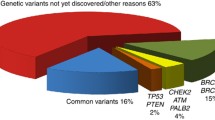
Abstract
Gene-environment interaction, a key idea in molecular epidemiology, has enabled the development of personalized medicine. This concept includes personalized prevention. While genome-wide association studies have identified a number of genetic susceptibility loci in breast cancer risk, however, the application of this knowledge to practical prevention is still underway. Here, we briefly review the history of molecular epidemiology and its progress in breast cancer epidemiology. We then introduce our experience with the trial combination of GWAS-identified loci and well-established lifestyle and reproductive risk factors in the risk prediction of breast cancer. Finally, we report our exploration of the cumulative risk of breast cancer based on this risk prediction model as a potential tool for individual risk communication, including genetic risk factors and gene-environment interaction with obesity.




Similar content being viewed by others
References
Lichtenstein P, Holm NV, Verkasalo PK, Iliadou A, Kaprio J, Koskenvuo M, et al. Environmental and heritable factors in the causation of cancer–analyses of cohorts of twins from Sweden, Denmark, and Finland. N Engl J Med. 2000;343:78–85.
Harris CC, Weston A, Willey JC, Trivers GE, Mann DL. Biochemical and molecular epidemiology of human cancer: indicators of carcinogen exposure, DNA damage, and genetic predisposition. Environ Health Perspect. 1987;75:109–19.
Perera FP, Poirier MC, Yuspa SH, Nakayama J, Jaretzki A, Curnen MM, et al. A pilot project in molecular cancer epidemiology: determination of benzo[a]pyrene–DNA adducts in animal and human tissues by immunoassays. Carcinogenesis. 1982;3:1405–10.
Lander ES, Linton LM, Birren B, Nusbaum C, Zody MC, Baldwin J, et al. Initial sequencing and analysis of the human genome. Nature. 2001;409:860–921.
International HapMap C. The International HapMap Project. Nature. 2003;426:789–96.
Consortium EP. An integrated encyclopedia of DNA elements in the human genome. Nature. 2012;489:57–74.
IARC. Alcoholic beverage consumption and ethyl carbamate (urethane). IARC monographs on the evaluation of carcinogenic risks to humans 96. Lyon: International Agency for Research on Cancer; 2010.
WCRF/AICR. Alcoholic drinks. Food, nutrition, physical activity, and the prevention of cancer: a global perspective. Washington, DC: American Institute for Cancer Research; 2007.
Yoshida A, Huang IY, Ikawa M. Molecular abnormality of an inactive aldehyde dehydrogenase variant commonly found in Orientals. Proc Natl Acad Sci USA. 1984;81:258–61.
Bosron WF, Crabb DW, Li TK. Relationship between kinetics of liver alcohol dehydrogenase and alcohol metabolism. Pharmacol Biochem Behav. 1983;18(Suppl 1):223–7.
Crabb DW, Edenberg HJ, Bosron WF, Li TK. Genotypes for aldehyde dehydrogenase deficiency and alcohol sensitivity. The inactive ALDH2(2) allele is dominant. J Clin Invest. 1989;83:314–6.
Li Y, Zhang D, Jin W, Shao C, Yan P, Xu C, et al. Mitochondrial aldehyde dehydrogenase-2 (ALDH2) Glu504Lys polymorphism contributes to the variation in efficacy of sublingual nitroglycerin. J Clin Invest. 2006;116:506–11.
Matsuo K, Wakai K, Hirose K, Ito H, Saito T, Tajima K. Alcohol dehydrogenase 2 His47Arg polymorphism influences drinking habit independently of aldehyde dehydrogenase 2 Glu487Lys polymorphism: analysis of 2,299 Japanese subjects. Cancer Epidemiol Biomarkers Prev. 2006;15:1009–13.
Matsuo K, Hamajima N, Shinoda M, Hatooka S, Inoue M, Takezaki T, et al. Gene-environment interaction between an aldehyde dehydrogenase-2 (ALDH2) polymorphism and alcohol consumption for the risk of esophageal cancer. Carcinogenesis. 2001;22:913–6.
Yang C, Wang H, Wang Z, Du H, Tao D, Mu X, et al. Risk factors for esophageal cancer: a case-control study in South-western China. Asian Pac J Cancer Prev. 2005;6:48–53.
Hiraki A, Matsuo K, Wakai K, Suzuki T, Hasegawa Y, Tajima K. Gene-gene and gene-environment interactions between alcohol drinking habit and polymorphisms in alcohol-metabolizing enzyme genes and the risk of head and neck cancer in Japan. Cancer Sci. 2007;98:1087–91.
Oze I, Matsuo K, Hosono S, Ito H, Kawase T, Watanabe M, et al. Comparison between self-reported facial flushing after alcohol consumption and ALDH2 Glu504Lys polymorphism for risk of upper aerodigestive tract cancer in a Japanese population. Cancer Sci. 2010;101:1875–80.
Matsuo K, Oze I, Hosono S, Ito H, Watanabe M, Ishioka K, et al. The aldehyde dehydrogenase 2 (ALDH2) Glu504Lys polymorphism interacts with alcohol drinking in the risk of stomach cancer. Carcinogenesis. 2013;34:1510–5.
Spitz MR, Bondy ML. The evolving discipline of molecular epidemiology of cancer. Carcinogenesis. 2010;31:127–34.
