBT:
-
Compressed breast thickness
- CI:
-
Confidence interval
- FG%:
-
Percentage of fibroglandular tissue volume to breast volume
- FGV:
-
Fibroglandular tissue volume
- IQR:
-
Interquartile range
- κ :
-
Kappa coefficient
- mCGT:
-
Mean compressed fibroglandular tissue thickness
- MLO:
-
Mediolateral oblique
- SVE:
-
Subjective visual estimation
References
Saquib N, Saquib J, Ioannidis JP. Does screening for disease save lives in asymptomatic adults? Systematic review of meta-analyses and randomized trials. Int J Epidemiol. 2015;44:264–77. https://doi.org/10.1093/ije/dyu140.
Mandelson MT, Oestreicher N, Porter PL, White D, Finder CA, Taplin SH, et al. Breast density as a predictor of mammographic detection: comparison of interval- and screen-detected cancers. J Natl Cancer Inst. 2000;92:1081–7. https://doi.org/10.1093/jnci/92.13.1081.
Kolb TM, Lichy J, Newhouse JH. Comparison of the performance of screening mammography, physical examination, and breast US and evaluation of factors that influence them: an analysis of 27,825 patient evaluations. Radiology. 2002;225:165–75. https://doi.org/10.1148/radiol.2251011667.
Carney PA, Miglioretti DL, Yankaskas BC, Kerlikowske K, Rosenberg R, Rutter CM, et al. Individual and combined effects of age, breast density, and hormone replacement therapy use on the accuracy of screening mammography. Ann Intern Med. 2003;138:168–75. https://doi.org/10.7326/0003-4819-138-3-200302040-00008.
Wolfe JN. Breast patterns as an index of risk for developing breast cancer. AJR Am J Roentgenol. 1976;126:1130–7.
Boyd NF, Guo H, Martin LJ, Sun L, Stone J, Fishell E, et al. Mammographic density and the risk and detection of breast cancer. N Engl J Med. 2007;356:227–36.
Tabár L, Dean PB. Mammographic parenchymal patterns. Risk indicator for breast cancer? JAMA. 1982;247:185–9.
Sickles EA, D’Orsi CJ, Bassett LW, Appleton CM, Berg WA, Burnside ES et al. Breast Imaging Reporting and Data System (BI-RADS) atlas. 5th ed. Reston, VA: American College of Radiology; 2013.
Ciatto S, Houssami N, Apruzzese A, Bassetti E, Brancato B, Carozzi F, et al. Categorizing breast mammographic density: intra- and interobserver reproducibility of BI-RADS density categories. Breast. 2005;14:269–75. https://doi.org/10.1016/j.breast.2004.12.004.
Redondo A, Comas M, Macià F, Ferrer F, Murta-Nascimento C, Maristany MT, et al. Inter- and intraradiologist variability in the BI-RADS assessment and breast density categories for screening mammograms. Br J Radiol. 2012;85:1465–70. https://doi.org/10.1259/bjr/21256379.
Bernardi D, Pellegrini M, Di Michele S, Tuttobene P, Fantò C, Valentini M, et al. Interobserver agreement in breast radiological density attribution according to BI-RADS quantitative classification. Radiol Med. 2012;117:519–28. https://doi.org/10.1007/s11547-011-0777-3.
Eng A, Gallant Z, Shepherd J, McCormack V, Li J, Dowsett M, et al. Digital mammographic density and breast cancer risk: a case–control study of six alternative density assessment methods. Breast Cancer Res. 2014;16:439. https://doi.org/10.1186/s13058-014-0439-1.
Brandt KR, Scott CG, Ma L, Mahmoudzadeh AP, Jensen MR, Whaley DH, et al. Comparison of clinical and automated breast density measurements: implications for risk prediction and supplemental screening. Radiology. 2016;279:710–9. https://doi.org/10.1148/radiol.2015151261.
Astley SM, Harkness EF, Sergeant JC, Warwick J, Stavrinos P, Warren R, et al. A comparison of five methods of measuring mammographic density: a case–control study. Breast Cancer Res. 2018;20:10. https://doi.org/10.1186/s13058-018-0932-z.
Oiwa M, Endo T, Suda N, Morita T, Sato Y, Kawasaki T, et al. Can quantitative evaluation of mammographic breast density, “volumetric measurement”, predict the masking risk with dense breast tissue? Investigation by comparison with subjective visual estimation by Japanese radiologists. Breast Cancer. 2019;26:349–58. https://doi.org/10.1007/s12282-018-0930-0.
Ohnuki K, Umemura A, Asano S, Usami S, Watanabe M. The clinical significance of compressed breast thickness in mammography: the reality of dense breast and efficient breast cancer screening. Jpn J Breast Cancer. 2019;34:55–63 (in Japanese with English abstract).
Noma M, Matsuura K, Itamoto T, Ishikura M, Saito H. Investigation of the relationship between breast density and detection of breast cancer by mammography. J Jpn Assoc Cancer Detect Diagn. 2018;25:160–4 (in Japanese with English abstract).
Machida Y, Saita A, Namba H, Fukuma E. Automated volumetric breast density estimation out of digital breast tomosynthesis data: feasibility study of a new software version. Springerplus. 2016;5:780. https://doi.org/10.1186/s40064-016-2519-4.
Wang J, Azziz A, Fan B, Malkov S, Klifa C, Newitt D, et al. Agreement of mammographic measures of volumetric breast density to MRI. PLoS One. 2013;8: e81653. https://doi.org/10.1371/journal.pone.0081653.
