Abstract
Background
To investigate the feasibility of in vivo proton magnetic resonance spectroscopy (1H-MRS) for characterizing malignant breast lesions using a clinical 3-T scanner, and to compare the measured total choline-containing compound (tCho) levels in this work with the previously published results at 1.5 T and 4 T.
Methods
Twenty-six patients (35–72 years old, mean age 53 years), with biopsy-confirmed breast cancer, underwent anatomical imaging, dynamic contrast-enhanced MR imaging, and single-voxel 1H-MRS. The absolute tCho concentration in malignant tumors was calculated and was expressed as a concentration unit in mmol/kg. All lesions were divided into two groups (mass- and non-mass-type lesions), based on the morphological pattern of enhancement, and analyzed.
Results
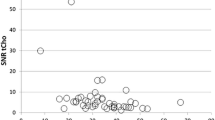
The measured tCho levels had a range of 0.10–10.1 (mean ± SD, 1.62 ± 2.29 mmol/kg) in 15 (58 %) of 26 malignant breast spectra, which was consistent with the previous in vivo estimates at 1.5 T and 4.0 T. There were 11 (42 %) false-negative cases, which resulted in 58 % sensitivity. The sensitivity of the 1H-MRS in size-dependent subgroups (e.g., <2.0 and ≥2.0 cm) increased from 14 to 66 %, in a statistically significant manner (P = 0.019). In addition, the tCho detection rate was higher in mass-type lesions (14/21, 66 %) than in non-mass-type lesions (1/5, 20 %), reaching a significant level (P = 0.03). The sensitivity based on the dynamic contrast-enhanced kinetics was 100 %.
Conclusions
Quantitative in vivo 1H-MRS at 3 T may provide useful information for quantifying tCho concentration levels and characterizing tumor types (e.g., mass vs diffuse) of breast cancer. However, its limitation in characterizing small lesions has to be considered.





Similar content being viewed by others
References
Roebuck JR, Cecil KM, Schnall MD, Lenkinski RE. Human breast lesions: characterization with proton MR spectroscopy. Radiology. 1998;209:269–75.
Kvistad KA, Bakken IJ, Gribbestad IS, Ehrnholm B, Lundgren S, Fjosne HE, Haraldseth O. Characterization of neoplastic and normal human breast tissues with in vivo 1H MR spectroscopy. J Magn Reson Imaging. 1999;10:159–64.
Jagannathan NR, Singh M, Govindaraju V, Raghunathan P, Coshic O, Julka PK, Rath GK. Volume localized in vivo proton MR spectroscopy of breast carcinoma: variation of water-fat ratio in patients receiving chemotherapy. NMR Biomed. 1998;11:414–22.
Jagannathan NR, Kumar M, Seenu V, Coshic O, Dwivedi SN, Julka PK, Srivastava A, Rath GK. Evaluation of total choline from in vivo volume localized proton MR spectroscopy and its response to neoadjuvant chemotherapy in locally advanced breast cancer. Br J Cancer. 2001;84:1016–22.
Yeung DK, Cheung HS, Tse GM. Human breast lesions: characterization with contrast-enhanced in vivo proton MR spectroscopy—initial results. Radiology. 2001;220:40–60.
Cecil KM, Schnall MD, Siegelman ES, Lenkinski RE. The evaluation of human breast lesions with magnetic resonance imaging and proton magnetic resonance spectroscopy. Breast Cancer Res Treat. 2001;68:45–54.
Tse GM, Ceung HS, Pang LM, Chu WC, Law BK, Kung FY, Yeung DK. Characterization of lesions of the breast with proton MR spectroscopy: comparison of carcinomas, benign lesions, and phyllodes tumors. Am J Roentgenol. 2003;181:1267–72.
Huang W, Fisher PR, Dulaimy K, Tudorica LA, Ohea B, Mutton TM. Detection of breast malignancy: diagnostic MR protocol for improved specificity. Radiology. 2004;232(2):585–91.
Bartella L, Morris EA, Dershaw DD, Liberman L, Thakur SB, Moskowitz C, Guido J, Huang W. Proton MR spectroscopy with choline peak as malignancy marker improves positive predictive value for breast cancer diagnosis: preliminary study. Radiology. 2006;239:686–92.
Baltzer PA, Dietzel M. Breast lesions: diagnosis by using proton MR spectroscopy at 1.5 T and 3.0 T—systematic review and meta-analysis. Radiology. 2013;267:735–46.
Begley JKP, Redpath TW, Jagpal B, Gilbert FJ. Single voxel proton magnetic resonance spectroscopy of breast cancer at 3 T. Breast Cancer Res. 2010;12(Suppl 3):P47.
Gruber S, Debski BK, Pinker K, Chmelik M, Grabner G, Helbich T, Trattnig S, Bogner W. Three-dimensional proton MR spectroscopic imaging at 3 T for the differentiation of benign and malignant breast lesions. Radiology. 2011;261:752–61.
Kousi E, Tsougos I, Vasiou K, Theodorou K, Poultsidi A, Fezoulidis I, Kappas C. Magnetic resonance spectroscopy of the breast at 3 T: pre-and post-contrast evaluation for breast lesion characterization. Sci World J. 2012;2012:754380.
Baek HM, Chen JH, Yu HJ, Mehta R, Nalicoglu O, Su MY. Detection of choline signal in human breast lesions with chemical-shift imaging. J Magn Reson Imaging. 2008;17:1114–21.
Haddadin IS, Mclntosh A, Meisamy S, Corum C, Styczynski Snyder AL, Powell NJ, Nelson MT, Yee D, Garwood M, Bolan PJ. Metabolite quantification and high-field MRS in breast cancer. NMR Biomed. 2009;22:65–76.
