Abstract
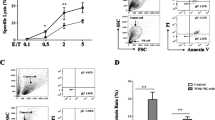
The susceptibility of cancer cells to natural killer (NK) cell–mediated cytotoxicity depends on the balance of activating and inhibitory ligands expressed on their surface. Although many types of cancer cells are killed by NK cells, non-small-cell lung cancer (NSCLC) cells are relatively resistant to NK cell–mediated cytotoxicity. In this study, we showed that several NSCLC cell lines have differential sensitivity to NK cell–mediated cytotoxicity: NCI-H522 cells were highly sensitive, but A549, NCI-H23, NCI-H1915, and NCI-H1299 were resistant. Among activating ligands such as CD48, HLA-A/B/G, ICAM-1, MICA/B, and ULBPs, only CD48 rendered NCI-H522 cells susceptible to NK cell–mediated cytotoxicity, which was proved by using CD48 siRNA and neutralizing antibody. CD48-positive NCI-H522 cells established a more stable contact with NK cells than did CD48-negative A549 and CD48 siRNA cell–transfected NCI-H522 cells. Taken together, these data demonstrate that CD48-positive NSCLC cells might be susceptible to NK cell–mediated cytotoxicity, which provide information on how to stratify NSCLC patients potentially responsive to NK-cell therapy.





Similar content being viewed by others
References
Abel AM, Yang C, Thakar MS, Malarkannan S (2018) Natural killer cells: development, maturation, and clinical utilization. Front Immunol 9:1869. https://doi.org/10.3389/fimmu.2018.01869
Arulanandam AR, Moingeon P, Concino MF, Recny MA, Kato K, Yagita H, Koyasu S, Reinherz EL (1993) A soluble multimeric recombinant CD2 protein identifies CD48 as a low affinity ligand for human CD2: divergence of CD2 ligands during the evolution of humans and mice. J Exp Med 177:1439–1450. https://doi.org/10.1084/jem.177.5.1439
Bae EA, Seo H, Kim IK, Jeon I, Kang CY (2019) Roles of NKT cells in cancer immunotherapy. Arch Pharm Res 42:543–548.
Bradley M, Zeytun A, Rafi-Janajreh A, Nagarkatti PS, Nagarkatti M (1998) Role of spontaneous and interleukin-2-induced natural killer cell activity in the cytotoxicity and rejection of Fas+ and Fas- tumor cells. Blood 92:4248–4255.
Brown MH, Boles K, Van Der Merwe PA, Kumar V, Mathew PA, Barclay AN (1998) 2B4, the natural killer and T cell immunoglobulin superfamily surface protein, is a ligand for CD48. J Exp Med 188:2083–2090. https://doi.org/10.1084/jem.188.11.2083
Campbell KS, Hasegawa J (2013) Natural killer cell biology: an update and future directions. J Allergy Clin Immunol 132:536–544. https://doi.org/10.1016/j.jaci.2013.07.006
Chlewicki LK, Velikovsky CA, Balakrishnan V, Mariuzza RA, Kumar V (2008) Molecular basis of the dual functions of 2B4 (CD244). J Immunol 180:8159–8167. https://doi.org/10.4049/jimmunol.180.12.8159
Claus M, Wingert S, Watzl C (2016) Modulation of natural killer cell functions by interactions between 2B4 and CD48 in cis and in trans. Open Biol. https://doi.org/10.1098/rsob.160010
Garni-Wagner BA, Purohit A, Mathew PA, Bennett M, Kumar V (1993) A novel function-associated molecule related to non-MHC-restricted cytotoxicity mediated by activated natural killer cells and T cells. J Immunol 151:60–70. PMID: 8326140
Hamilton G, Plangger A (2021) The impact of NK cell-based therapeutics for the treatment of lung cancer for biologics: targets and therapy. Biologics 15:265–277. https://doi.org/10.2147/BTT.S290305
Heng TS, Painter MW, Immunological Genome Project C (2008) The Immunological Genome Project: networks of gene expression in immune cells. Nat Immunol 9:1091–1094. https://doi.org/10.1038/ni1008-1091
Hoffmann SC, Cohnen A, Ludwig T, Watzl C (2011) 2B4 engagement mediates rapid LFA-1 and actin-dependent NK cell adhesion to tumor cells as measured by single cell force spectroscopy. J Immunol 186:2757–2764. https://doi.org/10.4049/jimmunol.1002867