Thompson PA, Ambrosone C. Molecular epidemiology of genetic polymorphisms in estrogen metabolizing enzymes in human breast cancer. J Natl Cancer Inst Monogr. 2000;27:125–34.
Manolio TA. Genomewide association studies and assessment of the risk of disease. N Engl J Med. 2010;363:166–76.
Hunter DJ, Kraft P, Jacobs KB, Cox DG, Yeager M, Hankinson SE, et al. A genome-wide association study identifies alleles in FGFR2 associated with risk of sporadic postmenopausal breast cancer. Nat Genet. 2007;39:870–4.
Easton DF, Pooley KA, Dunning AM, Pharoah PD, Thompson D, Ballinger DG, et al. Genome-wide association study identifies novel breast cancer susceptibility loci. Nature. 2007;447:1087–93.
Sakoda LC, Jorgenson E, Witte JS. Turning of COGS moves forward findings for hormonally mediated cancers. Nat Genet. 2013;45:345–8.
Gail MH, Brinton LA, Byar DP, Corle DK, Green SB, Schairer C, et al. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst. 1989;81:1879–86.
Costantino JP, Gail MH, Pee D, Anderson S, Redmond CK, Benichou J, et al. Validation studies for models projecting the risk of invasive and total breast cancer incidence. J Natl Cancer Inst. 1999;91:1541–8.
Gail MH, Costantino JP, Pee D, Bondy M, Newman L, Selvan M, et al. Projecting individualized absolute invasive breast cancer risk in African American women. J Natl Cancer Inst. 2007;99:1782–92.
Matsuno RK, Costantino JP, Ziegler RG, Anderson GL, Li H, Pee D, et al. Projecting individualized absolute invasive breast cancer risk in Asian and Pacific Islander American women. J Natl Cancer Inst. 2011;103:951–61.
Lee AJ, Cunningham AP, Kuchenbaecker KB, Mavaddat N, Easton DF, Antoniou AC, et al. BOADICEA breast cancer risk prediction model: updates to cancer incidences, tumour pathology and web interface. Br J Cancer. 2014;110:535–45.
Sueta A, Ito H, Kawase T, Hirose K, Hosono S, Yatabe Y, et al. A genetic risk predictor for breast cancer using a combination of low-penetrance polymorphisms in a Japanese population. Breast Cancer Res Treat. 2012;132:711–21.
Wacholder S, Hartge P, Prentice R, Garcia-Closas M, Feigelson HS, Diver WR, et al. Performance of common genetic variants in breast-cancer risk models. N Engl J Med. 2010;362:986–93.
Zheng W, Wen W, Gao YT, Shyr Y, Zheng Y, Long J, et al. Genetic and clinical predictors for breast cancer risk assessment and stratification among Chinese women. J Natl Cancer Inst. 2010;102:972–81.
Tajima K, Hirose K, Inoue M, Takezaki T, Hamajima N, Kuroishi T. A Model of practical cancer prevention for out-patients visiting a hospital: the Hospital-based Epidemiologic Research Program at Aichi Cancer Center (HERPACC). Asian Pac J Cancer Prev. 2000;1:35–47.
Hamajima N, Matsuo K, Saito T, Hirose K, Inoue M, Takezaki T, et al. Gene-environment interactions and polymorphism studies of cancer risk in the Hospital-based Epidemiologic Research Program at Aichi Cancer Center II (HERPACC-II). Asian Pac J Cancer Prev. 2001;2:99–107.
Kawase T, Matsuo K, Suzuki T, Hiraki A, Watanabe M, Iwata H, et al. FGFR2 intronic polymorphisms interact with reproductive risk factors of breast cancer: results of a case control study in Japan. Int J Cancer. 2009;125:1946–52.
Travis RC, Reeves GK, Green J, Bull D, Tipper SJ, Baker K, et al. Gene-environment interactions in 7610 women with breast cancer: prospective evidence from the Million Women Study. Lancet. 2010;375:2143–51.
Brennan P, Crispo A, Zaridze D, Szeszenia-Dabrowska N, Rudnai P, Lissowska J, et al. High cumulative risk of lung cancer death among smokers and nonsmokers in Central and Eastern Europe. Am J Epidemiol. 2006;164:1233–41.
Peto R, Darby S, Deo H, Silcocks P, Whitley E, Doll R. Smoking, smoking cessation, and lung cancer in the UK since 1950: combination of national statistics with two case-control studies. BMJ. 2000;321:323–9.
Zipkin DA, Umscheid CA, Keating NL, Allen E, Aung K, Beyth R, et al. Evidence-based risk communication: a systematic review. Ann Intern Med. 2014;161:270–80.
Acknowledgments
The authors appreciate the efforts of the many contributors to the HERPACC study. This study was supported by Grants-in-Aid for Scientific Research on Priority Areas and Grant-in-Aid for Scientific Research (A) and (C) from the Ministry of Education, Science, Sports, Culture and Technology of Japan; by a Grant-in-Aid for the Third Term Comprehensive 10-year Strategy for Cancer Control from the Ministry of Health, Labour and Welfare of Japan; and by research grant from Takeda Science Foundation. These grantors were not involved in the study design, subject enrollment, study analysis or interpretation, or submission of the manuscript for this study.
Conflict of interest
Authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ito, H., Matsuo, K. Molecular epidemiology, and possible real-world applications in breast cancer. Breast Cancer 23, 33–38 (2016). https://doi.org/10.1007/s12282-015-0609-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-015-0609-8