Bakken IJ, Gribbestad IS, Singstad TE, Kvistad KA. External standard method for the in vivo quantification of choline-containing compounds in breast tumors by proton MR spectroscopy at 1.5 Tesla. Magn Reson Med. 2001;46:189–92.
Bolan PJ, Meisamy S, Baker EH, Lin J, Emory T, Nelson M, Everson LI, Yee D, Garwood M. In vivo quantification of choline compounds in the breast with 1H MR spectroscopy. Magn Reson Med. 2003;50:1134–43.
Baek HM, Su MY, Yu H, Mehta R, Nalcioglu O. Quantification of choline-containing compounds in malignant breast tumors by 1H MR spectroscopy using water as an internal reference at 1.5 T. Magn Reson Mater Phys. 2006;19:96–104.
Baek HM, Yu H, Chen JH, Nalcioglu O, Su MY. Quantitative correlation between 1H MRS and dynamic contrast-enhanced MRI of human breast cancer. Magn Reson Imaging. 2008;26:523–31.
Meisamy S, Bolan PJ, Baker EH, et al. Neoadjuvant chemotherapy of locally advanced breast cancer: predicting response with in vivo 1H MR spectroscopy—a pilot study at 4 T. Radiology. 2004;233:424–31.
Baek HM, Chen JH, Nie K, et al. Predicting pathologic response to neoadjuvant chemotherapy in breast cancer by using MR imaging and quantitative 1H MR spectroscopy. Radiology. 2009;251:653–62.
Bottomley PA. Spatial localization in NMR spectroscopy in vivo. Ann N Y Acad Sci. 1987;508:333–48.
Ordidge RJ, Bendall MR, Gordon RE, Connelly A. Volume selection for in vivo spectroscopy. In: Govil G, Khetrapal CL, Sarans A, editors. Magnetic resonance in biology and medicine. New Delhi: Tata-McGraw-Hill; 1985. p. 387–97.
Haase A, Frahm J, Hanicke W, Mattaei D. 1H NMR chemical shift selective (CHESS) imaging. Phys Med Biol. 1985;30:431.
Doddrell DM, Galloway G, Brooks W, Filed J, Bulsing J, Irving M, Baddeley H. Water signal elimination in vivo, using suppression by mistimed echo and repetitive gradient episodes. J Magn Reson. 1986;70:176–80.
American College of Radiology. ACR breast imaging reporting and data system, breast imaging atlas. Reston: American College of Radiology; 2003.
Naressim A, Couturier C, Devos JM, Janssen M, Mangeat C, de Beer R, Graveron-Demilly D. Java-based graphical user interface for the MRUI quantitation package. MAGMA. 2001;12:141–52.
Vanhamme L, van den Boogaart A, Huffel SV. Improved method for accurate and efficient quantification of MRS data with use of prior knowledge. J Magn Reson. 1997;129:35–43.
Knijn A, de Beer R, van Ormondt D. Frequency-selective quantification in the time domain. J Magn Reson. 1992;97:444–50.
Tan PC, Pickles MD, Lowry M, Manton DJ, Turnbull LW. Lesion T2 relaxation times and volumes predict the response of malignant breast lesions to neoadjuvant chemotherapy. Magn Reson Imaging. 2008;26:26–34.
Ratiney H, Sdika M, Coenradie Y, Cavassila S, van Ormondt D, Graveron-Demilly D. Time-domain semi-parametric estimation based on a metabolite basis set. NMR Biomed. 2005;18:1–13.
Aboagye EO, Bhujwalla ZM. Malignant transformation alters membrane choline phospholipids metabolism of human mammary epithelial cells. Cancer Res. 1999;59:80–4.
Sitter B, Sonnewald U, Spraul M, Fjosne HE, Gribbestad IS. High-resolution magic angle spinning MRS of breast cancer tissue. NMR Biomed. 2002;15:327–37.
Ramires de Molina A, Gutierrez R, Ramos MA, et al. Increased choline kinase activity in human breast carcinoma: clinical evidence for a potential novel antitumor strategy. Oncogene. 2002;21:4317–22.
Ting Y-LT, Sherr D, Degani H. Variations in energy and phospholipid metabolism in normal and cancer human mammary epithelial cells. Anticancer Res. 1996;16:1381–8.
Gribbestad IS, Fjosne HE, Haugen OA, Nilsen G, Krane J, Petersen SB, Kvinnsland S. In vitro proton NMR spectroscopy of extracts from breast carcinoma and non-involved breast tissue. Anticancer Res. 1993;13:1973–80.
Bolan PJ, DelaBarre L, Baker EH, Merkle H, Everson LI, Yee D, Garwood M. Eliminating spurious lipid sidebands in 1H MRS of breast lesions. Magn Reson Med. 2002;48:215–22.
Bolan PJ, Henry PG, Baker EH, Meisamy S, Garwood M. Measurement and correction of respiration-induced B0 variations in breast 1H MRS at 4 Tesla. Magn Reson Med. 2004;52:1239–45.
Maril N, Collins CM, Greenman RL, Lenkinski RE. Strategies for shimming the breast. Magn Reson Med. 2005;54:1139–45.
Acknowledgments
This work was supported in part by KBSI (Korea Basic Science Institute) grant #E33601 and #T33416.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Baek, HM., Lee, YJ. Feasibility of MR spectroscopy for characterizing malignant breast lesions using a clinical 3-T scanner. Breast Cancer 22, 510–519 (2015). https://doi.org/10.1007/s12282-013-0514-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-013-0514-y