Hosen N, Ichihara H, Mugitani A, Aoyama Y, Fukuda Y, Kishida S, Matsuoka Y, Nakajima H, Kawakami M, Yamagami T, Fuji S, Tamaki H, Nakao T, Nishida S, Tsuboi A, Iida S, Hino M, Oka Y, Oji Y, Sugiyama H (2012) CD48 as a novel molecular target for antibody therapy in multiple myeloma. Br J Haematol 156:213–224. https://doi.org/10.1111/j.1365-2141.2011.08941.x
Kim JS, Shin BR, Lee HK, Lee JH, Kim KH, Choi JE, Ji AY, Hong JT, Kim Y, Han SB (2017) Cd226(-/-) natural killer cells fail to establish stable contacts with cancer cells and show impaired control of tumor metastasis in vivo. Oncoimmunology 6:e1338994. https://doi.org/10.1080/2162402X.2017.1338994
Kim J, Kim JS, Lee HK, Kim HS, Park EJ, Choi JE, Choi YJ, Shin BR, Kim EY, Hong JT, Kim Y, Han SB (2018) CXCR3-deficient natural killer cells fail to migrate to B16F10 melanoma cells. Int Immunopharmacol 63:66–73. https://doi.org/10.1016/j.intimp.2018.07.026
Kim HS, Lee JS, Lee HK, Park EJ, Jeon HW, Kang YJ, Lee TY, Kim KS, Bae SC, Park JH, Han SB (2019a) Mesenchymal stem cells ameliorate renal inflammation in adriamycin-induced nephropathy. Immune Netw 19:e36. https://doi.org/10.4110/in.2019.19.e36
Kim JS, Kim B, Lee HK, Kim HS, Park EJ, Choi YJ, Ahn GB, Yun J, Hong JT, Kim Y, Han SB (2019b) Characterization of morphological changes of B16 melanoma cells under natural killer cell attack. Int Immunopharmacol 67:366–371. https://doi.org/10.1016/j.intimp.2018.12.037
Kim N, Lee HH, Lee HJ, Choi WS, Lee J, Kim HS (2019c) Natural killer cells as a promising therapeutic target for cancer immunotherapy. Arch Pharm Res 42:591–606. https://doi.org/10.1007/s12272-019-01143-y
Klingemann H, Boissel L, Toneguzzo F (2016) Natural killer cells for immunotherapy—advantages of the NK-92 cell line over blood NK cells. Front Immunol 7:91. https://doi.org/10.3389/fimmu.2016.00091
Lee JK (2020) Sesamolin promotes cytolysis and migration activity of natural killer cells via dendritic cells. Arch Pharm Res 43:462–474. https://doi.org/10.1007/s12272-020-01229-y
Lee KM, Mcnerney ME, Stepp SE, Mathew PA, Schatzle JD, Bennett M, Kumar V (2004) 2B4 acts as a non-major histocompatibility complex binding inhibitory receptor on mouse natural killer cells. J Exp Med 199:1245–1254. https://doi.org/10.1084/jem.20031989
Lee HK, Kim EY, Kim HS, Park EJ, Lee HJ, Lee TY, Kim KS, Bae SC, Hong JT, Kim Y, Han SB (2020a) Effect of human mesenchymal stem cells on xenogeneic T and B cells isolated from lupus-prone MRL.Fas (lpr) mice. Stem Cells Int 2020:5617192. https://doi.org/10.1155/2020/5617192
Lee HK, Kim HS, Pyo M, Park EJ, Jang S, Jun HW, Lee TY, Kim KS, Bae SC, Kim Y, Hong JT, Yun J, Han SB (2020b) Phorbol ester activates human mesenchymal stem cells to inhibit B cells and ameliorate lupus symptoms in MRL.Fas (lpr) mice. Theranostics 10:10186–10199. https://doi.org/10.7150/thno.46835
Lin M, Luo H, Liang S, Chen J, Liu A, Niu L, Jiang Y (2020) Pembrolizumab plus allogeneic NK cells in advanced non-small cell lung cancer patients. J Clin Investig 130:2560–2569. https://doi.org/10.1172/JCI132712
Low MG, Saltiel AR (1988) Structural and functional roles of glycosyl-phosphatidylinositol in membranes. Science 239:268–275. https://doi.org/10.1126/science.3276003
Mcardel SL, Terhorst C, Sharpe AH (2016) Roles of CD48 in regulating immunity and tolerance. Clin Immunol 164:10–20. https://doi.org/10.1016/j.clim.2016.01.008
Minetto P, Guolo F, Pesce S, Greppi M, Obino V, Ferretti E, Sivori S, Genova C, Lemoli RM, Marcenaro E (2019) Harnessing NK cells for cancer treatment. Front Immunol 10:2836. https://doi.org/10.3389/fimmu.2019.02836
