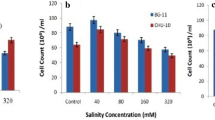
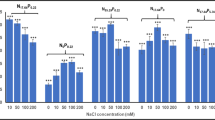
Abstract
Increased lipid accumulation of algal cells as a response to environmental stress factors attracted much attention of researchers to incorporate this stress response into industrial algal cultivation process with the aim of enhancing algal lipid productivity. This study applies high-salinity stress condition to a two-phase process in which microalgal cells are initially grown in freshwater medium until late exponential phase and subsequently subjected to high-salinity condition that induces excessive lipid accumulation. Our initial experiment revealed that the concentrated culture of Chlorella sorokiniana HS1 exhibited the intense fluorescence of Nile red at the NaCl concentration of 60 g/L along with 1 g/L of supplemental bicarbonate after 48 h of induction period without significantly compromising cultural integrity. These conditions were further verified with the algal culture grown for 7 days in a 1 L bottle reactor that reached late exponential phase; a 12% increment in the lipid content of harvested biomass was observed upon inducing high lipid accumulation in the concentrated algal culture at the density of 5.0 g DW/L. Although an increase in the sum of carbohydrate and lipid contents of harvested biomass indicated that the external carbon source supplemented during the induction period increased overall carbon assimilation, a decrease in carbohydrate content suggested the potential reallocation of cellular carbon that promoted lipid droplet formation under high-salinity stress. These results thus emphasize that the two-phase process can be successfully implemented to enhance algal lipid productivity by incorporating high-salinity stress conditions into the pre-concentrated sedimentation ponds of industrial algal production system.
Similar content being viewed by others
References
Ahmad, I. and Hellebust, J.A. 1984. Osmoregulation in the extremely euryhaline marine micro-alga Chlorella autotrophica. Plant Physiol. 74, 1010–1015.
Almshawit, H., Pouniotis, D., and Macreadie, I. 2014. Cell density impacts on Candida glabrata survival in hypo-osmotic stress. FEMS Yeast Res. 14, 508–516.
Azachi, M., Sadka, A., Fisher, M., Goldshlag, P., Gokhman, I., and Zamir, A. 2002. Salt induction of fatty acid elongase and membrane lipid modifications in the extreme halotolerant alga Dunaliella salina. Plant Physiol. 129, 1320–1329.
Bligh, E.G. and Dyer, W.J. 1959. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917.
Brennan, L. and Owende, P. 2010. Biofuels from microalgae-a review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sust. Energ. Rev. 14, 557–577.
Chen, C.Y., Yeh, K.L., Aisyah, R., Lee, D.J., and Chang, J.S. 2011. Cultivation, photobioreactor design and harvesting of microalgae for biodiesel production: a critical review. Bioresour. Technol. 102, 71–81.
Cho, D.H., Ramanan, R., Heo, J., Lee, J., Kim, B.H., Oh, H.M., and Kim, H.S. 2015. Enhancing microalgal biomass productivity by engineering a microalgal-bacterial community. Bioresour. Technol. 175, 578–585.
Cho, D.H., Ramanan, R., Kim, B.H., Lee, J., Kim, S., Yoo, C., Choi, G.G., Oh, H.M., and Kim, H.S. 2013. Novel approach for the development of axenic microalgal cultures from environmental samples. J. Phycol. 49, 802–810.
Church, J., Hwang, J.H., Kim, K.T., McLean, R., Oh, Y.K., Nam, B., Joo, J.C., and Lee, W.H. 2017. Effect of salt type and concentration on the growth and lipid content of Chlorella vulgaris in synthetic saline wastewater for biofuel production. Bioresour. Technol. 243, 147–153.
Davis, R., Aden, A., and Pienkos, P.T. 2011. Techno-economic analysis of autotrophic microalgae for fuel production. Appl. Energy 88, 3524–3531.
El-Kassas, H.Y. 2013. Growth and fatty acid profile of the marine microalga Picochlorum sp. grown under nutrient stress conditions. Egypt. J. Aquat. Res. 39, 233–239.
Fan, J., Cui, Y., Wan, M., Wang, W., and Li, Y. 2014. Lipid accumulation and biosynthesis genes response of the oleaginous Chlorella pyrenoidosa under three nutrition stressors. Biotechnol. Biofuels 7, 7–17.
González, L.E., Díaz, G.C., Aranda, D.A.G., Cruz, Y.R., and Fortes, M.M. 2015. Biodiesel production based in microalgae: a biorefinery approach. Nat. Sci. 7, 358.
