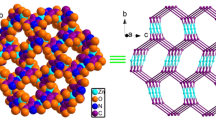
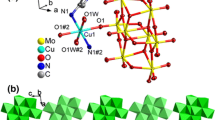
Abstract
In the development of metal-organic frameworks (MOFs), secondary building units (SBUs) have been utilized as molecular modules for the construction of nanoporous materials with robust structures. Under solvothermal synthetic conditions, dynamic changes in the metal coordination environments and ligand coordination modes of SBUs determine the resultant product structures. Alternatively, MOF phases with new topologies can also be achieved by post-synthetic treatment of as-synthesized MOFs via the introduction of acidic or basic moieties that cause the simultaneous cleavage/reformation of coordination bonds in the solid state. In this sense, we studied the solid-state transformation of two ndc-based Zn-MOFs (ndc = 1,4-naphthalene dicarboxylate) with different SBUs but the same pcu topology to another MOF with sev topology. One of the chosen MOFs with pcu nets is [Zn2(ndc)2(bpy)]n (bpy = 4,4′-bipyridine), (6Cbpy-MOF) consisting of a 6-connected pillared-paddlewheel SBU, and the other is IRMOF-7 composed of 6-connected Zn4O(COO)6 SBUs and ndc. Upon post-structural modification, these pcu MOFs were converted into the same MOF with sev topology constructed from the uncommon 7-connected Zn4O(COO)7 SBU (7C-MOF). The appropriate post-synthetic conditions for the transformation of each SBUs were systematically examined. In addition, the effect of the pillar molecules in the pillared-paddlewheel MOFs on the topology conversion was studied in terms of the linker basicity, which determines the inertness during the solid-state phase transformation. This post-synthetic modification approach is expected to expand the available methods for designing and synthesizing MOFs with controlled topologies.

Similar content being viewed by others
References
Yaghi, O. M.; Kalmutzki, M. J.; Diercks, C. S. Introduction to Reticular Chemistry: Metal-Organic Frameworks and Covalent Organic Frameworks; Wiley-VCH: Weinheim, 2019.
Kalmutzki, M. J.; Hanikel, N.; Yaghi, O. M. Secondary building units as the turning point in the development of the reticular chemistry of MOFs. Sci. Adv.2018, 4, eaat9180.
Tranchemontagne, D. J.; Mendoza-Cortés, J. L.; O’Keeffe, M.; Yaghi, O. M. Secondary building units, nets and bonding in the chemistry of metal-organic frameworks. Chem. Soc. Rev.2009, 38, 1257–1283.
Ha, J. S.; Lee, J. H.; Moon, H. R. Alterations to secondary building units of metal-organic frameworks for the development of new functions. Inorg. Chem. Front.2020, 7, 12–27.
Lee, J. H.; Jeoung, S.; Chung, Y. G.; Moon, H. R. Elucidation of flexible metal-organic frameworks: Research progresses and recent developments. Coord. Chem. Rev.2019, 389, 161–188.
Dighe, A. V.; Nemade, R. Y.; Singh, M. R. Modeling and simulation of crystallization of metal-organic frameworks. Processes2019, 7, 527.
Aggarwal, H.; Bhatt, P. M.; Bezuidenhout, C. X.; Barbour, L. J. Direct evidence for single-crystal to single-crystal switching of degree of interpenetration in a metal-organic framework. J. Am. Chem. Soc.2014, 136, 3776–3779.
Wei, R. J.; Huo, Q.; Tao, J.; Huang, R. B.; Zheng, L. S. Spin-crossover FeII4 squares: Two-step complete spin transition and reversible single-crystal-to-single-crystal transformation. Angew. Chem., Int. Ed.2011, 50, 8940–8943.
Wang, X. P.; Chen, W. M.; Qi, H.; Li, X. Y.; Rajnák, C.; Feng, Z. Y.; Kurmoo, M.; Boča, R.; Jia, C. J.; Tung, C. H. et al. Solvent-controlled phase transition of a CoII-organic framework: From achiral to chiral and two to three dimensions. Chem.—Eur. J.2017, 23, 7990–7996.
Yan, Z. H.; Li, X. Y.; Liu, L. W.; Yu, S. Q.; Wang, X. P.; Sun, D. Single-crystal to single-crystal phase transition and segmented thermochromic luminescence in a dynamic 3D interpenetrated AgI coordination network. Inorg. Chem.2016, 55, 1096–1101.