Multhoff G, Seier S, Stangl S, Sievert W, Shevtsov M, Werner C, Pockley AG, Blankenstein C, Hildebrandt M, Offner R, Ahrens N, Kokowski K, Hautmann M, Rodel C, Fietkau R, Lubgan D, Huber R, Hautmann H, Duell T, Molls M, Specht H, Haller B, Devecka M, Sauter A, Combs SE (2020) Targeted natural killer cell-based adoptive immunotherapy for the treatment of patients with NSCLC after radiochemotherapy: a randomized Phase II Clinical Trial. Clin Cancer Res 26:5368–5379. https://doi.org/10.1158/1078-0432.CCR-20-1141
Phung CD, Tran TH, Kim JO (2020) Engineered nanoparticles to enhance natural killer cell activity towards onco-immunotherapy: a review. Arch Pharm Res 43:32–45. https://doi.org/10.1007/s12272-020-01218-1
Pockley AG, Vaupel P, Multhoff G (2020) NK cell-based therapeutics for lung cancer. Expert Opin Biol Ther 20:23–33. https://doi.org/10.1080/14712598.2020.1688298
Poznanski SM, Ritchie TM, Fan IY, El-Sayes A, Portillo AL, Ben-Avi R, Rojas EA, Chew MV, Shargall Y, Ashkar AA (2021) Expanded human NK cells from lung cancer patients sensitize patients’ PDL1-negative tumors to PD1-blockade therapy. J Immunother Cancer. https://doi.org/10.1136/jitc-2020-001933
Roda-Navarro P, Mittelbrunn M, Ortega M, Howie D, Terhorst C, Sanchez-Madrid F, Fernandez-Ruiz E (2004) Dynamic redistribution of the activating 2B4/SAP complex at the cytotoxic NK cell immune synapse. J Immunol 173:3640–3646. https://doi.org/10.4049/jimmunol.173.6.3640
Ryu D, Lee JH, Kwak MK (2020) NRF2 level is negatively correlated with TGF-beta1-induced lung cancer motility and migration via NOX4-ROS signaling. Arch Pharm Res 43:1297–1310. https://doi.org/10.1007/s12272-020-01298-z
Sanchez-Correa B, Morgado S, Gayoso I, Bergua JM, Casado JG, Arcos MJ, Bengochea ML, Duran E, Solana R, Tarazona R (2011) Human NK cells in acute myeloid leukaemia patients: analysis of NK cell-activating receptors and their ligands. Cancer Immunol Immunother 60:1195–1205. https://doi.org/10.1007/s00262-011-1050-2
Sanchez-Correa B, Gayoso I, Bergua JM, Casado JG, Morgado S, Solana R, Tarazona R (2012) Decreased expression of DNAM-1 on NK cells from acute myeloid leukemia patients. Immunol Cell Biol 90:109–115. https://doi.org/10.1038/icb.2011.15
Schatzle JD, Sheu S, Stepp SE, Mathew PA, Bennett M, Kumar V (1999) Characterization of inhibitory and stimulatory forms of the murine natural killer cell receptor 2B4. Proc Natl Acad Sci USA 96:3870–3875. https://doi.org/10.1073/pnas.96.7.3870
Shevtsov M, Pitkin E, Ischenko A, Stangl S, Khachatryan W, Galibin O, Edmond S, Lobinger D, Multhoff G (2019) Ex vivo Hsp70-activated NK cells in combination with PD-1 inhibition significantly increase overall survival in preclinical models of glioblastoma and lung cancer. Front Immunol 10:454. https://doi.org/10.3389/fimmu.2019.00454
Sun L, Gang X, Li Z, Zhao X, Zhou T, Zhang S, Wang G (2021) Advances in understanding the roles of CD244 (SLAMF4) in immune regulation and associated diseases. Front Immunol 12:648182. https://doi.org/10.3389/fimmu.2021.648182
Talanian RV, Yang X, Turbov J, Seth P, Ghayur T, Casiano CA, Orth K, Froelich CJ (1997) Granule-mediated killing: pathways for granzyme B-initiated apoptosis. J Exp Med 186:1323–1331. https://doi.org/10.1084/jem.186.8.1323
Tangye SG, Lazetic S, Woollatt E, Sutherland GR, Lanier LL, Phillips JH (1999) Cutting edge: human 2B4, an activating NK cell receptor, recruits the protein tyrosine phosphatase SHP-2 and the adaptor signaling protein SAP. J Immunol 162:6981–6985. PMID: 10358138
Tonn T, Schwabe D, Klingemann HG, Becker S, Esser R, Koehl U, Suttorp M, Seifried E, Ottmann OG, Bug G (2020) Treatment of patients with advanced cancer with the natural killer cell line NK-92. Cytotherapy 15(12):1563–1570. https://doi.org/10.1016/j.jcyt.2013.06.017
Tripathi SK, Rengasamy KRR, Biswal BK (2020) Plumbagin engenders apoptosis in lung cancer cells via caspase-9 activation and targeting mitochondrial-mediated ROS induction. Arch Pharm Res 43:242–256. https://doi.org/10.1007/s12272-020-01221-6