Habiby, H., Afyuni, M., Khoshgoftarmanesh, A.H., and Schulin, R. 2014. Effect of preceding crops and their residues on availability of zinc in a calcareous Zn-deficient soil. Biol. Fertil Soils 50, 1061–1067.
Ho, S.H., Chen, C.N.N., Lai, Y.Y., Lu, W.B., and Chang, J.S. 2014. Exploring the high lipid production potential of a thermotolerant microalga using statistical optimization and semi-continuous cultivation. Bioresour. Technol. 163, 128–135.
Hu, Q., Sommerfeld, M., Jarvis, E., Ghirardi, M., Posewitz, M., Seibert, M., and Darzins, A. 2008. Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J. 54, 621–639.
Kang, Z., Kim, B.H., Ramanan, R., Choi, J.E., Yang, J.W., Oh, H.M., and Kim, H.S. 2015. A cost analysis of microalgal biomass and biodiesel production in open raceways treating municipal waste water and under optimum light wavelength. J. Microbiol. Biotechnol. 25, 109–118.
Kim, H.S., Guzman, A.R., Thapa, H.R., Devarenne, T.P., and Han, A. 2016a. A droplet microfluidics platform for rapid microalgal growth and oil production analysis. Biotechnol. Bioeng. 113, 1691–1701.
Kim, G.Y., Heo, J., Kim, H.S., and Han, J.I. 2017. Bicarbonate-based cultivation of Dunaliella salina for enhancing carbon utilization efficiency. Bioresour. Technol. 237, 72–77.
Kim, B.H., Ramanan, R., Kang, Z., Cho, D.H., Oh, H.M., and Kim, H.S. 2016b. Chlorella sorokiniana HS1, a novel freshwater green algal strain, grows and hyperaccumulates lipid droplets in seawater salinity. Biomass Bioenerg. 85, 300–305.
Kobayashi, M., Kurimura, Y., and Tsuji, Y. 1997. Light-independent, astaxanthin production by the green microalga Haematococcus pluvialis under salt stress. Biotechnol. Lett. 19, 507–509.
Kobayashi, N., Noel, E.A., Barnes, A., Rosenberg, J., DiRusso, C., Black, P., and Oyler, G.A. 2013. Rapid detection and quantification of triacylglycerol by HPLC-ELSD in Chlamydomonas reinhardtii and Chlorella strains. Lipids 48, 1035–1049.
Lee, J.Y., Yoo, C., Jun, S.Y., Ahn, C.Y., and Oh, H.M. 2010. Comparison of several methods for effective lipid extraction from microalgae. Bioresour. Technol. 101, S75–S77.
Lefebvre, O. and Moletta, R. 2006. Treatment of organic pollution in industrial saline wastewater: a literature review. Water Res. 40, 3671–3682.
Li, Z., Sun, M., Li, Q., Li, A., and Zhang, C. 2012. Profiling of carotenoids in six microalgae (Eustigmatophyceae) and assessment of their β-carotene productions in bubble column photobioreactor. Biotechnol. Lett. 34, 2049–2053.
Linaric, M., Markic, M., and Sipos, L. 2013. High salinity wastewater treatment. Water Sci. Technol. 68, 1400–1405.
Lohman, E.J., Gardner, R.D., Pedersen, T., Peyton, B.M., Cooksey, K.E., and Gerlach, R. 2015. Optimized inorganic carbon regime for enhanced growth and lipid accumulation in Chlorella vulgaris. Biotechnol. Biofuels 8, 82.
Mata, T.M., Martins, A.A., and Caetano, N.S. 2010. Microalgae for biodiesel production and other applications: a review. Renew. Sust. Energ. Rev. 14, 217–232.
Miller, L. and Houghton, J.A. 1945. The micro-Kjeldahl determination of the nitrogen content of amino acids and proteins. J. Biol. Chem. 169, 373–383.
Minhas, A.K., Hodgson, P., Barrow, C.J., and Adholeya, A. 2016. A review on the assessment of stress conditions for simultaneous production of microalgal lipids and carotenoids. Front. Microbiol. 7, 546.
Mulders, K.J., Lamers, P.P., Martens, D.E., and Wijffels, R.H. 2014. Phototrophic pigment production with microalgae: biological constraints and opportunities. J. Phycol. 50, 229–242.