Chaemchuen, S.; Zhou, K.; Yusubov, M. S.; Postnikov, P. S.; Klomkliang, N.; Verpoort, F. Solid-state transformation in porous metal-organic frameworks based on polymorphic-pillared net structure: Generation of tubular shaped MOFs. Micro. Meso. Mater.2019, 278, 99–104.
Schweighauser, L.; Harano, K.; Nakamura, E. Experimental study on interconversion between cubic MOF-5 and square MOF-2 arrays. Inorg. Chem. Commun.2017, 84, 1–4.
Xing, J. F.; Schweighauser, L.; Okada, S.; Harano, K.; Nakamura, E. Atomistic structures and dynamics of prenucleation clusters in MOF-2 and MOF-5 syntheses. Nat. Commun.2019, 10, 3068.
McKinstry, C.; Cussen, E. J.; Fletcher, A. J.; Patwardhan, S. V.; Sefcik, J. Effect of synthesis conditions on formation pathways of metal organic framework (MOF-5) crystals. Cryst. Growth Des.2013, 13, 5481–5486.
Kim, J.; Dolgos, M. R.; Lear, B. J. Isolation and chemical transformations involving a reactive intermediate of MOF-5. Cryst. Growth Des.2015, 15, 4781–4786.
Iannaccone, G.; Bernardi, A.; Suriano, R.; Bianchi, C. L.; Levi, M.; Turri, S.; Griffini, G. The role of sol-gel chemistry in the low-temperature formation of ZnO buffer layers for polymer solar cells with improved performance. RSC Adv.2016, 6, 46915–46924.
Yeh, C. C.; Liu, H. C.; Heni, W.; Berling, D.; Zan, H. W.; Soppera, O. Chemical and structural investigation of zinc-oxo cluster photoresists for DUV lithography. J. Mater. Chem. C2017, 5, 2611–2619.
Hirai, K.; Reboul, J.; Morone, N.; Heuser, J. E.; Furukawa, S.; Kitagawa, S. Diffusion-coupled molecular assembly: Structuring of coordination polymers across multiple length scales. J. Am. Chem. Soc.2014, 136, 14966–14973.
Lee, S. J.; Doussot, C.; Baux, A.; Liu, L. J.; Jameson, G. B.; Richardson, C.; Pak, J. J.; Trousselet, F.; Coudert, F. X.; Telfer, S. G. Multicomponent metal-organic frameworks as defect-tolerant materials. Chem. Mater.2016, 28, 368–375.
Beddoe, S. V.; Lonergan, R. F.; Pitak, M. B.; Price, J. R.; Coles, S. J.; Kitchen, J. A.; Keene, T. D. All about that base: Investigating the role of ligand basicity in pyridyl complexes derived from a copper-Schiff base coordination polymer. Dalton Trans.2019, 48, 15553–15559.
Karagiaridi, O.; Bury, W.; Tylianakis, E.; Sarjeant, A. A.; Hupp, J. T.; Farha, O. K. Opening metal-organic frameworks vol. 2: Inserting longer pillars into pillared-paddlewheel structures through solventassisted linker exchange. Chem. Mater.2013, 25, 3499–3503.
Burnett, B. J.; Choe, W. Stepwise pillar insertion into metal-organic frameworks: A sequential self-assembly approach. CrystEngComm2012, 14, 6129–6131.
Jeong, S.; Kim, D.; Song, X. K.; Choi, M.; Park, N.; Lah, M. S. Postsynthetic exchanges of the pillaring ligand in three-dimensional metal-organic frameworks. Chem. Mater.2013, 25, 1047–1054.
Misono, M. Heterogeneous Catalysis of Mixed Oxides: Perovskite and Heteropoly Catalysts; Elsevier: Oxford, 2013.
Pan, Y.; Ding, Q. J.; Xu, H. J.; Shi, C. Y.; Singh, A.; Kumar, A.; Liu, J. Q. A new Zn(II)-based 3D metal-organic framework with uncommon sev topology and its photocatalytic properties for the degradation of organic dyes. CrystEngComm2019, 21, 4578–4585.
Bai, S. Z.; Zhang, W. Q.; Ling, Y.; Yang, F. L.; Deng, M. L.; Chen, Z. X.; Weng, L. H.; Zhou, Y. M. Predicting and creating 7-connected Zn4O vertices for the construction of an exceptional metal-organic framework with nanoscale cages. CrystEngComm2015, 17, 1923–1926.
Duan, J. G.; Higuchi, M.; Kitagawa, S. Predesign and systematic synthesis of 11 highly porous coordination polymers with unprecedented topology. Inorg. Chem.2015, 54, 1645–1649.