Vaidya SV, Stepp SE, Mcnerney ME, Lee JK, Bennett M, Lee KM, Stewart CL, Kumar V, Mathew PA (2005) Targeted disruption of the 2B4 gene in mice reveals an in vivo role of 2B4 (CD244) in the rejection of B16 melanoma cells. J Immunol 174:800–807. https://doi.org/10.4049/jimmunol.174.2.800
Veillette A (2010) SLAM-family receptors: immune regulators with or without SAP-family adaptors. Cold Spring Harb Perspect Biol 2:a002469. https://doi.org/10.1101/cshperspect.a002469
Wang Z, Xiao Y, Guan W, Wang M, Chen J, Zhang L, Li Y, Xiong Q, Wang H, Wang M, Li Y, Lv N, Li Y, Wang L, Yu L (2020) Acute myeloid leukemia immune escape by epigenetic CD48 silencing. Clin Sci 134:261–271. https://doi.org/10.1042/CS20191170
Wang Z, Guan W, Wang M, Chen J, Zhang L, Xiao Y, Wang L, Li Y, Yu L (2021) AML1-ETO inhibits acute myeloid leukemia immune escape by CD48. Leuk Lymphoma 62:937–943. https://doi.org/10.1080/10428194.2020.1849680
Xie S, Wu Z, Niu L, Chen J, Ma Y, Zhang M (2019) Preparation of highly activated natural killer cells for advanced lung cancer therapy. Onco Targets Ther 12:5077–5086. https://doi.org/10.2147/OTT.S201924
Yang S, Cao B, Zhou G, Zhu L, Wang L, Zhang L, Kwok HF, Zhang Z, Zhao Q (2020) Targeting B7–H3 immune checkpoint with chimeric antigen receptor-engineered natural killer cells exhibits potent cytotoxicity against non-small cell lung cancer. Front Pharmacol 11:1089. https://doi.org/10.3389/fphar.2020.01089
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grants funded by the Korea government (MSIT) (2017R1A5A2015541 and 2020R1A2C2004200).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 4825 kb) Movie S1. NK-92 cell cytotoxicity against A549 cells. Cells were imaged every 2 min for 8 h (magnification, 100×; accelerated 1,200× over real time).
Supplementary file2 (MP4 15564 kb) Movie S2. NK-92 cell cytotoxicity against NCI-H522 cells. Cells were imaged every 2 min for 8 h (magnification, 100×; accelerated 1,200× over real time).
Supplementary file3 (MP4 6249 kb) Movie S3. NK-92 cell cytotoxicity against NCI-H522 cells transfected with CD48 siRNA. Cells were imaged every 2 min for 8 h (magnification, 100×; accelerated 1,200× over real time).
Supplementary file4 (MP4 4596 kb) Movie S4. NK-92 cell cytotoxicity against NCI-H522 cells in the presence of anti-2B4 blocking antibodies. Cells were imaged every 2 min for 8 h (magnification, 100×; accelerated 1,200× over real time).
Supplementary file5 (MP4 2188 kb) Movie S5. Non-killing contacts between NK-92 cells and NCI-H522 cells. Cells were imaged every 2 min for 8 h (magnification, 100×; electronically zoomed; accelerated 1,200× over real time).
Supplementary file6 (MP4 1155 kb) Movie S6. Killing contacts between NK-92 cells and NCI-H522 cells. NK-92 cells and NCI-H522 cells were imaged every 2 min for 8 h (magnification, 100 ×; electronically zoomed; accelerated 1,200x over real-time).
Supplementary file7 (MP4 2946 kb) Movie S7. Stable contacts between NK-92 cells and NCI-H522 cells. NK-92 cells and NCI-H522 cells were imaged every 2 min for 8 h (magnification, 100 ×; electronically zoomed; accelerated 1,200x over real-time).
Supplementary file8 (MP4 2183 kb) Movie S8. Unstable contacts between NK-92 cells and NCI-H522 cells. NK-92 cells and NCI-H522 cells were imaged every 2 min for 8 h (magnification, 100 ×; electronically zoomed; accelerated 1,200x over real-time).
Rights and permissions
About this article
Cite this article
Park, E.J., Jun, H.W., Na, I.H. et al. CD48-expressing non-small-cell lung cancer cells are susceptible to natural killer cell–mediated cytotoxicity. Arch. Pharm. Res. 45, 1–10 (2022). https://doi.org/10.1007/s12272-021-01365-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-021-01365-z