Narala, R.R., Garg, S., Sharma, K.K., Thomas-Hall, S.R., Deme, M., Li, Y., and Schenk, P.M. 2016. Comparison of microalgae cultivation in photobioreactor, open raceway pond, and a two-stage hybrid system. Front. Energy Res. 4, 29.
Pal, D., Khozin-Goldberg, I., Cohen, Z., and Boussiba, S. 2011. The effect of light, salinity, and nitrogen availability on lipid production by Nannochloropsis sp. Appl. Microbiol. Biotechnol. 90, 1429–1441.
Pandit, P.R., Fulekar, M.H., and Karuna, M.S.L. 2017. Effect of salinity stress on growth, lipid productivity, fatty acid composition, and biodiesel properties in Acutodesmus obliquus and Chlorella vulgaris. Environ. Sci. Pollut. Res. 24, 13437–13451.
Ra, C.H., Kang, C.H., Kim, N.K., Lee, C.G., and Kim, S.K. 2015. Cultivation of four microalgae for biomass and oil production using a two-stage culture strategy with salt stress. Renew. Energy 80, 117–122.
Ramanan, R., Kim, B.H., Cho, D.H., Ko, S.R., Oh, H.M., and Kim, H.S. 2013. Lipid droplet synthesis is limited by acetate availability in starchless mutant of Chlamydomonas reinhardtii. FEBS Lett. 587, 370–377.
Rippka, R., Deruelles, J., Waterbury, J.B., Herdman, M., and Stanier, R.Y. 1979. Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Microbiology 111, 1–61.
Roberts, G.W., Fortier, M.O.P., Sturm, B.S., and Stagg-Williams, S.M. 2013. Promising pathway for algal biofuels through wastewater cultivation and hydrothermal conversion. Energ. Fuel. 27, 857–867.
Schlagermann, P., Göttlicher, G., Dillschneider, R., Rosello-Sastre, R., and Posten, C. 2012. Composition of algal oil and its potential as biofuel. J. Combust. 201, 285185.
Shurin, J.B., Abbott, R.L., Deal, M.S., Kwan, G.T., Litchman, E., Mc-Bride, R.C., Mandal, S., and Smith, V.H. 2013. Industrial-strength ecology: trade-offs and opportunities in algal biofuel production. Ecol. Lett. 16, 1393–1404.
Smith, V.H., Sturm, B.S., de Noyelles, F.J., and Billings, S.A. 2010. The ecology of algal biodiesel production. Trends Ecol. Evol. 25, 301–309.
Sung, M.G., Lee, B., Kim, C.W., Nam, K., and Chang, Y.K. 2017. Enhancement of lipid productivity by adopting multi-stage continuous cultivation strategy in Nannochloropsis gaditana. Bioresour. Technol. 229, 20–25.
Uduman, N., Qi, Y., Danquah, M.K., Forde, G.M., and Hoadley, A. 2010. Dewatering of microalgal cultures: a major bottleneck to algae-based fuels. J. Renew. Sustain. Energy 2, 012701.
Wang, H., Pampati, N., McCormick, W.M., and Bhattacharyya, L. 2016. Protein nitrogen determination by Kjeldahl digestion and ion chromatography. J. Pharm. Sci. 105, 1851–1857.
Xu, X.Q. and Beardall, J. 1997. Effect of salinity on fatty acid composition of a green microalga from an antarctic hypersaline lake. Phytochemistry 45, 655–658.
Xu, N., Zhang, X., Fan, X., Han, L., and Zeng, C. 2001. Effects of nitrogen source and concentration on growth rate and fatty acid composition of Ellipsoidion sp. (Eustigmatophyta). J. Appl. Phycol. 13, 463–469.
Yeesang, C. and Cheirsilp, B. 2011. Effect of nitrogen, salt, and iron content in the growth medium and light intensity on lipid production by microalgae isolated from freshwater sources in Thailand. Bioresour. Technol. 102, 3034–3040.
Yoo, G., Park, W.K., Kim, C.W., Choi, Y.E., and Yang, J.W. 2012. Direct lipid extraction from wet Chlamydomonas reinhardtii biomass using osmotic shock. Bioresour. Technol. 123, 717–722.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Kakarla, R., Choi, JW., Yun, JH. et al. Application of high-salinity stress for enhancing the lipid productivity of Chlorella sorokiniana HS1 in a two-phase process. J Microbiol. 56, 56–64 (2018). https://doi.org/10.1007/s12275-018-7488-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-018-7488-6