Abstract
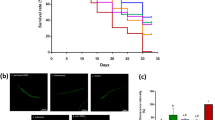
In this study, we utilized the nematode Caenorhabditis elegans to assess potential life-expanding effect of Lactobacillus salivarius strain FDB89 (FDB89) isolated from feces of centenarians in Bama County (Guangxi, China). This study showed that feeding FDB89 extended the mean life span in C. elegans by up to 11.9% compared to that of control nematodes. The reduced reproductive capacities, pharyngeal pumping rate, growth, and increased superoxide dismutase (SOD) activity and XTT reduction capacity were also observed in FDB89 feeding worms. To probe the anti-aging mechanism further, we incorporated a food gradient feeding assay and assayed the life span of eat-2 mutant. The results demonstrated that the maximal life span of C. elegans fed on FDB89 was achieved at the concentration of 1.0 mg bacterial cells/plate, which was 10-fold greater than that of C. elegans fed on E. coli OP50 (0.1 mg bacterial cells/plate). However, feeding FDB89 could not further extend the life span of eat-2 mutant. These results indicated that FDB89 modulated the longevity of C. elegans in a dietary restriction-dependent manner and expanded the understanding of anti-aging effect of probiotics.
Similar content being viewed by others
References
Braeckman, B.P., Houthoofd, K., De Vreese, A., and Vanfleteren, J.R. 2002. Assaying metabolic activity in ageing Caenorhabditis elegans. Mech. Ageing Dev. 123, 105–119.
Brown, M.K., Evans, J.L., and Luo, Y. 2006. Beneficial effects of natural antioxidants EGCG and alpha-lipoic acid on life span and age-dependent behavioral declines in Caenorhabditis elegans. Pharmacol. Biochem. Behav. 85, 620–628.
Crawford, D., Libina, N., and Kenyon, C. 2007. Caenorhabditis elegans integrates food and reproductive signals in lifespan determination. Aging Cell 6, 715–721.
Doonan, R., McElwee, J.J., Matthijssens, F., Walker, G.A., Houthoofd, K., Back, P., Matscheski, A., Vanfleteren, J.R., and Gems, D. 2008. Against the oxidative damage theory of aging: superoxide dismutases protect against oxidative stress but have little or no effect on life span in Caenorhabditis elegans. Genes Dev. 22, 3236–3241.
Fabian, T.J. and Johnson, T.E. 1994. Production of age-synchronous mass-cultures of Caenorhabditis elegans. J. Gerontol. 49, B145–B156.
Gems, D. and Doonan, R. 2009. Antioxidant defense and aging in C. elegans is the oxidative damage theory of aging wrong? Cell Cycle 8, 1681–1687.
Gruber, J., Ng, L.F., Poovathingal, S.K., and Halliwell, B. 2009. Deceptively simple but simply deceptive — Caenorhabditis elegans lifespan studies: Considerations for aging and antioxidant effects. FEBS Lett. 583, 3377–3387.
Harrington, L.A. and Harley, C.B. 1988. Effect of vitamin-E on lifespan and reproduction in Caenorhabditis elegans. Mech. Ageing Dev. 43, 71–78.
Houthoofd, K., Braeckman, B.P., Lenaerts, I., Brys, K., De Vreese, A., Van Eygen, S., and Vanfleteren, J.R. 2002a. Ageing is reversed, and metabolism is reset to young levels in recovering dauer larvae of C. elegans. Exp. Gerontol. 37, 1015–1021.
Houthoofd, K., Braeckman, B.P., Lenaerts, I., Brys, K., De Vreese, A., Van Eygen, S., and Vanfleteren, J.R. 2002b. Axenic growth up-regulates mass-specific metabolic rate, stress resistance, and extends life span in Caenorhabditis elegans. Exp. Gerontol. 37, 1371–1378.
Houthoofd, K., Braeckman, B.P., Lenaerts, I., Brys, K., De Vreese, A., Van Eygn, S., and Vanfleteren, J.R. 2002c. No reduction of metabolic rate in food restricted Caenorhabditis elegans. Exp. Gerontol. 37, 1359–1369.
Ikeda, T., Yasui, C., Hoshino, K., Arikawa, K., and Nishikawa, Y. 2007. Influence of lactic acid bacteria on longevity of Caenorhabditis elegans and host defense against Salmonella entetica serovar enteritidis. Appl. Environ. Microbiol. 73, 6404–6409.
Katewa, S.D. and Kapahi, P. 2010. Dietary restriction and aging. Aging Cell 9, 105–112.
Kimoto-Nira, H., Suzuki, C., Kobayashi, M., Sasaki, K., Kurisaki, J.I., and Mizumachi, K. 2007. Anti-ageing effect of a lactococcal strain: analysis using senescence-accelerated mice. Brit. J. Nutr. 98, 1178–1186.
