Abstract
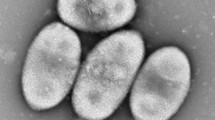
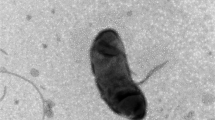
A Gram-stain-negative strictly aerobic, marine bacterium, designated GH2-2T, was isolated from a rhizosphere mudflat of a halophyte (Carex scabrifolia) in Gangwha Island, the Republic of Korea. The cells of the organism were oxidase-positive, catalase-positive, flagellated, short rods that grew at 10–40°C, pH 4–10, and 0–13% (w/v) NaCl. The predominant ubiquinone was Q-10. The major polar lipids were phosphatidylcholine, phosphatidylethanolamine, and phosphatidylglycerol. The major fatty acid is C18:1. Phylogenetic analysis based on 16S rRNA gene sequences revealed that the novel isolate formed an independent lineage at the base of the radiation encompassing members of the genus Thioclava, except for Thioclava arenosa. The closest relatives were T. nitratireducens (96.03% sequence similarity) and T. dalianensis (95.97%). The genome size and DNA G+C content were 3.77 Mbp and 59.6 mol%, respectively. Phylogenomic analysis supported phylogenetic distinctness based on 16S rRNA gene sequences. Average nucleotide identity values were 73.6–74.0% between the novel strain and members of the genus Thioclava. On the basis of data obtained from a polyphasic approach, the strain GH2-2T (= KCTC 62124T = DSM 105743) represents a novel species of a new genus for which the name Hahyoungchilella caricis gen. nov., sp. nov. is proposed. Moreover, the transfer of Thioclava arenosa Thongphrom et al. 2017 to Pseudothioclava gen. nov. as Pseudothioclava arenosa comb. nov. is also proposed. Finally, Thioclava electrotropha Chang et al. 2018 is proposed to be a later heterosynonym of Thioclava sediminum Liu et al. 2017.
Similar content being viewed by others
Change history
01 April 2020
In the article by Kim and Lee published in Journal of Microbiology 2019; 57, 1048–1055, The sentence ‘The type strain of <Emphasis Type="Italic">Pseudothioclava arenosa</Emphasis> is CAU 1312<Superscript>T</Superscript> (=KCTC 52190<Superscript>T</Superscript> = NBRC 111989<Superscript>T</Superscript>).’ should be added in the section ‘Description of <Emphasis Type="Italic">Pseudothioclava arenosa</Emphasis> comb. nov’ on 12th line of page 1054.
References
Brosius, J., Palmer, M.L., Kennedy, P.J., and Noller, H.F. 1978. Complete nucleotide sequence of a 16S ribosomal RNA gene from Escherichia coli. Proc. Natl. Acad. Sci. USA75, 4801–4805.
Chang, R., Bird, L., Barr, C., Osburn, M., Wilbanks, E., Nealson, K., and Rowe, A. 2018. Thioclava electrotropha sp. nov., a versatile electrode and sulfur-oxidizing bacterium from marine sediments. Int. J. Syst. Evol. Microbiol.68, 1652–1658.
Farris, J.S. 1972. Estimating phylogenetic trees from distance matrices. Am. Nat.106, 645–667.
Felsenstein J. 2002. PHYLIP (phylogeny inference package), version 3.6a. Department of Genome Sciences, University of Washington, Seattle, USA.
Goris, J., Konstantinidis, L.T., Klappenbach, J.A., Coenye, T., Vandamme, P., and Tiedje, J.M. 2007. DNA-DNA hybridization values and their relationship to whole-genome sequence similities. Int. J. Syst. Evol. MIcrobiol.57, 81–91.
Hopwood, D.A., Bibb, M.J., Chater, K.F., Kieser, T., Bruton, C.J., Kieser, H.M., Lydiate, D.J., Smith, C.P., Ward, J.M., and Schrempf, H. 1985. Genetic Manipulation of Streptomyces: a Laboratory Manual. John Innes Foundation, Norwich, UK.
Jukes, T.H. and Cantor, C.R. 1969. Evolution of protein molecules, pp. 21–132. In Munro, H.N. (ed.), Mammalian Protein Metabolism. Academic Press, New York, USA.
Jung, Y.T., Kim, B.H., Oh, T.K., and Yoon, J.H. 2010. Pseudoruegeria lutimaris sp. nov., isolated from a tidal flat sediment, and emended description of the genus Pseudoruegeria. Int. J. Syst. Evol. Microbiol.60, 1177–1181.
Kim, Y.J. and Lee, S.D. 2019. Martelella lutilitoris sp. nov., isolated from a tidal mudflat. J. Microbiol. (In press).
Kroppenstedt, R.M. 1985. Fatty acid and menaquinone analysis of actinomycetes and related organisms, pp. 173–199. In Goodfellow, M. and Minnikin, D.E. (eds.). Chemical Methods in Bacterial Systematics, Academic Press, London, UK.
Lai, Q., Li, S., Xu, H., Jiang, L., Zhang, R., and Shio, Z. 2014. Thioclava atlantica sp. nov., isolated from deep sea sediment of the Atlantic Ocean. Antonie van Leeuwenhoek106, 919–925.
Lane, D.J. 1991. 16S/23S rRNA Sequencing, pp. 115–175. In Stackebrandt, E. and Goodfellow, M. (eds.). Nucleic Acid Techniques in Bacterial Systematics, John Wiley and Sons, London, UK.