Qiu, Y. C.; Yuan, S.; Li, X. X.; Du, D. Y.; Wang, C.; Qin, J. S.; Drake, H. F.; Lan, Y. Q.; Jiang, L.; Zhou, H. C. Face-sharing archimedean solids stacking for the construction of mixed-ligand metal-organic frameworks. J. Am. Chem. Soc.2019, 141, 13841–13848.
He, W. W.; Li, S. L.; Yang, G. S.; Lan, Y. Q.; Su, Z. M.; Fu, Q. Controllable synthesis of a non-interpenetrating microporous metal-organic framework based on octahedral cage-like building units for highly efficient reversible adsorption of iodine. Chem. Commun.2012, 48, 10001–10003.
Yu, D. B.; Shao, Q.; Song, Q. J.; Cui, J. W.; Zhang, Y. L.; Wu, B.; Ge, L.; Wang, Y.; Zhang, Y.; Qin, Y. Q. et al. A solvent-assisted ligand exchange approach enables metal-organic frameworks with diverse and complex architectures. Nat. Commun.2020, 11, 927.
Swain, M. Chemicalize.org. J. Chem. Inf. Model.2012, 52, 613–615.
Chen, B. L.; Liang, C. D.; Yang, J.; Contreras, D. S.; Clancy, Y. L.; Lobkovsky, E. B.; Yaghi, O. M.; Dai, S. A microporous metal-organic framework for gas-chromatographic separation of alkanes. Angew. Chem., Int. Ed.2006, 45, 1390–1393.
Chun, H.; Dybtsev, D. N.; Kim, H.; Kim, K. Synthesis, X-ray crystal structures, and gas sorption properties of pillared square grid nets based on paddle-wheel motifs: Implications for hydrogen storage in porous materials. Chem.—Eur. J.2005, 11, 3521–3529.
Shekhah, O.; Wang, H.; Paradinas, M.; Ocal, C.; Schüpbach, B.; Terfort, A.; Zacher, D.; Fischer, R. A.; Wöll, C. Controlling interpenetration in metal-organic frameworks by liquid-phase epitaxy. Nat. Mater.2009, 8, 481–484.
Jiang, H. L.; Makal, T. A.; Zhou, H. C. Interpenetration control in metal-organic frameworks for functional applications. Coord. Chem. Rev.2013, 257, 2232–2249.
Ding, M. L.; Cai, X. C.; Jiang, H. L. Improving MOF stability: Approaches and applications. Chem. Sci.2019, 10, 10209–10230.
Kong, L. D.; Zou, R. Y.; Bi, W. Z.; Zhong, R. Q.; Mu, W. J.; Liu, J.; Han, R. P. S.; Zou, R. Q. Selective adsorption of CO2/CH4 and CO2/N2 within a charged metal-organic framework. J. Mater. Chem. A2014, 2, 17771–17778.
Shang, J.; Li, G.; Singh, R.; Gu, Q. F.; Nairn, K. M.; Bastow, T. J.; Medhekar, N.; Doherty, C. M.; Hill, A. J.; Liu, J. Z. et al. Discriminative separation of gases by a “molecular trapdoor” mechanism in chabazite zeolites. J. Am. Chem. Soc.2012, 134, 19246–19253.
Guha, S.; Saha, S. Fluoride ion sensing by an anion-π interaction. J. Am. Chem. Soc.2010, 132, 17674–17677.
Hosono, N.; Terashima, A.; Kusaka, S.; Matsuda, R.; Kitagawa, S. Highly responsive nature of porous coordination polymer surfaces imaged by in situ atomic force microscopy. Nat. Chem.2019, 11, 109–116.
Furukawa, S; Hirai, K.; Takashima, Y.; Nakagawa, K.; Kondo, M.; Tsuruoka, T.; Sakata, O.; Kitagawa, S. A block PCP crystal: Anisotropic hybridization of porous coordination polymers by face-selective epitaxial growth. Chem. Commun.2009, 5097–5099.
Rosi, N. L.; Eckert, J.; Eddaoudi, M.; Vodak, D. T.; Kim, J.; O’Keeffe, M.; Yaghi, O. M. Hydrogen storage in microporous metal-organic frameworks. Science2003, 300, 1127–1129.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) Grant funded by the Ministry of Science and ICT (Nos. NRF-2016R1A5A1009405, NRF-2019M3E6A1103980, and NRF-2019R1A6A3A01096867).
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Kim, J., Ha, J., Lee, J.H. et al. Solid-state phase transformations toward a metal-organic framework of 7-connected Zn4O secondary building units. Nano Res. 14, 411–416 (2021). https://doi.org/10.1007/s12274-020-2873-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-020-2873-y