Klass, M.R. 1977. Aging in the nematode Caenorhabditis elegans: Major biological and environmental factors influncing life span. Mech. Ageing Dev. 6, 413–429.
Mair, W. and Dillin, A. 2008. Aging and survival: The genetics of life span extension by dietary restriction. Annu. Rev. Biochem. 77, 727–754.
Martin, B., Golden, E., Carlson, O.D., Egan, J.M., Mattson, M.P., and Maudsley, S. 2008. Caloric restriction: Impact upon pituitary function and reproduction. Ageing Res. Rev. 7, 209–224.
Mehta, L.H. and Roth, G.S. 2009. Caloric restriction and longevity The science and the ascetic experience. In Bushell, W.C., Olivo, E.L., and Theise, N.D. (eds.), Longevity, Regeneration, and Optimal Health: Integrating Eastern and Westen Perspectives, Vol. 1172, pp. 28–33. Blackwell Publishing, Oxford, UK.
Metchnikoff, E. 1908. The microbes of intestinal putrefaction. C. R. A. Cad. Sci. 147, 579–582.
Morck, C. and Pilon, M. 2006. C. elegans feeding defective mutants have shorter body lengths and increased autophagy. BMC Dev. Biol. 6, 39.
Parvez, S., Malik, K.A., Kang, S.A., and Kim, H.Y. 2006. Probiotics and their fermented food products are beneficial for health. J. Appl. Microbiol. 100, 1171–1185.
Paull, K.D., Shoemaker, R.H., Boyd, M.R., Parsons, J.L., Risbood, P.A., Barbera, W.A., Sharma, M.N., Baker, D.C., Hand, E., Scudiero, D.A., and et al. 1988. The synthesis of XTT-a new tetrazolium reagent that is bioreducible to a water-soluble formazan. J. Heterocycl. Chem. 25, 911–914.
Pun, P.B.L., Gruber, J., Tang, S.Y., Schaffer, S., Ong, R.L.S., Fong, S., Ng, L.F., Cheah, I., and Halliwell, B. 2010. Ageing in nematodes: do antioxidants extend lifespan in Caenorhabditis elegans? Biogerontology 11, 17–30.
Ristow, M. and Schmeisser, S. 2011. Extending life span by increasing oxidative stress. Free Radical Biol. Med. 51, 327–336.
Salinas, L.S., Maldonado, E., and Navarro, R.E. 2006. Stress-induced germ cell apoptosis by a p53 independent pathway in Caenorhabditis elegans. Cell Death Differ. 13, 2129–2139.
Saul, N., Pietsch, K., Menzel, R., Sturzenbaum, S.R., and Steinberg, C.E.W. 2010. The longevity effect of tannic acid in Caenorhabditis elegans: Disposable soma meets hormesis. J. Gerontol. A. Biol. Sci. Med. Sci. 65, 626–635.
Sohal, R.S. and Weindruch, R. 1996. Oxidative stress, caloric restriction, and aging. Science 273, 59–63.
Stiernagle, T. 2006. Maintenance of C. elegans. pp. 1–11. Worm-Book: the online review of C. elegans biology, In The C. elegans Research Community. Pasadena, CA, USA.
Sulston, J. and Hodgkin, J. 1988. The nematode Caenorhabditis elegans. Methods. Cold Spring Harbor Monograph Series 17, 587–606.
Van Raamsdonk, J.M. and Hekimi, S. 2010. Reactive oxygen species and aging in Caenorhabditis elegans: Causal or casual relationship? Antioxid. Redox. Sign. 13, 1911–1953.
Vasquez, N., Suau, A., Magne, F., Pochart, P., and Pelissier, M.A. 2009. Differential effects of Bifidobacterium pseudolongum strain patronus and metronidazole in the rat gut. Appl. Environ. Microbiol. 75, 381–386.
Vina, J., Borras, C., and Miquel, J. 2007. Theories of ageing. IUBMB Life 59, 249–254.
Wang, F., Jiang, L., Liu, A.P., Guo, X.H., and Ren, F.Z. 2008. Analysis of antigenotoxicity of Lactobacillus salivarius by high performance liquid chromatography. Chin. J. Anal. Chem. 36, 740–744.
Zhang, H., Wang, Y., Liu, M., Chen, S., Zhang, H.J., Wang, Y., Liu, M.F., and Chen, S.H. 2008. Effect of lipoteichoic acid of Bifidobacterium on senile phenotypes of aging mice induced by D-galactose. Chin. J. Microecology 20, 219–221.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, Y., Zhao, L., Zheng, X. et al. Lactobacillus salivarius strain FDB89 induced longevity in Caenorhabditis elegans by dietary restriction. J Microbiol. 51, 183–188 (2013). https://doi.org/10.1007/s12275-013-2076-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-013-2076-2