Lee, S.D. 2018. Maribius pontilimi sp. nov., isolated from a tidal mudflat. Int. J. Syst. Evol. Microbiol.68, 353–357.
Lee, S.D. 2019. Martelella caricis sp. nov., isolated from a rhizosphere mudflat. Int. J. Syst. Evol. Microbiol.69, 266–270.
Lee, S.D., Kim, Y.J., and Kim, I.S. 2019a. Erythrobacter suaedae sp. nov., isolated from a rhizosphere mudflat of a halophyte (Suaeda japonica). Int. J. Syst. Evol. Microbiol. doi: https://doi.org/10.1099/ijsem.0.003625.
Lee, S.D., Kim, Y.J., and Kim, I.S. 2019b. Rhodococcus subtropicus sp. nov., a new actinobacterium isolated from a cave. Int. J. Syst. Evol. Microbiol. doi: https://doi.org/10.1099/ijsem.0.003601.
Lefort, V., Desper, R., and Gascuel, O. 2015. FastME 2.0: A comprehensive, accurate, and fast distance-based phylogeny inference program. Mol. Biol. Evol.32, 2798–2800.
Liu, Y., Lai, Q., Du, J., Xu, H., Jiang, L., and Shio, Z. 2015. Thioclava indica sp. nov., isolated from surface seawater of the Indian Ocean. Antonie van Leeuwenhoek107, 297–304.
Liu, Y., Lai, Q., and Shao, Z. 2017a. A multilocus sequence analysis scheme for phylogeny of Thioclava bacteria and proposal of two novel species. Front. Microbiol.8, 1322.
Liu, Y., Lai, Q., and Shao, Z. 2017b. Thioclava nitratireducens sp. nov., isolated from surface seawater. Int. J. Syst. Evol. Microbiol.67, 2109–2113.
Liu, Y., Lai, Q., Wang, W., and Shao, Z. 2017c. Defluviimonas nitratireducens sp. nov., isolated from surface seawater. Int. J. Syst. Evol. Microbiol.67, 2752–2757.
Meier-Kolthoff, J.P., Auch, A.F., Klenk, H.P., and Göker, M. 2013. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics14, 60.
Meier-Kolthoff, J.P. and Göker, M. 2019. TYGS is an automated high-throughput platform for state-of-the-art genome-based taxonomy. Nat. Commun.10, 2182.
Mesbah, M., Premachandran, U., and Whitman, W.B. 1989. Precise measurement of the G+C content of deoxyribonucleic acid by high-performance liquid chromatography. Int. J. Syst. Bacteriol.39, 159–167.
Minnikin, D.E., O’Donnell, A.G., Goodfellow, M., Alderson, G., Athalye, M., Schaal, A., and Parlett, J.H. 1984. An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J. Microbiol. Methods2, 233–241.
Minnikin, D.E., Patel, P.V., Alshamaony, L., and Goodfellow, M. 1977. Polar lipid composition in the classification of Nocardia and related bacteria. Int. J. Syst. Bacteriol.27, 104–117.
Richter, M. and Rosselló-Móra, R. 2009. Shifting the genomic gold standard for the prokaryotic species definition. Proc. Natl. Acad. Sci. USA106, 19126–19131.
Sorokin, D.Y., Tourova, T.P., Spiridonova, E.M., Rainey, F.A., and Muyzer, G. 2005. Thioclava pacifica gen. nov., sp. nov., a novel facultatively autotrophic, marine, sulfur-oxidizing bacterium from a near-shore sulfidic hydrothermal area. Int. J. Syst. Evol. Microbiol.55, 1069–1075.
Thompson, J.D., Gibson, T.J., Plewniak, F., Jeanmougin, F., and Higgins, D.G. 1997. The Clustal_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res.24, 4876–4882.
Thongphrom, C., Kim, J.H., Bora, N., and Kim, W. 2017. Thioclava arenosa sp. nov., isolated from sea sand. Int. J. Syst. Evol. Microbiol.67, 1735–1739.
Zhang, R., Lai, Q., Wang, W., Li, S., and Shao, Z. 2013. Thioclava dalianensis sp. nov., isolated from surface sea water. Int. J. Syst. Evol. Microbiol.63, 2981–2985.
Acknowledgements
This research was carried out by the project for the survey and excavation of Korean indigenous species of the National Institute of Biological Resources (NIBR) under the Ministry of Environment, Korea, and through the partial support of the National Research Foundation of Korea (no. 2019015605). The authors are thankful for Dr. J. S. Lee (KCTC) for providing the type strains of T. arenosa and T. nitratireducens.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Supporting Information
Rights and permissions
About this article
Cite this article
Kim, YJ., Lee, S.D. Hahyoungchilella caricis gen. nov., sp. nov., isolated from a rhizosphere mudflat of a halophyte (Carex scabrifolia), transfer of Thioclava arenosa Thongphrom et al. 2017 to Pseudothioclava as Pseudothioclava arenosa gen. nov., comb. nov. and proposal of Thioclava electrotropha Chang et al. 2018 as a later heterosynonym of Thioclava sediminum. J Microbiol. 57, 1048–1055 (2019). https://doi.org/10.1007/s12275-019-9260-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-019-9260-y