Gubern-Mérida A, Kallenberg M, Platel B, Mann RM, Martí R, Karssemeijer N. Volumetric breast density estimation from full-field digital mammograms: a validation study. PLoS One. 2014;9: e85952. https://doi.org/10.1371/journal.pone.0085952.
Sartor H, Lång K, Rosso A, Borgquist S, Zackrisson S, Timberg P. Measuring mammographic density: comparing a fully automated volumetric assessment versus European radiologists’ qualitative classification. Eur Radiol. 2016;26:4354–60. https://doi.org/10.1007/s00330-016-4309-3.
Gemici AA, Arıbal E, Özaydın AN, Gürdal SÖ, Özçınar B, Cabioğlu N, et al. Comparison of qualitative and volumetric assessments of breast density and analyses of breast compression parameters and breast volume of women in Bahcesehir mammography screening project. Eur J Breast Health. 2020;16:110–6. https://doi.org/10.5152/ejbh.2020.4943.
Youk JH, Gweon HM, Son EJ, Kim JA. Automated volumetric breast density measurements in the era of the BI-RADS fifth edition: a comparison with visual assessment. AJR. 2016;206:1056–62. https://doi.org/10.2214/AJR.15.15472.
Destounis S, Johnston L, Highnam R, Arieno A, Morgan R, Chan A. Using volumetric breast density to quantify the potential masking risk of mammographic density. AJR Am J Roentgenol. 2017;208:222–7. https://doi.org/10.2214/AJR.16.16489.
Sawada T, Akashi S, Nakamura S, Kuwayama T, Enokido K, Yoshida M, et al. Digital volumetric measurement of mammographic density and the risk of overlooking cancer in Japanese women. Breast Cancer. 2017;24:708–13. https://doi.org/10.1007/s12282-017-0763-2.
Moshina N, Roman M, Waade GG, Sebuødegård S, Ursin G, Hofvind S. Breast compression parameters and mammographic density in the Norwegian Breast Cancer Screening Programme. Eur Radiol. 2018;28:1662–72. https://doi.org/10.1007/s00330-017-5104-5.
Holland K, van Gils CH, Mann RM, Karssemeijer N. Quantification of masking risk in screening mammography with volumetric breast density maps. Breast Cancer Res Treat. 2017;162:541–8. https://doi.org/10.1007/s10549-017-4137-4.
Burt JR, Torosdagli N, Khosravan N, Raviprakash H, Mortazi A, Tissavirasingham F, et al. Deep learning beyond cats and dogs: recent advances in diagnosing breast cancer with deep neural networks. Br J Radiol. 2018;91:20170545. https://doi.org/10.1259/bjr.20170545.
Rodriguez-Ruiz A, Lång K, Gubern-Merida A, Broeders M, Gennaro G, Clauser P, et al. Stand-alone artificial intelligence for breast cancer detection in mammography: comparison with 101 radiologists. J Natl Cancer Inst. 2019;111:916–22. https://doi.org/10.1093/jnci/djy222.
Sasaki M, Tozaki M, Rodríguez-Ruiz A, Yotsumoto D, Ichiki Y, Terawaki A, et al. Artificial intelligence for breast cancer detection in mammography: experience of use of the ScreenPoint Medical Transpara system in 310 Japanese women. Breast Cancer. 2020;27:642–51. https://doi.org/10.1007/s12282-020-01061-8.
Arieno A, Chan A, Destounis SV. A review of the role of augmented intelligence in breast imaging: from automated breast density assessment to risk stratification. AJR Am J Roentgenol. 2019;212:259–70. https://doi.org/10.2214/AJR.18.20391.
Chan HP, Helvie MA. Deep learning for mammographic breast density assessment and beyond. Radiology. 2019;290:59–60. https://doi.org/10.1148/radiol.2018182116.
Waade GG, Highnam R, Hauge IHR, McEntee MF, Hofvind S, Denton E, et al. Impact of errors in recorded compressed breast thickness measurements on volumetric density classification using volpara v1.5.0 software. Med Phys. 2016;43:2870–6. https://doi.org/10.1118/1.4948503.
Acknowledgements
The authors thank A. Iwakoshi, Y. Murakami, H. Usami, Y. Ando, E. Matsuda, Y. Sasada, C. Suzuki, A. Morishita, N. Hara, Y. Araki, M. Ohashi, A. Ishida, A. Iwama, A. Okamoto, and K. Yokoyama from the NHO Nagoya Medical Center for breast cancer examination, as well as Editage (http://www.editage.com) for English language editing. We would like to particularly thank M. Noma from the Department of Breast Surgery, Hiroshima Prefectural Hospital, for her helpful suggestions.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
MO, YS, and TE designed the study, collected and analyzed the data, and wrote the manuscript. MO and NS re-evaluated the mammograms. TM, YT, TH and AK carried out the clinical data collection and reviewed the medical charts. RN and SI made the diagnoses and carried out the pathological interpretations. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical standards
This study received approval from the Ethical Review Board of Nagoya Medical Center (approval number: 2021-008). For this type of study formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Oiwa, M., Suda, N., Morita, T. et al. Validity of computed mean compressed fibroglandular tissue thickness and breast composition for stratification of masking risk in Japanese women. Breast Cancer 30, 541–551 (2023). https://doi.org/10.1007/s12282-023-01444-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-023-01444-7